An Informational Coral Guide
Florida's shallow-water coral reefs have seen large declines in coral cover over the last 40 years. The following information is meant to be a guide to common Scleractinian (stony) corals of Florida. Although several of the corals presented here are no longer common on the reefs today, they are still considered common due to their historical prevalence on Florida's reefs. Florida's Coral Reef Protection Act prohibits damaging coral reef habitats in any way, and several of these species are federally protected under the Endangered Species Act. In their natural environment, these corals should never be handled or touched in any way. It is important to note that although several of the species discussed here are growing more numerous due to their wide tolerance, the overall condition of coral reefs along Florida's coastline is declining, and the reefs are highly threatened because the most important reef-building corals are in a state of decline.
Scleractinian Coral
The order Scleractinia falls under the subclass Hexacorallia. Hexacorallia is known for its polyps that contain tubular tentacles in multiples of six (Humann and DeLoach 2013). This paper focuses on Scleractinian corals, also known as "stony" or "hard" corals, which are native to Florida. These corals form dense networks of colonial polyps built around a calcium carbonate skeleton. Figure 1 portrays the complex structure of the coral polyp and labels important terminology. A glossary, located at the end of this document, describes these terms in greater detail along with other terminology used throughout the guide. The rigid and complex habitat formed by scleractinian corals makes them a critical element to the formation of south Florida's primary reef structure (Figure 2). Scleractinia can be subdivided into six colony types as described in Humann and DeLoach (2013). Five colony types from the order Scleractinia covered in this document are:
- Branching and pillar corals
- Brain corals
- Encrusting, mound, and boulder corals
- Leafy, plate, and sheet corals
- Flowering and cup corals
In this guide, each profile contains information on physical characteristics and habitat preferences, as well as current distribution and population status for each species. Two images accompanying each species profile provide a full-colony perspective and close-up of polyp and corallite structure. If present, the text "Mov." next to the species name will link to a video of each species in the wild.
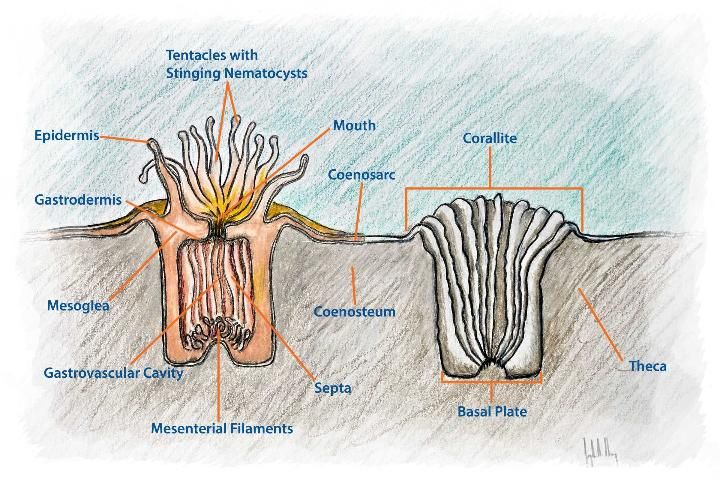
Credit: Joseph Henry, UF/IFAS SFRC. Adapted From: NOAA, Anatomy of a Coral Polyp
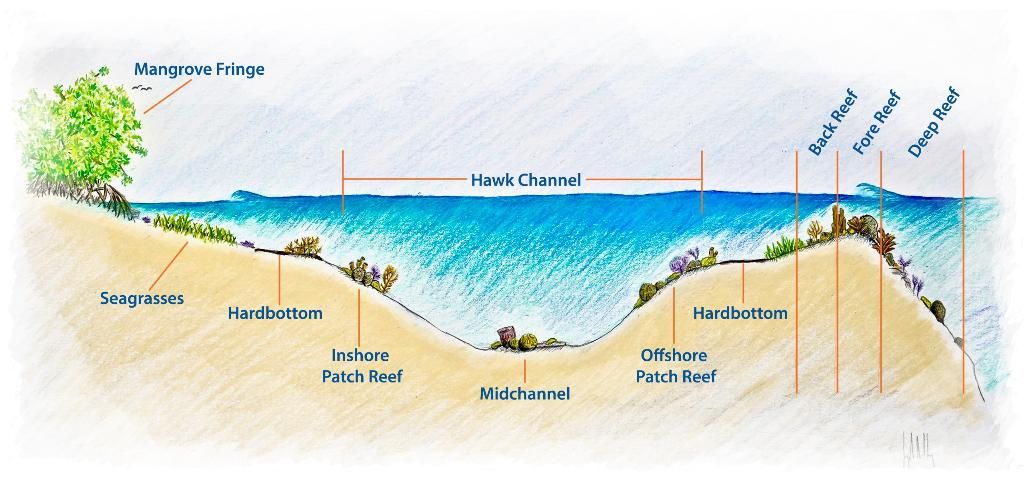
Credit: Joseph Henry, UF/IFAS SFRC. Adapted from: Florida's Coral Reef Ecosystem Poster: NOAA, Florida DEP, FWC. Florida Keys NMS
Branching and Pillar Corals
Staghorn Coral (Acropora cervicornis)
Characteristics
Staghorn coral (A. cervicornis) has been one of the most important reef-building species in southern Florida. This species has narrow, cylindrical branches that historically formed large thickets for other species of invertebrates and fish to occupy (Gilbert 1986). These branches are commonly yellow, brown, or golden brown and contain angled corallites that extend towards the tips of the branches. This species is considered one of the fastest growing corals in regard to linear extension, with single branches extending as much as 10 inches (26 cm) per year in ideal conditions (Lewis, Axelson, Goodbody, Page, and Chislett 1968).
Habitat
Found from 3 to 82 feet (1 to 25 m) underwater, but more commonly seen in Florida at depths less than 30 feet (9 m). Staghorn coral favors areas of low wave exposure and greater light availability (Aronson and Precht 2001). Staghorn coral is generally found in areas with water temperatures varying from 66°F to 86°F (19°C to 30°C) and salinities of 33 to 37 parts per thousand (ppt) (NOAA Fisheries 2002). In southeast Florida, staghorn coral historically dominated the outer reef platforms, but climate change, anthropogenic stressors and disease have greatly reduced the species presence in these areas. Currently, in Florida, small populations of staghorn coral can be found on near shore patch reefs and hard bottom habitat (Acropora Biological Review Team 2005).
Distribution
Staghorn coral is one of only two Acropora species found in southern Florida and the Caribbean. These species form a natural hybrid known as Acropora prolifera. Staghorn coral can also be found throughout the Western Central Atlantic, the Bahamas, and the Gulf of Mexico (Goldberg 1973).
Population Status
Staghorn coral is currently listed as threatened under the Endangered Species Act. Heavily impacted by climate change, anthropogenic stressors and disease, populations have declined by more than 80%–98% throughout their historical range (Aronson et al. 2008b).

