The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Bagous affinis Hustache (Figure 1) is a semi-aquatic weevil that feeds on the aquatic invasive plant Hydrilla verticillata (L.f.) Royle. The larvae of the weevil mine hydrilla tubers, whereas the adults feed on the submerged stems and leaves. The weevil was discovered during surveys for biological control agents for hydrilla in Pakistan in 1980 and was first introduced to the U.S. in Florida from India in 1987. A closely related weevil from Australia, Bagous hydrillae O'Brien (Insecta: Coleoptera: Curculionidae), was introduced to the U.S. in 1991.

Credit: Gary Buckingham USDA-Agricultural Research Service
Distribution
Bagous affinis is native to India and Pakistan. It feeds on stranded hydrilla above the waterline around water bodies in areas that have a long dry season (Buckingham and Bennett 1994).
Description
Eggs
Eggs are elongated and 0.52 mm long and 0.32 mm wide (Buckingham and Bennett 1994). The egg is white, and the chorion or outer membrane is transparent (Figure 2). The larvae can often be seen moving around in mature eggs (Buckingham and Bennett 1994). Eggs are laid in waterlogged wood, hydrilla stems, and soil.

Credit: USDA Agricultural Research Service Archive, USDA-Agricultural Research Service, Bugwood.org.
Larvae
There are three larval stages, and all three instars are white with brown heads (Figure 3). The first instars hatch from the eggs and appear more dorsoventrally flattened than the later instars (Buckingham and Bennett 1994). The setae are also more noticeable in this stage (Buckingham and Bennett 1994). The instars can be differentiated by head capsule width, which in the first instar is 0.22 mm (Buckingham and Bennett 1994). The head capsule width in the second instar is 0.40 mm and that of the third instar is 0.66 mm (Buckingham and Bennett 1994). The first instar bores into the tubers of hydrilla, and larval development occurs within the tuber.

Credit: USDA-Agricultural Research Service.
Pupae
Pupae of Bagous affinis (Figure 4) are off white to yellow with small red-brown bristles on the head and dorsum (Buckingham and Bennett 1994). Pupae are 3.75 mm long and 1.81 mm wide (Buckingham and Bennett 1994). The pupae turn yellow as they approach pupation. One day before eclosion, adult features become apparent, the eyes and leg joints darken, and the tips of the mandibles turn red (Buckingham and Bennett 1994).
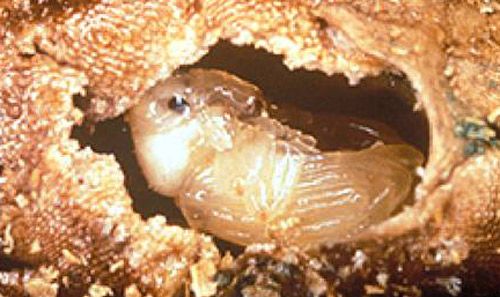
Credit: U.S. Army Engineer Research and Development Center and USDA-ARS.
Adult
Adult Bagous affinis are small brown weevils with light-colored mottling (Figure 5). Adults are on average 3.5 mm long and 1.5 mm wide (Buckingham and Bennett 1994). The males and females can be distinguished by the shape of the first abdominal sternite. The sternite is concave in males and convex in females (Buckingham and Bennett 1994).

