Family: Primulaceae (formerly in Theophrastaceae)
Common Names: Joewood; barbasco; cudjo wood; ironwood; joebush; washwood
Native Distribution: South Florida; Bahamas; Cayman Islands; Cuba; Haiti; Jamaica
USDA Zone: 10A-12B (27°F minimum)
Plant Type: Shrub or small tree
Growth Rate: Slow
Typical Dimensions: 8 ft tall x 12 ft wide
Leaf Persistence: Evergreen
Leaf Type: Simple, entire
Flowering Months: Sporadic but heaviest between June and November
Flower Color: White; ivory
Fruit Color: Ivory; orange-red; yellow
Light Requirements: Full sun to light shade
Soil Tolerances: Clay; sand; loam; acidic; alkaline; brackish; well-drained; rarely inundated
Drought Tolerance: High
Salt Tolerance: High
Wind Tolerance: High
Nutritional Requirements: Low
Propagation: Seeds; cuttings
IFAS Assessment: Not evaluated (native)
Human Hazards: Potentially poisonous but toxic compound undetermined
Major Insect and Disease Problems: None
Uses: Specimen; small flowering tree; coastal locations; container; streetscapes; wildlife host
Florida Distribution
Joewood (Jacquinia keyensis) is most common in the Florida Keys (Monroe County), where it occurs in coastal scrubs, on shell mounds, and is associated with rockland hammocks and dry sites. It prefers transition areas between coastal thickets and maritime hammocks. Joewood is also found on the northern shoreline of Florida Bay in Monroe County, in the Long Pine Key region of Everglades National Park, in Miami-Dade County, and on a few barrier islands in Lee and Charlotte Counties. It is listed as a threatened species by the state of Florida due to habitat loss and the rapid decline in the number of plants within the state. Population losses have not sufficiently decreased the number of plants to cause them to be endangered or listed as threatened. Joewood is best protected and preserved on naturally occurring sites.
In 1989, the city of Sanibel in Lee County, at the recommendation of its vegetation committee, declared the joewood to be its "city plant" in hopes that the declaration would increase respect for this native species and enhance its protection and distribution on the island.
Growth Habit
Joewood is a very slow growing, long-lived shrub or small tree. Seedlings under nursery conditions initially grow about six inches a year and are about three feet when six or seven years old. Plants found in the wild are seldom above 12 feet tall.
Joewood may be single- or multi-trunked with as many as seven smaller trunks sometimes emerging from a single main trunk close to the ground. Thus, large, multi-trunked specimens may be mistaken as a clump of tightly grown shrubs or trees. Even old joewood trees remain relatively small. One of the larger specimens identified by the primary author is an 18-foot-tall specimen on Sanibel Island. The tree has five trunks, the largest of which is 11 inches in diameter. It has been designated by the Florida Forest Service as the state's champion Joewood tree.
The trunks of joewood are smooth and gray at first, later becoming mottled by lichens. The interior wood lacks distinct growth rings. The twigs are gray when young. The plant is evergreen, and growth can occur at any time of the year. Much of the attraction of the plant is in its shape. No central leader develops. Instead, joewood produces numerous short, rigid branches, giving the plant a compact rounded crown and a distinctive appearance. If grown in full sun with plenty of room to spread, the canopy of joewood will be wider than tall. Multi-trunked plants are significantly wider than single-trunked specimens of similar heights.

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
Leaves
Leaves are simple, alternately, and spirally arranged, sometimes with very short internodes that may give a false appearance of being whorled. The false whorled characteristic is more common among other Jacquinia species and is referred to as pseudoverticillate. Leaves are yellow-green in color shortly after emerging and remain mostly upright. The leaf blades are stiff, shiny, leathery, and mostly recurved slightly to strongly curved downward. They have inconspicuous venation and margins that are entire and revolute (rolled under). The shape of the blades varies from broadly to narrowly obovate to broadly elliptic. Leaf bases are attenuate (tapering towards a very short petiole). The apices are rounded to retuse (shallowly notched), often with a mucro (short sharp abrupt point formed by extension of the midrib). The blades are approximately 1.5 to 2 inches long and 0.5 inch wide, and the petioles mostly 0.2 inches in length.