Credit: Joseph Henry, UF/IFAS

Credit: Josh Patterson, UF/IFAS
Elkhorn Coral (Acropora palmata) Mov.
Characteristics
Elkhorn coral (A. palmata) is one of the more important reef-building corals for southern Florida. Its large, flattened branches create ideal habitat for reef fish and reduce wave energy (Randall and Szmant 2009). As seen below, this species' branches grow at acute angles from the parent blade, which become more rounded when found in turbulent waters. At maturity, the blades of elkhorn coral are typically greater than 1.5 feet (0.5 m) in length (Porter 1987).
Habitat
Elkhorn coral is commonly found attached to the rocky substrate of shallow fore-reefs and outer reef slopes with exposure to higher current and wave action (Williams, Miller, and Kramer 2008). The common depth range for this species is very shallow, between 1 and 16 feet (0.5 to 5 m), but more commonly seen in Florida between 3 and 30 feet (1 to 9 m) (Goreau and Wells 1967).
Distribution
Elkhorn coral can be found distributed throughout the shallow waters of the Western Atlantic. In the Florida Keys, this species can be found as far north as Fowey Rocks and Biscayne National Park near Miami. Other known areas include the Gulf of Mexico, the Bahamas, and the Caribbean (Porter 1987).
Population Status
Similar to staghorn coral, elkhorn coral populations have undergone large-scale declines throughout their historical range. Once common on the shallow reefs of south Florida, elkhorn coral populations have declined by roughly 95% (Williams, Miller, and Kramer 2008). The impacts of climate change, anthropogenic stressors, and disease have all heavily impacted this species and resulted in elkhorn coral being classified as threatened under the Endangered Species Act in 2006 (NMFS 2006).

