What is bluetongue?
Bluetongue is a disease of ruminant livestock that is caused by the bluetongue virus. Bluetongue virus does not cause disease in humans. This virus is transmitted to livestock by selected species in the genus Culicoides (Diptera: Ceratopogonidae). These small insects are biting midges and in some areas are called no-see-ums, sand flies or punkies. Bluetongue virus was among the first recognized insect borne viruses. The disease was first described in livestock in South Africa in 1902. Bluetongue virus is widely distributed throughout the tropical, subtropical and temperate regions of the world wherever different competent species of Culicoides occur. Bluetongue outbreaks can result in millions of dollars in losses due to the impact on livestock health and notably in the loss of markets for livestock due to regulatory restrictions on animal movement to limit the spread of the disease.
The virus
Bluetongue virus, or BTV, is a member of the Orbivirus genus in the family Reoviridae. BTV is related to other orbiviruses that also cause disease in animals like African horse sickness and epizootic hemorrhagic disease viruses. The orbiviruses contain genomes that are in the form of RNA segments. BTV occurs in the form of 24 different serotypes (a serological form of the virus) and these serotypes are distributed in different regions of the world. It is thought that the distribution of the different serotypes in different regions is influenced by the different species of Culicoides that are present in specific regions. For example, historically only five BTV serotypes, BTV-2, 10, 11, 13, and 17 have occurred in the US Surveillance in the Caribbean Basin showed the presence of BTV-1, 3, 4, 6, 8, 8, 12, 14, and 17. It is believed that the difference in the types of serotypes in these two regions is due to different Culicoides vectors in the two regions. In the US Culicoides sonorensis is the primary vector, while in the Basin region the vector is Culicoides insignis. In other regions of the world, i.e., Africa, South America, Asia, and Europe, there are additional BTV serotypes and each region has different species of Culicoides that are the primary vectors.
What are the clinical signs of bluetongue in livestock?
Bluetongue virus may infect several different domestic and wild ruminant species including cattle, deer, goats, and sheep. However clinical signs are most apparent in sheep where signs include 5–7 days of increasing temperature (up to 107.6°F), excessive salivation, hyperemia and swelling of the nasal and buccal mucosa, hemorrhaging in the mucosal membranes in the mouth, oral erosions, swollen tongue, loss of wool, depression and hemorrhages in the coronary bands of the hoof that often result in lameness, and difficulty standing (Figure 1).

Credit: A. J. Leudke, USDA/ARS
Sheep with lesions in the mouth and esophagus may vomit and this may lead to aspiration of the ruminal contents, pneumonia and death. Bluetongue can be a severe disease in sheep with mortality varying from 5–80% depending on the virus serotype and on the immune status of the sheep populations.
Although cattle can serve as reservoirs for BTV, clinical disease in cattle has generally been rare. When this has been encountered, clinical disease is usually evident in fewer than 5% of infected animals, and there is very rarely any mortality. Cattle develop a prolonged viremia lasting several weeks. This is believed to enable the virus to survive during cold winters in the absence of insects, or at other times when vector populations are absent or reduced. Therefore, cattle are considered reservoirs for BTV and a means of introducing BTV into different areas through cattle movement and trade. Early prenatal infection with BTV may lead to embryonic death in cattle so that epidemics can influence cattle reproduction. However cattle fetuses infected later in gestation usually survive without persistent infection and these animals develop antibodies to the specific infecting virus serotype.
Impact
Bluetongue can have a severe impact on the livestock economy of a country or region. The disease itself can cause catastrophic losses to sheep populations. Although mortality to bluetongue is often low, it is not uncommon to see mortality rates approaching 50%–100% in susceptible flocks, and there are other losses due to morbidity and the need to provide care for sick animals. Costs associated with morbidity of sick animals include weight loss, reduced milk yield, abortion and associated veterinary costs.
During a BTV-2 epidemic in Italy in 2000–2001, approximately 263,000 diseased sheep and goats were reported (18% morbidity) and 48,000 sheep and goats died (3% mortality). During a second epidemic in 2001–2002, approximately 251,000 diseased sheep and goats were reported (18% morbidity) and 73,000 sheep and goats died (5% mortality) (Calistri 2004). In 2007 BTV-8 outbreaks occurred on over 20,000 farms in Germany with disease in ca, 35,000 cattle, sheep or goats.
