Miscellaneous Insect Pests of Florida Sugarcane
The insects and mites discussed in this publication are occasional pests of sugarcane in Florida or are arthropods of concern although their pest status has not been determined. They have received less research interest and less is known about them, because they have not been as important as other sugarcane pests such as the corn wireworm (Melanotus communis) or the sugarcane borer (Diatraea saccharalis). However, these arthropods have the potential to become a problem if conditions favor an outbreak.
American Grasshopper
The American grasshopper (Figure 1), Schistocerca americana, is a birdwing grasshopper that occasionally causes serious injury to sugarcane. This grasshopper eats leaf blades down to the midrib in jagged, irregular patterns, removing valuable photosynthetic tissue from the plant (Figure 2). The insect has been common in southern Florida since 2009 and has been more commonly found feeding on sugarcane grown on the sandy soils west and southwest of Lake Okeechobee. The eggs are deposited in groups in the soil. Nymphs emerge to feed on many different plants throughout the landscape. Current status as a pest of sugarcane has not been determined.

Credit: Gregg Nuessly, UF/IFAS

Credit: Gregg Nuessly, UF/IFAS
Diaprepes Root Weevil
The Diaprepes root weevil, Diaprepes abbreviatus, is invasive to Florida and is an important pest of sugarcane in Barbados and Puerto Rico. This insect is a root weevil native to the Caribbean and was first reported during the 1960s in Florida, where it damaged citrus and ornamental plants. Although this root weevil was observed in or near Florida sugarcane areas, damage to Florida sugarcane was not observed until 2010. Eggs are yellowish white and are deposited between leaves cemented together by the female (Figure 3). Larvae are white and look like grubs but have no legs. Larvae feed on roots and tunnel into the stool and belowground stalks (Figure 4). Adult coloration is variable, ranging from all black with a few white to orange markings to nearly all white to orange with black markings (Figure 5).

Credit: Alvin Wilson, UF/IFAS

Credit: Alvin Wilson, UF/IFAS

Credit: Alvin Wilson, UF/IFAS
Injury is similar to grub injury whereby sugarcane plants suffer yellow discoloration, become highly stunted, and are prone to lodging. Damage has been significant locally with stalk lodging, exposed root balls, and stand loss (Figure 6). Several weed species found in Florida sugarcane fields are suitable food sources and egg-laying sites for Diaprepes root weevils. These include the Brazilian pepper tree, a highly invasive woody plant found throughout central and southern Florida, including around sugarcane fields. Weed control is important in preventing Diaprepes root weevil infestations in sugarcane.

Credit: Alvin Wilson, UF/IFAS
Grass Leaf Tier
Larvae of the grass leaf tier, Marasmia trapezalis, feed within the youngest leaves at the top of sugarcane plants. The larvae also feed on sorghum, corn, and rice. They tie the edges of the leaves together with silk to form a protected environment to feed and molt (Figure 7). It is not uncommon during the spring months to find the youngest leaf tied together with a dead terminal end (Figure 8); however, this injury is not known to affect long-term growth or overall crop yield.

Credit: Gregg Nuessly UF/IFAS

Credit: Gregg Nuessly UF/IFAS
Spider Mites
Spider mites, primarily Oligonychus stickneyi, have been occasional pests in Florida sugarcane since the 1970s. These mites live and feed on the undersides of leaves. They form fine webs in which eggs are laid and young nymphs develop (Figure 9). Leaves infested by spider mites display an orange to reddish-brown discoloration (red russetting) similar to that associated with sugarcane lacebugs. Severe injury by spider mites can result in leaf death.
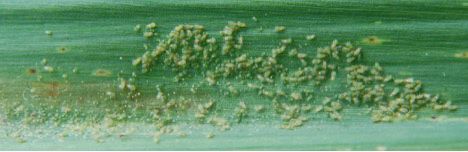
Credit: Gregg Nuessly, UF/IFAS
Spider mite infestations generally occur during March-June. The lower leaves of sugarcane are usually colonized first. Prolonged heavy infestations accompanied by extensive injury to the middle and upper leaves (Figure 10) of young plants may reduce growth. Predaceous mites, a predaceous thrips, and rainfall help control spider mites. Products with elemental sulfur are registered for use in sugarcane and may control spider mites. Multiple applications would be required to try to bring the mites under control, particularly because the mites are found predominantly under the leaves, rather than on the top of the leaves where most of the applied sulfur would be deposited.