Credit: Joseph Henry, UF/IFAS

Credit: DivePix, divepix.webdhd.com. Used with permission.
Clubbed Finger Coral (Porites porites) Mov.
Characteristics
Clubbed finger coral (P. porites) is a branching coral with a dense network of corallites and polyps that gives the coral a fuzzy appearance when the polyps are extended. Colonies grow to 1 to 4 feet (0.3 to 1.2 m) and the branches range in diameter between 0.5 and 1 inch (1.3 to 2.6 cm). This coral presents in three forms and a variety of colors, ranging from yellowish-brown, brown, tan, gray, and on rare occasions lavender to purple (Humann and DeLoach 2013). The three forms are P. porites (divaricata), which has thin branches and is generally found in shallow water with higher sedimentation, P. porites (furcata) whose branches divide and leave spaces between adjacent branches, and P. porites (porites), which presents with dense, thick branches (Warner 2012). Some scientists consider these three Porites forms as different species.
Habitat
Although this species can be found in most reef areas, it is most commonly found in shallow water less than 3 feet (1 m) deep or at depths greater than 66 feet (20 m). A high tolerance to sedimentation has allowed clubbed finger coral to extend its habitat into seagrass beds and attach to the roots of mangroves (Macintyre et al. 2000).
Distribution
Clubbed finger coral is found in south Florida, the Bahamas, Bermuda, the Caribbean and the west coast of Africa (Warner 2012).
Population Status
Clubbed finger coral populations are currently considered stable and are not at great risk for extinction. Although considered a low-risk species, studies have shown the species is vulnerable to bleaching and disease. For example, in 2005, populations in the US Virgin Islands experienced an extremely high level of loss due to bleaching and disease (Whelan et al. 2007).

Credit: Joseph Henry, UF/IFAS

Credit: Charlie Veron, Australian Institute of Marine Sciences. Used with permission
Pillar Coral (Dendrogyra cylindrus) Mov.
Characteristics
As the name suggests, pillar coral (D. cylindrus) grows in multiple cylindrical pillars that extend vertically from the base towards the water's surface. Mature colonies range in size from 4 to 10 feet (2 to 3 m) high with individual pillars ranging from 3 to 5 inches (8 to 13 cm) in diameter. Multiple colonies of pillar coral have been known to fuse, creating larger-than-average formations of this species. This species can appear brown, light tan, or golden-brown (Humann and DeLoach 2013). Unlike other stony corals, the polyps of pillar coral have long tentacles that are extended during the day.
Habitat
Colonies of pillar coral have an encrusting base that is most commonly attached to rocky, flat substrate or slightly sloped reefs. In Florida, the species is most commonly seen between 4 and 65 feet (1 to 20 m) under the surface. In the Florida Keys, pillar coral populations are seldom found near shore or on the back-reef. They are more common on the backside of the fore-reef (Brainard et al. 2011).
Distribution
Found only in the Western Atlantic, pillar coral populations are limited in southern Florida. The bulk of the species numbers are in the Bahamas and the Caribbean. This species is restricted to the southwestern part of the Gulf of Mexico (Tunnell 1988).
Population Status
Pillar coral was listed on the Endangered Species Act in October 2015. Several life history characteristics have made this species increasingly vulnerable to extinction. These characteristics include small population size, limited geographical range, slow growth and decreased reproductive success (Marhaver, Vermeij, and Medina 2015). As of early 2018, an ongoing disease outbreak of white plague has greatly reduced pillar coral populations in Florida (Precht 2018).

