The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The encyrtid parasitoid Ageniaspis citricola was first imported into Florida from Australia in 1994 in a classical biological control program against the citrus leafminer, Phyllocnistis citrella Stainton (Hoy and Nguyen 1994a). A second strain of Ageniaspis citricola was introduced into Florida from Taiwan in 1997, although there is no evidence that this second strain ever established (Hoy and Nguyen 1997). The citrus leafminer was first detected in Florida in 1993 and quickly spread throughout all 860,000 acres of citrus, posing a serious threat to the state's citrus industry. The population of A. citricola from Australia quickly established and dispersed throughout the state, reaching parasitism levels near 100% in some areas (Hoy et al. 1995a, 1995b; Knapp et al. 1995; Bullock et al. 1996; Pomerinke and Stansly 1998). Ageniaspis citricola is consistently the dominant parasitoid of citrus leafminer in Florida.
Credit: UF/IFAS Southwest Florida Research and Education Center
Distribution
Ageniaspis citricola is well-adapted to humid tropical and subtropical climates. Ageniaspis citricola was originally described from Vietnam (Logvinovskaya) and is also found in Thailand and Taiwan (Morakote and Nanta 1996a, Ujiye 1996). Through classical biological control programs, successful introductions of Ageniaspis citricola have been made in Queensland Australia, Florida, Louisiana, the Bahamas, Honduras, Brazil, and Peru. Releases have also been made in Morocco, Israel and Spain, although Ageniaspis citricola does not perform well in those arid Mediterranean climates.
When A. citricola was introduced into Florida from Australia, it was reported to disperse rapidly, up to 300 km within a year (Pomerinke and Stansly 1998). Ageniaspis citricola is now well established in all citrus-producing areas of the state.
Description
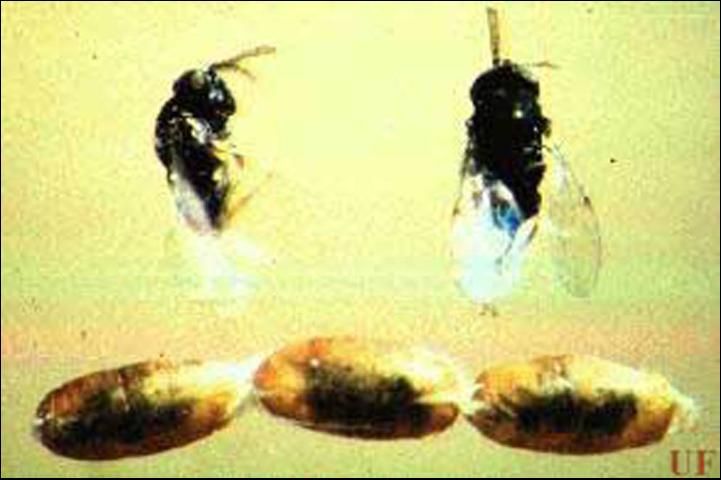
Ageniaspis citricola is approximately 0.7 to 0.8 mm long. The abdomen of the female is small and triangular, and the thorax is considerably longer than the abdomen. Both males and females are black in color, with fine silvery white pubescence, and with tarsi and portions of the tibiae colored yellow. Antennae are clubbed, 7-segmented and yellowish-brown in color, with a darker scape and pedicel.

Credit: Marjorie A. Hoy, UF/IFAS; and Ru Nguyen, Division of Plant Industry
Ageniaspis citricola pupae are small, elliptical, and linked together in chains of approximately two to eight pupae. Pupae are cream colored early in development but become darker brown and then black just before adult emergence. Pupae of A. citricola may be found by opening the pupal cell of the citrus leafminer, which often is at the leaf margin. Citrus leafminer larvae that have been parasitized by A. citricola appear slightly darkened.