Credit: Michael Shillingburg, Arizona State University Hasbrouck Insect Collection (ASUHIS, 2013).
Life Cycle and Biology
Life stages include the egg, three larval instars, pupa, and adult. The life cycle of Bagous affinis occurs in stranded plant material at the edge of water bodies when the water level recedes during dry periods (Buckingham and Bennett 1994). The adults prefer to lay eggs in waterlogged wood although they also will lay eggs in soil and into hydrilla stems (Buckingham and Bennett 1991). The female probes the substrate with her mouthparts and then inserts an egg into the probed area (Buckingham and Bennett 1994). In the hydrilla stems, eggs were laid most commonly near the leaf nodes. After 3-4 days (Buckingham and Bennett 1994), the first-instar larvae hatch and locate and mine the tubers of the hydrilla. They do not attack submersed hydrilla tubers, only those above the water level. Larval development, through the three instars, takes 14-15 days, and the larvae usually stay within the same tuber (Buckingham and Bennett 1994). The larvae will die if the tubers become submerged when the water level rises.
Pupation occurs in terrestrial habitats usually in stranded plant material or in the silt at the edge of the water body. The pupation period lasts for 5 days (Buckingham and Bennett 1994). The total time for immature development is around 22 days, and adults live for around 120-130 days on average (Buckingham and Bennett 1994). Successful development occurs between 64 °F (18 °C) and 90 °F (32 °C) (Godfrey and Anderson 1994a). Bagous affinis adults cannot swim, but they can remain submerged for several hours. They have been observed going underwater by clinging to hydrilla stems (Buckingham and Bennett 1994). When underwater, the weevils breathe using plastron respiration, i.e., air that is trapped close to the body by scales (Buckingham and Bennett 1994).
The larvae of a closely related species, the hydrilla stem weevil, Bagous hydrillae, feed on hydrilla stems. Although the female hydrilla stem weevil will lay eggs on tubers and the larvae will develop normally, the opposite is not true. The larvae of the hydrilla tuber weevil are not able to complete development on stem tissue and die before the prepupal stage (Wheeler and Center 2007).
Hosts
Host specificity studies were completed by Buckingham and Bennett (1998). In these tests, larvae were observed feeding on seven plant species of the 39 species in 20 families that were tested. Larvae developed on Brazilian waterweed (Egeria densa Planch.), dwarf arrowhead (Sagittaria spp.), American pondweed (Potamogeton nodosus Poir.), sago pondweed (Potamogeton pectinatus L.), Richardson's pondweed (Potamogeton richardsonii (A. Benn.) Rydb.), watercelery (Vallisneria americana Michx.), and hydrilla. However, very low levels of development occurred in all other plants compared to hydrilla. For adults, 52 plant species in 29 families were tested. With the exception of sago pondweed, feeding was limited to plants in the Hydrocharitaceae. Plants within the family Hydrocharitaceae that were fed upon by adults included Brazilian waterweed, watercelery, and frogbit (Limnobium spongia (Bosc.) Rich. ex Steud.). Females laid eggs on only three out of six species, namely Brazilian waterweed, watercelery and elodea (Elodea canadensis Michx.).
Damage
Adult Bagous affinis feed on hydrilla leaves, stems, turions, and tubers, although stems are preferred (Buckingham and Bennett 1994). When feeding, the adults often form aggregations with all individuals feeding on the same stem or tuber (Buckingham and Bennett 1994). On the stems, adults eat the tissue around the leaf nodes and often cause the stems to break. On the tubers, adults bore into and feed on the tissue as they move into the tuber. The larvae stay within the tubers, consuming plant tissue as they develop (Figure 6). This feeding degrades the tubers, so the tubers often fail to germinate once damaged.