Credit: Wikimedia Commons

Credit: Joseph Henry, UF/IFAS
Brain Corals
Symmetrical Brain Coral (Pseudodiploria strigosa)
Characteristics
Also known as smooth brain coral or common brain coral, symmetrical brain coral (P. strigosa, formerly Diploria strigosa) adopts a hemispherical shape in shallow water or a flat encrusting form at greater depths. Colonies range in diameter from a few inches to 6 feet (2 m) and are commonly yellowish-brown, grey or green in color (Humann and DeLoach 2013). A common way to differentiate this species from grooved brain coral is by looking closely at the ridges. Symmetrical brain coral ridges generally lack a groove running along the crest. However, some individuals may present with a fine line running along the ridges as seen below (Warner 2012).
Habitat
P. strigosa occupies a wide variety of marine habitats. This species can be found growing on the rocky substrate of protected back-reefs or growing on the sides of sea walls and ledges exposed to higher wave activity. The depth of this species ranges from 3 to 130 feet (1 to 40 m), but it is most commonly found in Florida at depths less than 33 feet (10 m) (Warner 2012).
Distribution
Isolated to the Western Atlantic, P. strigosa is the most common brain coral found in the Caribbean. It is also found in southern Florida, Bermuda, the Bahamas, and the Gulf of Mexico.
Population Status
Currently, the species is not under immediate threat, and there is no known trend of widespread population decline. Studies have found localized losses due to disease, but this species has not been as heavily impacted as other corals (Bruckner and Bruckner 1997).

Credit: Joseph Henry, UF/IFAS

Credit: Wikimedia Commons
Grooved Brain Coral (Diploria labyrinthiformis) Mov.
Characteristics
Grooved brain coral (D. labyrinthiformis) is recognized for its color, hemispherical shape, and weaving crevices surrounded by grooved ridges (Humann 2013). Variation in growth patterns can occur, and some corals may have wider grooves than those depicted below. Colony diameter typically ranges from 1 to 4 feet (0.3 to 1.2 m). Color varies from yellow-brown to brownish-grey (Humann and DeLoach 2013).
Habitat
Grooved brain coral is commonly found in Florida on inward slopes of offshore reefs at depths of 15 to 50 feet (4.5 to 15 m), although it can be found in other regions at depths as great as 135 feet (42 m) (Humann and DeLoach 2013). In the Florida Keys, this species is commonly found in mid-channel patch reefs. (Wagner, Kramer, and Van Woesik 2010).
Distribution
Grooved brain coral can be found in southern Florida, Bermuda, the Bahamas, and throughout the Caribbean (Humann and DeLoach 2013). In the Florida Keys, this species is present in Biscayne National Park extending to the Lower Keys, with a majority of their numbers being found in the northern portion of that range (Wagner, Kramer, and Van Woesik 2010).
Population Status
Grooved brain coral populations are currently considered stable. This species is still under considerable pressure from diseases, along with localized areas of bleaching (Aronson et al. 2008c). During a large coral bleaching event in 2005, grooved brain coral populations experienced population declines throughout the Florida Keys (Wagner, Kramer, and Van Woesik 2010).

Credit: Jan Derk, Wikimedia Commons

Credit: Ryan McMinds, Wikimedia Commons
Rose Coral (Manicina areolata)
Characteristics
Rose coral (M. areolata) is known for growing in two different formations; elliptical and hemispherical. Elliptical colonies grow to an average of 2–6 inches (5–15 cm), while hemispherical colonies can grow to 4–8 inches (10–20 cm)(Humann and DeLoach 2013). This small colony size is in contrast to the other brain corals, which are normally larger. Colonies commonly present in the colors of grey, green, brown, and pale orange.
Habitat
Location is another means of differentiating this species from the other brain corals. Rose coral is usually found growing in seagrass beds or open sandy substrate (Aronson et al. 2008). Elliptical colonies tend to occur on turtle grass beds and sandy substrate, where hemispherical colonies can be found on hard substrates. The depth range of this species in Florida is 2 to 200 feet (1 to 61 meters) (Humann and DeLoach 2013).
Distribution
Rose coral is commonly found in the Caribbean, the Bahamas, the Gulf of Mexico, and the seagrass beds of the Florida Keys (Humann and DeLoach 2013).
Population Status
Current populations of rose coral are stable and not under immediate threat.