The indirect costs associated with bluetongue can be much greater than the direct costs. Bluetongue is a "List A" disease of the Office of International Epizootics (OIE). List A diseases are those diseases which can spread rapidly and that have a considerable impact on the health of livestock. List A diseases are notifiable to the OIE, and often used as non-tariff trade barriers to prevent livestock from moving and potentially spreading disease. Livestock from bluetongue endemic areas, or regions where there are outbreaks, can be prevented through regulatory policy from moving to regions considered bluetongue-free. The economic impact in reducing livestock movement and trade can be considerable. Additional costs for bluetongue come from the required testing for the virus in animals being considered for movement.
The impact on cattle industries is through the effects of trade barriers. Bluetongue positive cattle are not allowed to be moved from outbreak areas because of the prolonged viremias. Cattle are capable of infecting Culicoides and spreading the virus for as along as a month after infection.
In 2007, a BTV-8 outbreak in France was estimated to cost $1.4 billion. Losses were largely due to the inability to trade cattle, a very substantial industry in France, on the international market. In 2007 a BTV-8 outbreak in the Netherlands cost approximately $85 million. The southern regions of the US have been endemic for certain BTV serotypes for many years though animal disease has been rare. Nevertheless the impact on the US has been losses due to restrictions on the international cattle trade in livestock and germ plasm to regions considered bluetongue-free like some countries in the European Union. The US losses in trade and associated testing of cattle for BTV status has been estimated at $130 million annually.
How is bluetongue transmitted to livestock?
The cycle of transmission is from a competent Culicoides to a ruminant animal, back to a susceptible Culicoides, to a ruminant and back to a Culicoides, etc. The female Culicoides requires a blood meal in order to lay eggs. Different Culicoides species prefer different types of habitats to lay eggs. Culicoides sonorensis prefers heavily organic rich water, with high manure content for egg laying and subsequent larval development. Some African Culicoides vectors lay eggs directly in the deposited dung of ruminants. Both types of insects, therefore, lay their eggs and complete larval development near ruminant livestock habitat.

Credit: E. T. Schmidtmann, USDA/ARS
How is bluetongue diagnosed?
In regions with known transmission, Bluetongue can be diagnosed based on the characteristic lesions in susceptible animals. However, the diagnosis should always be confirmed by virus isolation and identification using any of a variety of available methods capable of detecting BTV in blood. A rise in BTV antibody may also be used to confirm a diagnosis.
There are many immunological tests that can be used including enzyme immunoabsorbant assay, cell culture or inoculation of the blood into embryonated chicken eggs. Faster identification of the BTV in blood or other infected tissue can be achieved using polymerase chain reaction methods capable of detecting virus RNA.
What is the cure?
As with the other arthropod-borne viruses, there is no "cure" or effective treatment for bluetongue disease other then treating the clinical signs in an attempt to reduce the severity.
Where does bluetongue occur?
BTV occurs wherever there are species of Culicoides capable of transmitting it to animals. Historically BTV serotypes occur in Africa, Asia, South America, North America, Middle East, India, Australia (Figure 3).
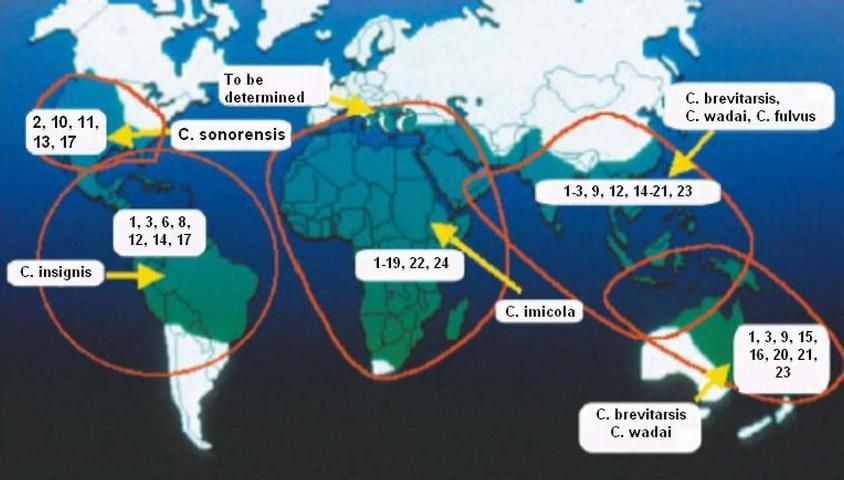
Credit: W. J. Tabachnick, UF/IFAS
US BTV serotypes 1,10,11,13 and 17 have been found in the southern and western states where they are transmitted by Culicoides sonorensis (Figure 2). The northern states do not support Culicoides sonorensis populations, and this region of the U.S. and adjacent regions of Canada are considered BTV-free.