Credit: Gregg Nuessly, UF/IFAS
Sugarcane spider mite injury to the lower leaves results in stripes of green healthy leaves alternated with stripes of yellow to bronzed leaves between the planted rows as viewed from the field edge (Figure 11). Injury to upper-canopy leaves results in a bronzed appearance across the entire infested portion of the field. Growers should look carefully for mites if they suspect an infestation, because substantial injury does not necessarily mean mites are still present.

Credit: Gregg Nuessly, UF/IFAS
Sugarcane Aphid
The sugarcane aphid, Melanaphis sacchari, was first observed in Florida in 1977. The aphid, sometimes referred to as the white sugarcane aphid, is dirty white or light gray to very light tan in color with very short cornicles (Figure 12) and feeds mostly on the undersurface of sugarcane leaves. Feeding does not result in yellow to purple areas on the leaf as with the yellow sugarcane aphid. However, sugarcane aphids produce large amounts of honeydew that collect mostly on the upper leaf surfaces and provide a substrate for sooty mold fungi. The fungal growth blocks sunlight and reduces photosynthesis, thereby negatively affecting yield. The sugarcane aphid also transmits viruses that reduce yield (e.g., Sugarcane yellow leaf virus). Large sugarcane aphid colonies are not common on sugarcane in Florida because the aphid is held under control by the same predators as for the yellow sugarcane aphid, as well as by a parasitic wasp (Lysiphlebus testaceipes) and fungal pathogens.

Credit: Julien Beuzelin, UF/IFAS
Sugarcane Delphacid
The sugarcane delphacid, Perkinsiella saccharicida (Figure 13), was first observed in Florida in 1982. Surveys quickly revealed that the delphacid ranged throughout the Florida sugarcane production area. To date, little economic damage from the pest has been reported. However, if a rapid population expansion were to occur, there could be serious economic damage due to its feeding and reproductive activities. The sugarcane delphacid is the vector of Fiji disease; however, the disease is not present in Florida.

Credit: Julien Beuzelin, UF/IFAS
Females deposit eggs into the leaf tissue. Two to twelve eggs are laid in a cluster on either side of the leaf, mainly along the midrib, leaf sheaths, and stalk internodes. The egg is 1.0 mm by 0.35 mm, elongate-cylindrical, and slightly curved. The narrow end of the egg protrudes just above the surface of the leaf and is covered with a white waxy material secreted by the female.
A female can lay up to 300 eggs during her normal 30-day life span. The incubation period varies from 2–3 weeks. The newly hatched nymph is pale and wingless, and it starts feeding shortly after hatching. The nymphal stage has five instars and lasts approximately 32 days.
Damage is caused primarily by feeding of the adults and nymphs on the sap of the sugarcane plant, which causes yellowing, reddening, and desiccation of the leaves. Additional damage results from decreased photosynthesis caused by growth of a sooty mold fungus on honeydew excreted by the insects. Finally, laceration of leaf tissue by the insect's saber-like ovipositor can cause reddening and desiccation of the leaves. There are several natural enemies of sugarcane delphacids, including predators, parasitoids, and pathogens.
Sugarcane Lacebug
The sugarcane lacebug, Leptodictya tabida, was first discovered in the United States on sugarcane in Palm Beach County, Florida, in 1990. The adult lacebug is about 3 mm long, flat and light brown or straw-colored (Figure 14). The forewings are semi-transparent and finely laced or netlike. Five long, erect spines are present on the head. Nymphs are flat and whitish in color with many long, branched, erect spines. Eggs are laid singly into leaf tissue, usually on the underside of the leaves. The tip of the egg is left outside the leaf tissue but is covered with a protective cap secreted by the adult female. A generation, from egg to adult, may take 20 to 30 days, with five nymphal molts.
Lacebug feeding results in light-green or yellow speckles on leaves. Many cultivars develop a red discoloration (russetting) on the leaves that expands beyond the area of feeding. Another characteristic diagnostic character of sugarcane lacebugs are the small, black, oily fecal deposits (i.e., frass spots) laid on the leaves (Figure 14).

Credit: Gregg Nuessly, UF/IFAS
Egg parasitoids of the sugarcane lacebug have been found in Costa Rica and Venezuela, but natural parasitoid populations have not been observed in Florida. Predators of these plant-sucking insects include earwigs and spiders.