Credit: Wikimedia Commons

Credit: Charles Sheppard, University of Warwick. Used with permission
Encrusting, Mound, and Boulder Corals
Mustard Hill Coral (Porites astreoides)
Characteristics
Mustard hill coral (P. astreoides) has a flat encrusting structure in areas of higher wave activity. In deeper water, however, this species takes on a more rounded structure with a lumpy surface covered with closely associated corallites as seen below. This dense network of corallites gives the coral a fuzzy appearance when the polyps are extended. Color variations of this species include yellow to yellowish-brown, beige, or gray. Size generally varies from 6 inches to 2 feet (15 to 61 cm) (Humann and DeLoach 2013).
Habitat
Mustard hill coral can be found throughout the reef environment, from the shallow fore-reef to the deep regions of the back-reef. This species has a depth range of 3 to 150 feet (1 to 46 m) but is commonly found in Florida at depths of 15 to 80 feet (5 to 24 m) (Humann and DeLoach 2013).
Distribution
This species has a very wide distribution in both the western and eastern Atlantic. In the western Atlantic, this species can be found in southern Florida, the Bahamas, Bermuda, the Caribbean, Brazil, and the southwestern regions of the Gulf of Mexico. In the eastern Atlantic, this species is found along the west coast of Africa (Warner 2012).
Population Status
Mustard hill coral populations are currently considered to be increasing given their high recruitment rates. Although more resilient to environmental change, this species is still vulnerable to disease and predation by stoplight parrotfish (Sparisoma viride) (Aronson et al. 2008e).

Credit: Wikimedia Commons

Credit: Wikimedia Commons
Massive Starlet Coral (Siderastrea siderea) Mov.
Characteristics
Massive starlet coral (S. siderea), also known as "round starlet" or "smooth starlet," is a light gray to golden brown coral covered with pitted round corallites. The characteristic depressed corallites range in size from 0.1 to 0.75 inches (3 to 20 mm) and give this coral a smooth appearance (Warner 2012). This species grows to a large boulder or dome shape. Younger colonies are smaller and encrusting. Colony diameter generally ranges from 1 to 6 feet (0.3 to 2 m) (Humann and DeLoach 2013).
Habitat
This species is common, extending from the fore-reefs to the shallow lagoons and on occasion growing out of sandy substrate (Warner 2012). Massive starlet coral prefers the clear water of protected reef areas at depths of 25 to 45 feet (8 to 14 m) in Florida but can be found as shallow as 2 feet (61 cm) and as deep as 220 ft (67 m) throughout the species range.
Distribution
This species is common to Florida, the Bahamas, Bermuda, and the southwestern regions of the Gulf of Mexico. Massive starlet coral is found along the eastern coast of Florida, and some studies have described populations as far north as North Carolina (Macintyre 2003).
Population Status
Massive starlet coral populations are currently considered stable; the species has shown a higher tolerance to changing environmental conditions. Although this species has had localized declines, no major losses have been seen across the species' historical range (Aronson et al. 2008f). As of early 2018, however, incidence of disease has increased markedly, especially in southeast Florida and the Upper Keys (CREMP 2017).

Credit: Wikimedia Commons

Credit: Charlie Veron, Australian Institute of Marine Sciences. Used with permission.
Lobed Star Coral (Orbicella annularis) Mov.
Characteristics
Lobed star coral (O. annularis) has closely associated corallites and a smooth but irregular surface, as seen below. Colonies grow to 10 feet (3 m) or more in diameter and can be yellow-brown, greenish brown, or gray. Historically, this species was known for its many growth forms, but due to the work of Weil and Knowlton in the 1990s, mountainous star coral (O. faveolata) and boulder star coral (O. franksi) were differentiated from lobed star coral based on depth and morphology (Weil and Knowlton 1994). O. faveolata presents in a singular large encrusting formation, and O. annularis presents as multiple encrusting colonies.
Habitat
This species can be found growing at depths from 1.5 to 270 feet (0.5 to 82 m) beneath the surface (Reed 1985). Despite the large depth range, it is most commonly found in semi-protected lagoons and upper reef slopes of Florida in shallow waters from 3 to 33 feet (1 to 10 m) deep.
Distribution
Lobed star coral is distributed in southern Florida and Bermuda, and throughout the Caribbean, the Bahamas, and the Gulf of Mexico. Biscayne National Park and the Florida Keys National Marine Sanctuary contain populations of lobed star coral (Brainard et al. 2011).
Population Status
This species is currently at a high risk of extinction and is listed as threatened under the Endangered Species Act. Lobed brain coral was once a dominant coral throughout its geographic range, but populations have declined extensively (Burke et al. 2004). Slow growth, low recruitment, and the preference for shallow water habitat make this species highly vulnerable to changing ocean conditions (Brainard et al. 2011).