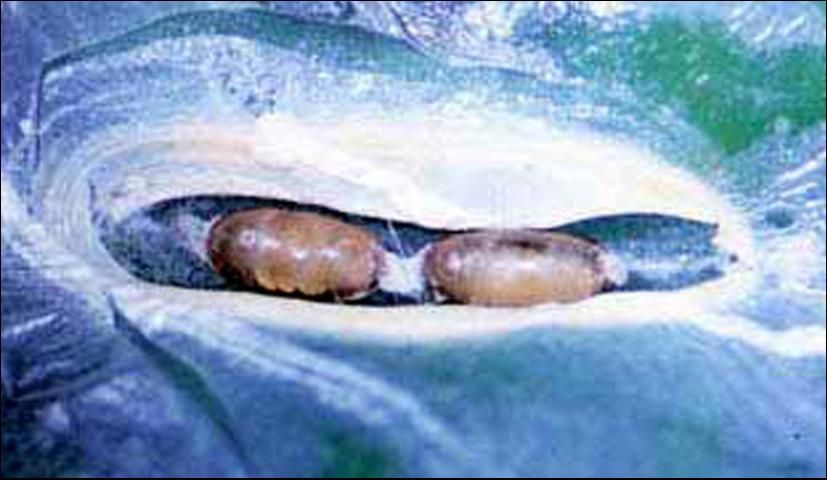
Credit: Marjorie A. Hoy, UF/IFAS; and Ru Nguyen, Division of Plant Industry
Biology
Ageniaspis citricola is a host-specific, koinobiont (not especially harmful to their host until they mature), endoparasitoid of citrus leafminer eggs and early instar larvae. All stages of the parasitoid develop within the citrus leafminer host. After being parasitized, the citrus leafminer will continue to feed and develop in leaf mines, going on to form a pupal chamber at the end of the mine. At this point, Ageniaspis citricola kills its host and pupates within the exoskeleton of the citrus leafminer in the pupal chamber. Adult Ageniaspis citricola emerge from the pupal chamber by chewing a hole in the silk holding the leaf chamber together. One female can produce one to 10 progeny per single host, with an average of 2.8 ± 1.1 Ageniaspis citricola emerging per pupal chamber under laboratory conditions (Smith and Hoy 1995).
Both females and males are produced, contrary to previous reports of thelytoky (Evans 1995). Thus, Ageniaspis citricola is arrhenotokous, meaning that unmated females will produce only haploid males. The average sex ratio in laboratory colonies is approximately 1 male: 1.8 females (Smith and Hoy 1995).
Ageniaspis citricola takes approximately 16 to 18 days to complete its life cycle at greenhouse conditions of approximately 30°C and 80 to 90% RH (Smith and Hoy 1995). Adults live an average of two to five days, with the greatest longevity at 96% humidity (Edwards and Hoy 1998).
Several authors (Neale et al. 1995, Smith and Hoy 1995, Argov and Rossler 1996, Smith and Beattie 1996, Smith and Neale 1996) have reported on the difficulties of mass-rearing Ageniaspis citricola. Adults require very high humidity levels (Yoder and Hoy 1998), are short-lived, are susceptible to desiccation and rough handling, i.e., aspiration, and are easily trapped and drowned in moisture or honey drops.
Hoy et al. (2000) and Alvarez and Hoy (2002) conducted genomic analyses (RAPD-PCR) and analyses of actin gene sequences of Ageniaspis citricola populations from Australia and Taiwan and found differences suggesting the two populations are cryptic species. The two strains were observed to have different net water loss rates in the adult stage (Yoder and Hoy 1998).
Classical Biological Control
Ageniaspis citricola was introduced into Queensland, Australia, from Thailand in 1991 to control the citrus leafminer. Based on the success of that introduction, Ageniaspis citricola was imported into Florida from Australia in 1994 upon discovery of the citrus leafminer. Approximately 3000 pupae were hand carried from Australia into quarantine facilities at the Division of Plant Industry, Gainesville, FL. The first releases of Ageniaspis citricola took place in Florida citrus groves on 30 April 1994. Parasitism rates by Ageniaspis citricola as high as 86% were observed in some groves (Hoy and Nguyen 1997, Pomerinke and Stansly 1998).