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
Flowers, Pollination, and Flowering
Flowers appear in conspicuous terminal racemes and are especially fragrant. The aroma is reminiscent of jasmine and is said to be unsurpassed by any other native in Florida's natural plant communities.
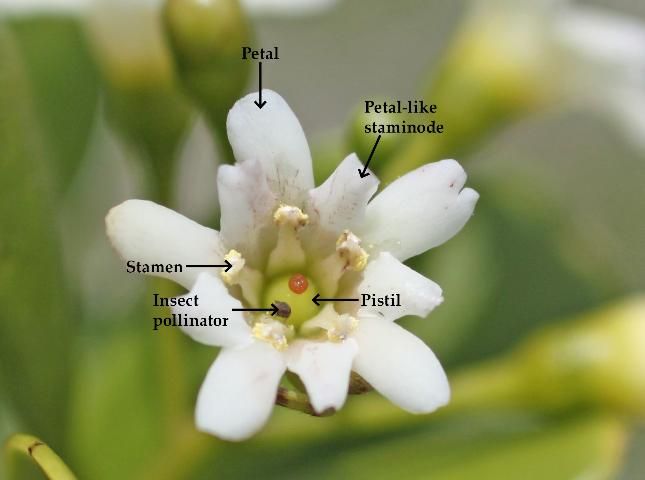
Racemes are up to three inches long. At the base of each flower is a short, green calyx, with five, indistinct, rounded lobes. There are five white or ivory colored petals which spread widely when the flowers are fully mature. The petals alternate with five shorter, petal-like staminodes (sterile stamens) of the same color, making it appear that each flower has ten petals. Five functional stamens lie opposite the five petals.
The flowers have both male and female reproductive parts (stamens and pistils) present on the same flower. However, dichogamy is pronounced, with the stamens and pistils maturing at different times to promote outcrossing. Joewood is protogynous; hence, female parts of flowers develop first and the male parts produce pollen after the stigmas of the same flower are receptive. Insect pollinators (thought to be bees and wasps seeking nectar) are attracted to the sweet-scented flowers.
Plants may flower at any time of year, but most commonly do so from mid-spring to early fall. Some individual plants or groups of plants may flower more abundantly than others. It is not uncommon for flowers and fruits to be present on a plant simultaneously.
Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
Fruits and Fruiting
With the female reproductive organs of joewood maturing before the male reproductive organs, a small green berry is often seen developing in the center of the flower while petals and functional stamens are still present. After pollination, the petals of the flowers fall, and the numerous green pistils (immature berries) remain on the terminal racemes. Fruit clusters are generally pendent, though they can be upright when supported by lower leaves. The fruits are round, smooth, hard-coated berries with the style persistent at the tip. They are about 0.35 inches in diameter and are connected to a pedicel of the same approximate length. On Sanibel Island, mature fruits are ivory-colored, but they are typically described elsewhere as being golden-orange to orange-red in color. The interior of the fruit has a sticky, gelatinous pulp containing several small embedded seeds that are fused together when the fruits are completely dried. White-crowned pigeons and other birds feed on the fruits of joewood, and the plant provides significant cover for wildlife.

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
Landscaping
Joewood makes a good specimen plant tolerant of sandy, high-pH soils, salt air, and short periods of salt water intrusion. It adapts to partial shade, but should be planted in full sun in order to develop a fully-rounded crown. Container plants can be planted at any time of the year. After planting, water it frequently for several weeks for establishment. Once established, it can withstand heat, drought, and severe wind. It rarely, if ever, needs pruning. Plant development is slow but adequate fertilizer applications and routine irrigation in the dry season should accelerate growth.
Best Ways to Identify Joewood

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
Comparison of Jacquinia keyensis Mez and Jacquinia arborea Vahl
Jacquinia arborea (previously known as Jacquinia armillaris var. arborea) braceletwood is a non-native relative of joewood. Identification of these species is important, as braceletwood has naturalized in Southern Florida and has been identified as an invasive plant species by the Florida Keys Invasive Species Task Force.
When compared side by side, these two species are readily distinguished. The leaf blades of the braceletwood are larger and not as leathery in appearance. They are commonly 3.0–5.0 inches long and about 1.5 inches wide. The petioles are approximately 0.3 inches long. In braceletwood, the internodes are extremely short, resulting in a strongly pseudoverticillate leaf arrangement; in joewood, the internodes are slightly longer, so the leaves are only weakly pseudoverticillate. The leaf margins of braceletwood may be flat or slightly revolute (rolled under), but are never strongly revolute, as often seen in joewood. Both species have highly fragrant flowers. Joewoods are found to have mostly ivory-colored berries in Lee County, with reports of otherwise golden-orange, orange-red, and yellow fruits. The berries of braceletwood are approximately the same size as joewood, but fruits are consistently orange-red or red.