Credit: Wikimedia Commons

Credit: Wikimedia Commons
Mountainous Star Coral (Orbicella faveolata) Mov.
Characteristics
The structure of mountainous star coral (O. faveolata) varies depending on the depth of the water where it grows. In shallow water, colonies form mounding structures with skirted edges around the periphery of the colony. At depths below 80 feet (24 m), colonies generally grow in flattened plates, which overlap each other like shingles. Individual corallites are small, approximately 0.13 inches (0.3 cm) in diameter. Color variation for the species ranges from yellow-brown to green and gray (Humann and DeLoach 2013). Mountainous star coral was identified as a distinct species in the 1990s.
Habitat
Mountainous star coral is found throughout many reef habitats, but most commonly in the fore-reef. This species can be found between 1.5 and 131 feet (0.5 to 40 m) underwater, but in Florida it is most commonly found at depths of 33 to 66 feet (10 to 20 m) (Weil and Knowlton 1994).
Distribution
Mountainous star coral has a wide range compared to other stony corals of Florida. It is commonly found in south Florida, the Bahamas, the Caribbean, and throughout the Gulf of Mexico.
Population Status
Mountainous star coral is currently listed as threatened under the Endangered Species Act, but due to the species' low growth and recruitment combined with high losses throughout its historical range, this coral is at a very high risk for extinction (Brainard et al. 2011). Studies done off the coast of Jamaica have revealed a loss of 90% of this species from 1980 to 1994 as the species struggled to compete with environmental changes and reef communities becoming dominated by macro algae (Hughes 1994.)

Credit: Wikimedia Commons

Credit: Wikimedia Commons
Great Star Coral (Montastraea cavernosa)
Characteristics
Great star coral (M. cavernosa) is known for building large colonies ranging from 2 to 8 feet (0.6 to 2.5 m) in diameter. The corallites of this species are known for being large. Each individual corallite can range from 0.25 to 0.5 inches (0.6 to 1.3 cm) in diameter. At shallow depths, this species forms large dome-like structures. In deeper water, it creates encrusting sheets (Warner 2012). On rare occasions, great star coral fluoresces red or orange, but normally the coral is yellowish brown or green (Humann and DeLoach 2013).
Habitat
This species is found throughout the coastal reef environment and is the dominant coral species at intermediate depths of 40 to 100 feet (12 to 30 m). With a depth range between 6 and 300 feet (2 to 91 m), great star coral is known for being one of the deepest-dwelling stony corals of Florida (Humann and DeLoach 2013).
Distribution
Great star coral is commonly found in Florida, the Bahamas, Bermuda, and the Caribbean. Populations have been identified along coastal Brazil and also in the Eastern Atlantic along the west coast of Africa (Goodbody‐Gringley, Woollacott, and Giribet 2012).
Population Status
Great star coral populations are currently considered stable, but localized mortality has been observed due to disease and environmental shifts. During a cold-water event that hit the Florida Keys in January of 2010, greater than 50% of great star coral colonies experienced partial or complete mortality (Colella et al. 2012).

Credit: Wikimedia Commons

Credit: Wikimedia Commons
Rough Cactus Coral (Mycetophyllia ferox)
Characteristics
Rough cactus coral (M. ferox) colonies generally grow encrusting formations or create overhangs on edged substrate. Colonies will grow to 1 to 2 feet (30–61 cm) in diameter (Humann and DeLoach 2013). Corallites generally grow in single rows, and colonies present in several colors including grey, blue, and brown (Wells 1973).
Habitat
This species is commonly found in Florida at depths of 30–70 feet (9–20 meters) in areas with very strong water flow. This species can occupy depths as shallow as 2 feet (61 cm) and as deep as 120 feet (37 m) but is more often found in shallow mid-channel patch reefs (Humann and DeLoach 2013).
Distribution
Rough cactus coral is commonly found in the Caribbean, Bahamas, and occasionally in south Florida and the Western Gulf of Mexico (Humann and DeLoach 2013).
Population Status
Rough cactus coral populations are currently considered vulnerable due to the species' high susceptibility to disease and bleaching (Aronson et al. 2008). During a white plague outbreak that swept through the northern keys in the 1970s, nearly all colonies of rough cactus corals were infected by the disease, resulting in a mortality rate of 30% (Dustan 1977). This coral is listed as threatened under the Endangered Species Act.

Credit: Charlie Veron, Australian Institute of Marine Sciences. Used with permission.