Credit: Gregg Nuessly, UF/IFAS
Egg parasitoids of the sugarcane lacebug have been found in Costa Rica and Venezuela, but natural parasite populations have not been observed in Florida. Predators of these plant-sucking insects include earwigs and spiders.
Sugarcane Rust Mite
The sugarcane rust mite, Abacarus sacchari, was discovered feeding on sugarcane at Canal Point, Florida, in September 2007. This was the first time this mite had been officially observed feeding on sugarcane in the United States. However, further examination of historical records and samples by acarologist Cal Welbourn of the Florida Department of Agricultural and Consumer Services revealed that this mite had been first observed on sugarcane in Belle Glade, Florida, in 1983, but incorrectly identified as Abacarus officinari. Rust mites are very difficult to identify to species, but modern techniques available today (e.g., low temperature scanning electron microscopy) made it possible to correctly identify this mite in Florida. Described from Asian specimens, the sugarcane rust mite has now been recorded in Africa, Australia, India, Costa Rica, Venezuela, Brazil, and Florida. In 2014, sugarcane rust mites infested sugarcane throughout the Florida commercial sugarcane industry.
All stages of the sugarcane rust mite are oval-elongate, tapering toward the end of the abdomen, and have two pairs of legs. Waxy growths from the body vary by environment and plant host (Figure 16). These mites develop through egg, larval, and nymphal stages before reaching adulthood. Development is completed in less than 7 days. The mites can be found throughout the year but reach elevated population levels only during the mid to late summer months when temperature and humidity are relatively high.

Credit: Gary Bauchan, USDA-ARS, Beltsville, Maryland
Sugarcane rust mite feeding symptoms are cultivar-specific and range from an orange to rusty reddish-brown to purple discoloration on the underside of leaves (Figure 17). Symptoms may be masked by the disease orange rust caused by Puccinia kuehnii. However, mite feeding symptoms are more uniformly distributed over the leaves and do not result in the raised pustules characteristic of rust infestation. Furthermore, mite feeding may be restricted to the midvein or leaf blade, or it may be spread across the leaf surface. Late in the summer, heavily infested plants also show symptoms on the upper side of leaves, including reddish asymmetric blotches on the normally white center of the leaves above the mid rib. Leaves injured by mite feeding display reduced photosynthesis associated with reduced chlorophyll content, transpiration, and stomatal conductance, but elevated intercellular CO2 concentrations.

Credit: Gregg Nuessly, UF/IFAS
Recent studies show that sugarcane rust mites can negatively affect yield components including Brix, pol, and commercial recoverable sucrose in the plant cane crop, but not in subsequent sugarcane crops.
Elemental sulfur is registered for use on sugarcane and may control sugarcane rust mites. Conventional miticides including abamectin, tolfenpyrad, and spiromesifen decrease sugarcane rust mite injury. However, these conventional miticides are not registered for use on sugarcane.
Sugarcane Thrips
The sugarcane thrips, Fulmekiola serrata, was first observed in the United States infesting sugarcane in Florida in 2017. The insect primarily feeds on sugarcane. All life stages, from egg to adult, are found on sugarcane plants, generally between unrolled whorl leaves or in rolled tips and margins of older leaves. Adults (Figure 18) are approximately 1 mm long and appear dark brown to black, whereas nymphs and pupae are light-colored, whitish to yellowish.

Credit: Felipe Soto-Adames, FDACS-DPI, Gainesville, Florida
Sugarcane thrips nymphs and adults use their rasping-sucking mouthparts to feed on leaf surfaces. Feeding causes dried and curled leaf tips and margins (Figure 19) that can cause injured whorl leaves to remain tied together with a whip-like appearance. Feeding also causes yellowish streaks and dried necrotic patches on injured leaves. Sugarcane leaf death can occur under severe thrips infestations.

Credit: Julien Beuzelin, UF/IFAS
Yield losses associated with sugarcane thrips in Florida sugarcane have not been determined; however, studies in South Africa showed that the insect could cause >20% yield loss when infesting susceptible cultivars. Young sugarcane is more susceptible to thrips infestations, and dry weather conditions exacerbate thrips severity.
West Indian Canefly
The West Indian canefly, Saccharosydne saccharivora, was first recorded in Florida in 1909. This insect is not a true fly, despite its common name, and is also called West Indian sugarcane fulgorid. The adult is light to medium green with wings that are nearly transparent. The head is narrower than the body and narrows to a point in front of the golden-yellow to orange eyes. Eggs are inserted into the leaf tissue, frequently along the underside of leaves into or along the midrib of sugarcane plants. A white, waxy plug produced by the females covers egg masses (Figure 20). The nymphs are yellow to light green in color and produce copious amounts of wax, which collects at the end of their abdomens and resembles a cotton string that can exceed twice their body length. The nymphs develop through five instars in 6.5 to 7 weeks at 80°F.