Ageniaspis citricola were subsequently shipped to Louisiana, where parasitism levels were reported to be 60 to 100% in some areas of the state within the first year (Johnson et al. 1998). Additional classical biological control programs for Ageniaspis citricola have been implemented in the Bahamas, Honduras, Brazil, and Peru.
Johnson et al. (1998) found that insecticide use may limit Ageniaspis citricola's effectiveness based on parasitism levels in routinely sprayed vs. unsprayed orchards in Louisiana. Villanueva and Hoy (1998) and Villanueva et al. (2000) evaluated the relative effects of pesticides on Ageniaspis citricola and identified several IPM-compatible pesticides for use in citrus groves and nurseries.
Ageniaspis citricola has all the attributes of a highly effective natural enemy: 1) host specificity, 2) high reproductive rate, 3) high dispersal rate, and 4) high searching rate. It has performed exceptionally well as a natural enemy given the correct climatic conditions, i.e., high relative humidity. However, several years of drought conditions in Florida had a negative effect on Ageniaspis citricola populations, resulting in parasitoid populations lagging behind leafminer populations. An additional parasitoid may be imported, evaluated, and released to complement the control provided by Ageniaspis citricola in Florida.
Selected References
Alvarez JM. 2000. Use of molecular tools for discriminating between two populations of the citrus leafminer parasitoid Ageniaspis (Hymenoptera: Encyrtidae). Ph.D. Dissertation, University of Florida, Gainesville.
Alvarez JM, Hoy MA. 2002. "Evaluation of the ribosomal ITS2 DNA sequences in separating closely related populations of the parasitoid Ageniaspis (Hymenoptera: Encyrtidae)." Annals of the Entomological Society of America 95: 250–256.
Argov Y, Rossler Y. 1996. "Introduction, release and recovery of several exotic natural enemies for biological control of the citrus leafminer, Phyllocnistis citrella, in Israel." Phytoparasitica 24: 33–38.
Beattie GAC, Smith D. 1993. Citrus Leafminer. Agfact H2.AE.4, second ed. New South Wales, Australia.
Bullock RC, Killer EE, Pelosi RR. 1996. The release and distribution of Ageniaspis citricola in St. Lucie County, Florida. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Cave R. Biological control of citrus leafminer in Honduras. 1996. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Edwards OR, Hoy MA. 1998. "Biology of Ageniaspis citricola (Hymenoptera: Encyrtidae), a parasitoid of the leafminer Phyllocnistis citrella (Lepidoptera: Gracillariidae)." Annals of the Entomological Society of America 91: 654–660.
Evans G. 1995. "Discovery of the male of Ageniaspis citricola (Hymenoptera: Encyrtidae), a parasitoid of the citrus leafminer, Phyllocnistis citrella (Lepidoptera: Gracillariidae)." Florida Entomologist 78: 134–36.
Heppner JB. 1993. "Citrus leafminer, Phyllocnistis citrella, in Florida (Lepidoptera: Gracillariidae: Phyllocnistinae)." Tropical Lepidoptera 4: 49–64.
Hoy MA, Nguyen R. 1994a. Classical biological control of the citrus leafminer in Florida. Citrus Industry (April): 22.
Hoy MA, Nguyen R. 1994b. Classical biological control of the citrus leafminer in Florida: A progress report. IPM Florida. (September 2019)
Hoy MA, Nguyen R. 1994c. "Current status of Ageniaspis citricola, a parasite of the citrus leafminer, in Florida." Citrus Industry (December): 30–32.
Hoy MA, Nguyen R. 1997. "Classical biological control of the citrus leafminer Phyllocnistis citrella Stainton." Tropical Lepidoptera 8: 1–19.
Hoy MA, Jeyaprakash A, Morakote R, Lo P, Nguyen R. 2000. "Genomic analyses of two populations of Ageniaspis citricola (Hymenoptera: Encyrtidae) suggest that a cryptic species may exist." Biological Control 17: 1–10.
Hoy MA, Johnson S, Nguyen R. 1996. Classical biological control of the citrus leafminer: In theory and practice. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Hoy MA, Nguyen R, Hall D, Bullock R, Pomerinke M, Pena J, Browning H, Stansly P. 1995a. "Establishment of citrus leafminer parasitoid, Ageniaspis citricola, in Florida." Citrus Industry (December): 12–17.