Credit: Charlie Veron, Australian Institute of Marine Sciences. Used with permission.
Leafy, Plate, and Sheet Corals
Lettuce Coral (Undaria agaricites)
Characteristics
Lettuce coral (U. agaricites, formerly Agaricia agaricites) forms encrusting or hemispherical structures with ridges running in multiple directions. Vertical extending formations have corallites on both sides, while horizontal or encrusting structures have corallites only on the top surface (Warner 2012). Colony size typically ranges from 4 inches to 3 feet (10 cm to 1 m) and colors include brown, yellow, grayish-brown, to purplish-blue (Humann and DeLoach 2013).
Habitat
This species displays a very high tolerance for habitat variability and depth, and is found in areas commonly uninhabitable for most other corals, including deep, dark crevices and shallow, turbid waters. This coral is found from the back-reef to the outer reef-wall and even into mangrove forests. The depth range for this species is 3 to 240 feet (1 to 73 m) (Humann and DeLoach 2013).
Distribution
Lettuce coral is found throughout Florida and the Bahamas. It is one of the most common coral species found in the Caribbean (Warner 2012). Unlike many species restricted to the Caribbean, this coral is also found along the eastern coast of Brazil, from Cape Sao Roque to Nova Almeida (Fernanda 2008).
Population Status
Lettuce coral populations are currently considered stable. This continues to be a dominant coral throughout its historical range.

Credit: Wikimedia Commons

Credit: Wikimedia Commons
Flowering and Cup Corals
Ivory Bush Coral (Oculina diffusa)
Characteristics
Ivory bush coral (O. diffusa) forms small colonies with short, dense branches and large, cup-like corallites. Mature colonies vary in size from 1 to 12 inches (2 to 30 cm), and branches are roughly 0.5 inch (1 cm) in diameter (Humann and DeLoach 2013). Each branch contains several widely spaced, slightly protruding corallites. This species is commonly yellowish-brown and encrusted with other organisms, but certain colonies found living in dark areas or at depth are white due to the absence of zooxanthellae (Warner 2012).
Habitat
Ivory bush coral is a very hardy species that is commonly found in reef areas characterized by sloping bottoms and shallow, high-sediment habitats. This species' depth ranges from 3 to 75 feet (1 to 23 m) (Humann and DeLoach 2013). This species' high tolerance of marginal temperatures, salinity, and turbidity has allowed it to occupy coastal inlets and lagoons.
Distribution
This species is commonly found in Florida, Bermuda, the Bahamas, and the Caribbean. Although this coral can be found in the Caribbean, distribution is sparse with limited populations in the West Indies (Aronson et al. 2008).
Population Status
Ivory bush coral populations are currently considered stable as the species is tolerant of a wide range of environmental conditions, including salinity, temperature, and sedimentation. Due to this species' ability to live in shallow, soft-bottom substrate, like seagrass beds, it is possible that physical damage from trawls, boat and anchor scars is one of the greater threats to the species (Aronson et al. 2008).