Europe and the Mediterranean Basin had been BTV free until the beginning of 2001 when BTV serotypes began to appear in Italy causing outbreaks and considerable economic losses in sheep populations. Substantial outbreaks of BTV-8 occurred in northern Europe livestock during 2006 and 2007 transmitted by European Culicoides for the first time. In 2007 BTV in the form of BTV-8 entered the United Kingdom causing outbreaks on farms in Southern England. The reason for the expansion of the range of BTV into Europe is unknown.
The recent spread of different BTV serotypes to northern Europe is alarming. The spread has been facilitated by a few species of northern European Culicoides. It is also alarming that these outbreaks have been accompanied by the appearance of more clinical cases of bluetongue in cattle than had ever been seen elsewhere.
What about Florida?
Culicoides sonorensis occurs throughout Florida. The South American vector of BTV, Culicoides insignis, occurs in the southern half of Florida. As a result Florida livestock are exposed to BTV. Tests on about 240 Florida cattle showed that approximately 40% of Florida cattle had antibody to BTV signifying that they had been previously infected with either BTV-10, 11, 13, or 17 (Metcalf et al. 1981). Fortunately clinical disease has not occurred, but Florida cattle are restricted in international trade to BTV-free countries.
In 2001 tests on a few sick sheep, goats and two cattle resulted in the first identifications of new BTV serotypes in the US. Between 2001 and 2007, 18 identifications were made and included the following exotic BTV serotypes: BTV 3, 5, 6, 14, 19, 22. It is noteworthy that two cattle were found with clinical signs. How these serotypes entered Florida, where they came from, how far they have spread, what their effect will be on livestock health and well being in Florida and the rest of the US is unknown.
How can it be prevented?
The most effective preventive measure to protect livestock from BTV is through vaccination. There are a variety of different BTV vaccines available but to be effective the vaccine must be specifically for the serotype that is being transmitted and causing the cases in the field. Vaccines exist in a variety of forms. Modified live or attenuated vaccines are available for several different serotypes and have been widely used in South Africa. Unfortunately, though effective and cheap to produce these vaccines produce viremia in vaccinated animals that can infect Culicoides, and there is evidence that the modified virus can revert to virulence. In addition attenuated vaccines can cause reproductive problems if given to sheep during reproduction. Killed vaccines consist of completely inactivated virus and are available.
Culicoides control through adult suppression, larval control through reduction of larval habitat, housing animals indoors when Culicoides activity is highest or using repellents to protect animals from attack have not been effective in mitigating outbreaks of bluetongue.
References
Calistri, P., Giovannini, A., Conte, A., Nannini, D., Santucci, U., Patta, C., Rolesu, S., and Caporale, V. 2004. Bluetongue in Italy. Vet. Ital. 40: 243–251.
MacLachlan, N.J. and Pearson, J.E. (Eds.). 2004. Bluetongue. Proceedings of the Third International Symposium. Vet. Ital. 40 (3): 1–730.
Metcalf, H.E., Pearson, J.E. and Klingsporn, A.L. 1981. Bluetongue in cattle: A serological survey of slaughter cattle in the United States. Amer. J. Vet. Med Res. 42: 1057–1061.
Tabachnick, W.J. 1996. Culicoides variipennis and bluetongue-virus epidemiology in the United States. Ann. Rev. Entomol. 41: 23–43.
Walton, T.E. and Osburn, B.I. (Eds.). 1992. Bluetongue, African Horse Sickness and Related Orbiviruses. Proceedings of the Second International Symposium. CRC Press, Boca Raton. 1042 pp.