Hoy MA, Nguyen R, Hall D, Bullock R, Pomerinke M, Pena J, Browning H, Stansly P. 1995b. "Distribution and abundance of Ageniaspis citricola, a parasite of the citrus leafminer in Florida." Citrus Industry (May): 51–52.
Johnson SJ, Vaughn A, Bourgeois WJ. 1996. Rearing and release methods for Ageniaspis citricola for a classical biocontrol program of the citrus leafminer in Louisiana. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Johnson SJ, Vaughn A, Bourgeois WJ. 1998. Biological control of the citrus leafminer in Louisiana. Louisiana Agriculture Summer 1998, pp. 11–14.
Knapp J, Pena J, Stansly P, Heppner J, Yang Y. 1993. Citrus leafminer, a new pest of citrus in Florida. Citrus Industry (October): 42–43.
Knapp, J, Albrigo LG, Browning HW, Bullock RC, Heppner J, Hall D, Hoy MA, Nguyen R, Pena J, Stansly P. 1995. Citrus leafminer, Phyllocnistis citrella Stainton: Current status in Florida—1995. Gainesville: University of Florida Institute of Food and Agricultural Sciences. 35 pp.
Logvinovskaya TV. 1983. "A new species of Ageniaspis Dahlbom 1857 (Hymenoptera: Encyrtidae) from Vietnam." Entomological Review 62: 150–152.
Morakote R, Nanta P. 1996a. Managing the citrus leafminer in Thailand. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Morakote R, Nanta P. 1996b. Natural enemies of citrus leafminer. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Neale C, Smith D, Beattie GAC, Miles M. 1995. "Importation, host specificity, testing, rearing and release of three parasitoids of Phyllocnistis citrella Stainton (Lepidoptera: Gracillariidae) in eastern Australia." Journal of the Australian Entomological Society 34: 343–348.
Pomerinke MA, Stansly PA. 1996. Establishment and dispersal of Ageniaspis citricola in southwest Florida. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Pomerinke MA, Stansly PA. 1998. "Establishment of Ageniaspis citricola (Hymenoptera: Encyrtidae) for biological control of Phyllocnistis citrella (Lepidoptera: Gracillariidae) in Florida." Florida Entomologist 81: 361–372.
Smith JM, Hoy MA. 1995. "Rearing methods for Ageniaspis citricola (Hymenoptera: Encyrtidae) and Cirrospilus quadristriatus (Hymenoptera: Eulophidae) released in a classical biological control program for citrus leafminer." Phyllocnistis citrella (Lepidoptera: Gracillariidae)." Florida Entomologist 78: 600–608.
Smith D, Neale C. 1996. Rearing techniques for citrus leafminer parasitoids in Queensland. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Smith D, Beattie GAC. 1996. Australian citrus IPM and citrus leafminer. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Ujiye T. 1996. Parasitoid fauna of the citrus leafminer in Japan, Taiwan and Thailand. In Hoy MA (ed.) Managing the citrus leafminer, Proceedings of the International Conference, Orlando, Fl. April 2–25, 1996. University of Florida, Gainesville.
Villanueva-Jimenez JA, Hoy MA. 1998. "Toxicity of pesticides to the citrus leafminer and its parasitoid Ageniaspis citricola evaluated to assess their suitability for an IPM program in citrus nurseries". BioControl 43: 357–388.
Villanueva-Jimenez JA, Hoy MA, Davies FS. 2000. "Field evaluation of integrated pest management-compatible pesticides for the citrus leafminer Phyllocnistis citrella (Lepidoptera: Gracillariidae) and its parasitoid Ageniaspis citricola (Hymenoptera: Encyrtidae)." Journal of Economic Entomology 93: 357–367.
Yoder JA, Hoy MA. 1998. "Differences in water relations among the citrus leafminer and two different populations of its parasitoid inhabiting the same apparent microhabitat." Entomologica Experimentalis et Appliciata 89: 169–173.