Credit: Charles Sheppard, University of Warwick. Used with permission

Credit: Charles Sheppard, University of Warwick. Used with permission.
Glossary of Terms
Coenosarc: The system of tissue tubes connecting polyps of colonial corals.
Coenosteum: The calcareous skeleton of a coral colony.
Corallite: The calcareous skeleton of a single coral polyp.
Epidermis: The outer layer of cells covering an animal.
Gastrodermis: The inner layer of cells that lines the membrane of the gastrovascular cavity.
Mesoglea: A jelly-like substance found between the internal and external cellular layers of coral polyps.
Nematocyst: A specialized cell found in cnidarians. Can be used to capture prey or in self-defense.
Order: A biological classification between the taxonomic ranks of Family and Class.
Polyp: The individual soft-bodied animal that collectively with other polyps forms a coral.
Recruitment: The process by which juvenile species complete larval stages, settle, and become part of the adult population.
Septa: Vertical plates radiating from corallite wall.
Substrate: The surface on which an organism lives or grows.
Theca: Corallite wall.
References
Aronson, R., A. Bruckner, J. Moore, B. Precht, and E. Weil. 2008. The IUCN Red List of Threatened Species 2008.
Gilbert, C. R. 1986. Species Profiles. Life Histories and Environmental Requirements of Coastal Fishes and Invertebrates (South Florida). Southern, Gulf, and Summer Flounders.
Goodbody‐Gringley, G., R. M. Woollacott, and G. Giribet. 2012. "Population structure and connectivity in the Atlantic scleractinian coral Montastraea cavernosa (Linnaeus, 1767)." Marine ecology 33(1): 32–48.
Goldberg, W. M. 1973. The ecology of the coral-octocoral community of the southeast Florida coast: geomorphology, species composition and zonation. Bulletin of Marine Science 23: 465–488.
Goreau, T. F., and J. W. Wells. 1967. The shallow-water Scleractinia of Jamaica: Revised list of species and their vertical distribution range. Bulletin of Marine Science 17: 442–453.
Hughes, T. P. 1994. Catastrophes, phase shifts, and large-scale degradation of a Caribbean coral reef. Science 265: 1547–1551
Humann, P., and N. DeLoach. 2013. Reef coral Identification: Florida, Caribbean, Bahamas. New World Publication, Inc., FL.
Lewis, J. B., F. Axelsen, I. Goodbody, C. Page, and G. Chislett. 1968. Comparative growth rates of some reef corals in the Caribbean. McGill Univ. Marine Science Manuscript. Rep. 10, p. 1–27.
Macintyre, I. G. 2003. "A classic marginal coral environment: tropical coral patches off North Carolina, USA." Coral Reefs 22: 474.
Macintyre, I. G., I. Goodbody, K. Ruetzler, D. S. Littler, and M. M. Littler. 2000. "A general biological and geological survey of the rims of ponds in the major mangrove islands of the Pelican Cays, Belize." Atoll Research Bulletin 467: 13–44.
NMFS. 2006. Endangered and threatened species: Final listing determinations for elkhorn coral and staghorn coral. Federal Register 71: 26852–26861
NOAA Fisheries. 2002. "General Fact Sheet: Atlantic Acropora Corals" (On-line pdf). NOAA Fisheries. https://www.fisheries.noaa.gov/species/acropora-pharaonis-coral
Porter, J. W. 1987. Species profiles: Life histories and environmental requirements of coastal fishes and invertebrates (south Florida) – Reef-building corals. US Fish and Wildlife Service Biological Report 82(11.73). US Army Corp of Engineers, TR EL-82-4, 23.
Precht, W. 2018. Coral disease ravages reef-building corals throughout southeast Florida. Inside Ecology Magazine. https://insideecology.com/2018/04/05/coral-disease-ravages-reef-building-corals-throughout-southeast-florida/
Randall, C. J., and A. M. Szmant. 2009. "Elevated temperature affects development, survivorship, and settlement of the elkhorn coral, Acropora palmata (Lamarck 1816)." The Biological Bulletin 217(3): 269–282.
Reed, J. K. 1985. "Deepest distribution of Atlantic hermatypic corals discovered in the Bahamas." Fifth International Coral Reef Congress 6: 249–254 Tahiti.
Tunnell, J. W. J. 1988. "Regional comparison of southwestern Gulf of Mexico to Caribbean Sea coral reefs." In Proc. 6th Int. Coral Reef Symp., Vol. 3, pp. 303–308, Townsville, Australia.
Wagner, D. E., P. Kramer, and R. Van Woesik. 2010. "Species composition, habitat, and water quality influence coral bleaching in southern Florida." Marine Ecology Progress Series 408: 65–78.
Warner, G. 2012. Corals of Florida & the Caribbean. University Press of Florida, Gainesville, FL.
Weil, E., and N. Knowton. 1994. "A multi-character analysis of the Caribbean coral Montastraea annularis (Ellis and Solander, 1786) and its two-sibling species, M. faveolata (Ellis and Solander, 1786) and M. franksi (Gregory, 1895)." Bulletin of Marine Science 55: 151–175.
Wells, J. W. 1973. "Coral reef project—papers in memory of Dr. Thomas F. Goreau. 2. New and old scleractinian corals from Jamaica." Bulletin of Marine Science 23(1): 16–58.
Whelan, K. R. T., J. Miller, O. Sanchez, and M. Patterson. 2007. "Impact of the 2005 coral bleaching event on Porites porites and Colpophyllia natans at Tektite Reef, US Virgin Islands." Coral Reefs 26: 689–693.
Williams, D. E., M. W. Miller, and K. L. Kramer. 2008. "Recruitment failure in Florida Keys Acropora palmata, a threatened Caribbean coral." Coral Reefs 27(3): 697–705.