Credit: USDA Agricultural Research Service Archive, USDA-Agricultural Research Service, Bugwood.org.
Importance as a Biological Control Agent
The hydrilla tuber weevil was selected as a potential biocontrol agent due to the organism's ability to damage hydrilla in its native range of India and Pakistan. Baloch et al. (1980) reported that Bagous affinis infested nearly all of the tubers collected at one field site in Pakistan. Infested tubers were less likely to germinate than uninfested tubers (Godfrey and Anderson 1994b).
In 1987, Bagous affinis was released in Florida and was the first insect species ever released for hydrilla control (Grodowitz et al. 1995). Releases continued in Florida until 1988. In July 1990, there was no establishment of the weevil at the release sites (Buckingham and Bennett 1994). The most likely reason for the lack of establishment of the hydrilla tuber weevil is the specific requirements of the life cycle. The larvae of Bagous affinis require relatively dry conditions for larval development and pupation. In the native range of India and Pakistan, the weevil completes its whole life cycle in stranded hydrilla at the edge of water bodies. In Florida and most of the southeastern U.S., where this insect has been released, these conditions (i.e., stranded hydrilla around water bodies) are not common and so the weevil is unable to successfully complete its lifecycle. The only evidence of field colonization, albeit temporary, was during a drawdown in a reservoir in north-central Florida (Buckingham and Bennett 1991). A drawdown is a method of hydrilla control used in water bodies with water level control structures. The water level is decreased so that the hydrilla dries out and dies. Unfortunately, the insects were killed when that water level returned to normal levels.
In 1992, it was realized that, although the necessary conditions for establishment of this species were not present in the southeastern U.S., Bagous affinis may be able to establish in other areas, specifically California. Releases were made to determine if Bagous affinis would establish and successfully overwinter in a river and a pond in northern California (Godfrey et al. 1994). No establishment occurred at the river site (Chowchilla River), but it was later discovered that not many tubers were present (Godfrey et al. 1994). At the pond site (Calaveras County), the weevils overwintered in cages over two years (Godfrey et al. 1994). When the eggs were released directly into the pond (i.e., not in cages), temporary establishment occurred over the summer with tuber damage visible on sentinel tubers (Godfrey et al. 1994). Although no adults were caught in the spring of the following year, tubers from the sentinel traps showed characteristic weevil damage (Godfrey et al. 1994).
In 1994, Bagous affinis was released at Choke Canyon Reservoir in Texas (Grodowitz et al. 1995). It was believed that this site would be conducive to weevil development as the water level could be altered and stranded tubers were visible at a rate of 90 tubers per m2 (Grodowitz et al. 1995). However, when the soil and tubers were examined three months later, no individuals were found and no damage was recorded (Grodowitz et al. 1995).
Monitoring and Management
Several methods can be used to monitor weevil activity including plant dissection and black lights. Firstly, hydrilla can be collected and the leaves examined for the characteristic damage caused by the adult weevils. Tubers may be dissected and larvae may be visualized under a light microscope. Monitoring for adult weevils can be done using ultraviolet (UV) lights (Buckingham and Bennett 1994). Management is not necessary for the hydrilla tuber weevil as it is not a pest in the U.S.
The authors would like to acknowledge funding provided by the USDA NIFA RAMP Grant 2010-02825 that helped pay for the production of this article. The authors would like to acknowledge the reviewers who provided feedback on an early draft of the article.
Selected References
Balciunas JK, Grodowitz MJ, Cofrancesco AF, Shearer JF. 2002. Hydrilla. In Van Driesche R, Blossey B, Hoddle M, Lyon S, Reardon R (editors). Biological Control of Invasive Plants in the Eastern United States [A1] , USDA Forest Service Publication FHTET-2002-04, 413P. Morgantown, WV. (8 February 2023)
Baloch GM, Sana-Ullah, Ghani MA. 1980. Some promising insects for the biological control of Hydrilla verticillata in Pakistan. Tropical Pest Management 26: 194–200
Buckingham GR, Bennett CA. 1991. Laboratory biologies of Bagous affinis and B. laevigatus (Coleoptera: Curculionidae) attacking tubers of Hydrilla verticillata (Hydrocharitaceae). Annals of the Entomological Society of America 84: 420–428.
Buckingham GR, Bennett CA. 1994. Biological and host range studies with Bagous affinis, an Indian weevil that destroys hydrilla tubers. Technical Report A-948, U.S. Army Corps of Engineers, Waterways Experiment Station, Vicksburg, MS. (27 July 2017)
Buckingham GR, Bennett CA. 1998. Host range studies with Bagous affinis (Coleoptera: Curculionidae), an Indian weevil that feeds on hydrilla tubers. Environmental Entomology 27: 469–479.
Cofrancesco A, Grodowitz M, Center T, Balciunas J. 2005. Bagous affinis - hydrilla tuber weevil. Noxious and Nuisance Plant Management Information Systems, U.S. Army Corps of Engineers, Engineer Research and Development Center, Vicksburg, MS. (27 June 2017).
Godfrey KE, Anderson LWJ. 1994a. Development rates of Bagous affinis (Coleoptera: Curculionidae) at constant temperatures. Florida Entomologist 77: 516–519.
Godfrey KE, Anderson LWJ. 1994b. Feeding by Bagous affinis (Coleoptera: Curculionidae) inhibits germination of hydrilla tubers. Florida Entomologist 77: 480–488.
Godfrey KE, Anderson LWJ, Perry SD, Dechoretz N. 1994. Overwintering and establishment potential of Bagous affinis (Coleoptera: Curculionidae) on Hydrilla verticillata (Hydrocharitaceae) in Northern California. Florida Entomologist 77: 221–230
Grodowitz J, Center T, Snoddy E. 1995. Current status of use of insect biocontrol agents for the management of hydrilla. Pages 134–141 In Proceedings of the 29th Annual Meeting of Aquatic Plant Control Research Program, 14–17 November 1994, Vicksburg, MS, Miscellaneous paper A-93-2, U.S. Army Engineer Waterways Experiment Station, Vicksburg, MS.
McCann JA, Arkin L, Williams JD. 1996. Nonindigenous aquatic and selected terrestrial species of Florida: status, pathway and time of introduction, present distribution, and significant ecological and economic effects. [A1] National Biological Service, Southeastern Biological Science Center, Gainesville, FL. (8 February 2023).
O'Brien C, Pajni HR. 1989. Two Indian Bagous weevils (Coleoptera: Curculionidae) tuber feeders of Hydrilla verticillata (Hydrocharitaceae), one a potential biocontrol agent in Florida. Florida Entomologist 72: 462–468.
Wheeler GS, Center TD. 2007. Hydrilla stems and tubers as hosts for three Bagous species: two introduced biological control agents (Bagous hydrillae and B. affinis) and one native species (B. restrictus). Environmental Entomology 36: 409–415.