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS

Credit: Stephen H. Brown, UF/IFAS
List of Botanical Terms
Apex (pl. apices): The point of a leaf furthest from the petiole.
Attenuate: Gradually tapering at the base or at the tip.
Berry: A fleshy fruit with many seeds developing from a single pistil.
Calyx: (pl. calyces, calyxes): Group or cluster of modified leaves enclosing a flower bud, each segment being a sepal.
Dichogamous: With the pistils and stamens maturing at different times to prevent self-fertilization.
Elliptic: A leaf blade broadest at the middle and narrower at two equal ends.
Entire: Complete, not toothed, notched, or divided, with smooth margins.
Internode: The portion of a stem between two nodes.
Margin: The edge, as in the edge of a leaf blade.
Mucro: A short, sharp, abrupt terminal point.
Obovate: Widest above the center. The opposite of ovate.
Pedicel: The stalk of a single flower.
Petal: An individual segment of the corolla, variously colored, seldom green.
Pistil: The female organ of a flower consisting of stigma, style and ovary.
Pseudoverticillate: Having very close internodes, thus giving the appearance of whorled leaves.
Petiole: The stalk of a leaf.
Protogyny: The female parts of a flower developing before the male parts.
Raceme: An unbranched inflorescence with stalked flowers on an elongated axis with flowers opening in succession towards the apex.
Recurved: Curved backward.
Retuse: Shallowly notched at the apex.
Revolute: With leaf margins rolled downward toward the underside.
Stamen: The male organ of a flower consisting of an anther and a filament.
Staminodes: Sterile stamens.
Stigma: The apex of a pistil (female part) that is receptive to pollen grains.
Style: The usually narrow portion of the pistil between the stigma and the ovary.
References
Broschat, T.K. and A.W. Meerow, 2001. Reference Guide to Florida Landscape Plants. Hollywood, Florida: Betrock Information System, Inc.
Farnsworth, Steve. "Cold Hardiness Report on Tropical Native Plants," The Palmetto, 1985. Orlando, Florida: Florida Native Plant Society, 5(3):12.
Gann, G.D., M.E. Adbo, J.W. Gann, G.D. Gann, Sr., S.W. Woodmansee, K.A. Bradley, E. Grahl and K.N. Hines. 2016. "Joewood: Jacquinia keyensis." Natives For Your Neighborhood: A program of the Institute for Regional Conservation, Delray Beach, Florida. http://www.regionalconservation.org/beta/nfyn/plantdetail.asp?tx=Jacqkeye
Flora of North America Editorial Committee, eds. 1993+. Flora of North America North of Mexico. 20+ vols. Jacquinia keyensis. New York and Oxford. Vol. 8, 2009. pp. 253–254. http://www.efloras.org/florataxon.aspx?flora_id=1&taxon_id=250092184
Mullins, Stephen. "Moving joewood trees," The Palmetto, 1993. Orlando, Florida. Florida Native Plant Society.13(2): 8–9. http://fnps.org/assets/pdf/palmetto/mullins_stephen_moving_joewood_trees_vol_13_no_2_summer_1993.pdf
Perkins, K.D., and W.W. Payne. 1980. Guide to the Poisonous and Irritant Plants of Florida. Circular 441. Gainesville: University of Florida Institute of Food and Agricultural Sciences.
The Angiosperm Phylogeny Group. 2003. "An update of the Angiosperm phylogeny group classification for the orders and families of flowering plants. APG II." Botanical Journal of the Linnean Society. 141: 399–436. https://onlinelibrary.wiley.com/doi/pdf/10.1046/j.1095-8339.2003.t01-1-00158.x
Tomlinson, P.B. 1986. The Biology of Trees Native to Tropical Florida. Massachusetts: Harvard University Printing Office.
Workman, R.W. 1980. Growing Native: Native plants for landscape use in Coastal South Florida. Sanibel, Florida: The Sanibel-Captiva Conservation Foundation, Inc.
Wunderlin, R.P., B.F. Hansen, A.R. Franck, and F.B. Essig. 2017. "Atlas of Florida Plants." Institute for Systematic Botany, University of South Florida, Tampa, Florida. http://florida.plantatlas.usf.edu/browse/genus