Credit: Gregg Nuessly, UF/IFAS
West Indian caneflies are commonly found on several additional cultivated and weedy grass species in Florida. Numbers can occasionally grow to damaging levels resulting in yield losses, as was observed in Louisiana in 2012. Sugarcane can be affected through direct loss of sap and through growth of sooty mold fungus on honeydew produced by the nymphs and adults. Accumulation of this fungus blocks photosynthesis as with the sugarcane aphid.
Parasitoids of West Indian canefly nymphs were observed in Florida at least as early as 1937. The black-colored larvae of the parasitic wasps feed externally on the nymphs. Research conducted in Louisiana showed that insecticides, including lambda-cyhalothrin, can control the insect during outbreak years.
West Indian Cane Weevil
The West Indian cane weevil, Metamasius hemipterus, was first found infesting sugarcane in Florida in 1994. This insect is sometimes referred to as the rotten cane stalk weevil or silky cane weevil. Adult weevils (Figure 21) are usually attracted to sugarcane stalks that are already injured (e.g., damage by other insects or rats). However, the weevils may be attracted to some cultivars such as CP85-1382 that have growth cracks, but no other obvious abnormalities.

Eggs of the West Indian cane weevil are laid into sugarcane stalks through cracks or into plant tissues damaged by rats, insects, wind, or mechanical implements. The white, legless larvae from these eggs feed within the stalk and grow to more than an inch in length. A pupal case made of shredded inner stalk fibers is usually constructed within the infested plant, although pupal cases may sometimes be found on the ground around infested stalks. The entire life cycle from egg to adult may be completed in 8 to 10 weeks, depending on temperature. The tunnels and injury by the weevil can be so extensive that stalks may break. General observations indicate the weevil can cause large yield reductions in susceptible cultivars like CP85-1382. Damaged stalks may be unfit for use as seed.
A trapping method exists for the adult weevils that may be useful for identifying when infestations of the pest begin to develop. With respect to biological control, no parasitoids, predators, or pathogens have yet to be observed in Florida. Growing sugarcane varieties resistant to the weevil and limiting rat and insect injury are the key management tactics. Population levels of adult weevils in an area may be reduced using traps during the summer and fall, which might help limit infestations in sugarcane.
Selected References
Asbani, N. 2018. The evaluation of sugarcane rust mite Abacarus sacchari Channabasavana as a potential pest of sugarcane. PhD Dissertation, University of Florida, Gainesville, FL.
Beuzelin, J. M., R. H. Cherry, G. S. Nuessly, and H. S. Sandhu. 2019. Insect Management in Sugarcane. ENY-406. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/ig065
Beuzelin, J. M., M.T. VanWeelden, F.N. Soto-Adames, H.S. Sandhu, R.W. Davidson, L. Baucum, and S. Swanson. 2019. "Effect of sugarcane cultivar and foliar insecticide treatment on infestations of the invasive sugarcane thrips, Fulmekiola serrata (Thysanoptera: Thripidae)", in Florida. J. Econ. Entomol. 112: 2703–2712 https://doi.org/10.1093/jee/toz188
Hall, D. G. 1987. "The sugarcane aphid, Melanaphis sacchari, in Florida sugarcane." J. Am. Soc. Sugar Cane Technol. 7: 26–29.
Hall, D. G. 1988. "Insects and mites associated with sugarcane in Florida." Fla. Entomol. 71: 138–150.
Hall, D. G., and O. Sosa, Jr. 1994. "Population levels of Leptodictya tabida (Hemiptera: Tingidae) in Florida sugarcane." Fla. Entomol. 77: 91–99.
Hall, D. G., A. S. Konstantinov, G. S. Hodges, O. Sosa, C. Welbourn, and R. L. Westcott. 2005. "Insects and mites new to Florida sugarcane." J. Am. Soc. Sugar Cane Technol. 25: 143–156.
Odero, D., R. Cherry and D. Hall. 2013. "Weedy host plants of the sugarcane root weevil (Coleoptera: Curculionidae) in Florida sugarcane." J. Entomol. Sci. 48: 81–89.
Odero, D. C., R. H. Cherry, A. L. Wright, H. S. Sandhu, and Y. Luo. 2015. "Woody host plants of the sugarcane root weevil (Coleoptera: Curculionidae)." Florida sugarcane. J. Entomol. Sci. 50: 3–13.
Rott, P., D. C. Odero, J. M. Beuzelin, R. N. Raid, M. VanWeelden, S. Swanson, and M. Mossler. 2018. Florida Crop/Pest Profile: Sugarcane. PI-171. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/pi207
Sosa, O., Jr. 1985. The sugarcane delphacid, Perkinsiella saccharicida (Homoptera: Delphacidae), a sugarcane pest new to North America detected in Florida. Fla. Entomol. 68: 357–360.



