Introduction
Drosophila or pomace flies are small insects commonly found in association with over-ripened or rotten fruits and vegetables. There are approximately 1,500 known species in the genus Drosophila (Markow and O'Grady 2006). Drosophila flies are sometimes called small fruit flies. However, true fruit flies belong to the family Tephritidae. Not only are they larger, but they are common and often important agricultural pests (Green 2002). The vast majority of Drosophila flies are associated with rotten or over-ripened fruits and are nuisance pests. However, a few species such as the spotted-wing drosophila, Drosophila suzukii (Matsumura), can infest un-ripened fruits and are of economic significance. First detected within the continental United States in August 2008, Drosophila suzukii has become a serious threat to fruit crops.
Distribution
Drosophila suzukii is native to southeast Asia and is widely distributed in China, India, Korea, Myanmar, Russia, and Thailand (Toda 1987, Oku 2003, Hauser et al. 2009). Within Europe, this species is also widely distributed in France, Italy and Spain (European and Mediterranean Plant Protection Organization 2010). Within the United States, it has been established in Hawaii since 1980 (Kaneshiro 1983). In the continental US, it was first detected in Santa Cruz County, California, on caneberry and strawberry plants (Bolda et al. 2010). Since then, it has rapidly spread to new areas including Florida, Louisiana, Michigan, North Carolina, Oregon, South Carolina, Utah, Washington, and Wisconsin in the US, and British Columbia in Canada (Walsh et al. 2011).
Description
Adults
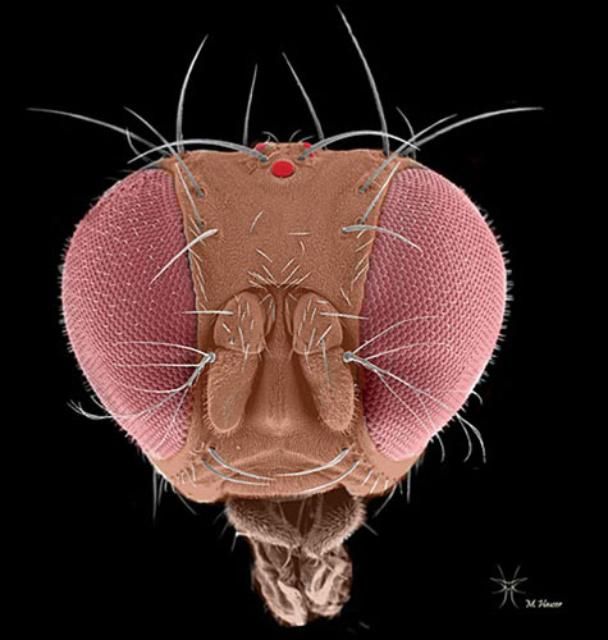
Florida is home to at least 27 addiional Drosophila spp. including Drosophila melanogaster. Some of these could easily be confused with Drosophila suzukii due to their spotted-wings. Therefore, expert examination by a specialist is needed for positive identification and confirmation (Steck et al. 2009).
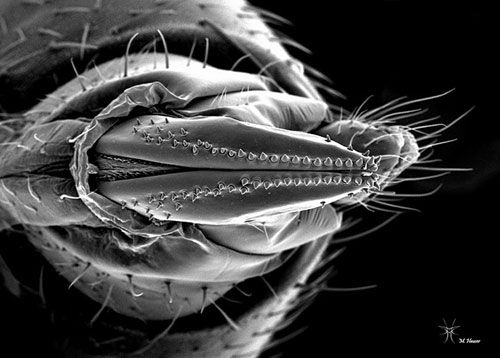
Drosophila suzukii adults are small (3–4 mm) yellowish-brown flies with red eyes. The adults have a pale brown or yellowish-brown thorax with black bands on the abdomen. The antennae are short and stubby with branched arista. Males have a distinguishing dark spot along the front edge of each wing. Spotless males are also possible, but are rarely observed in the field and should be verified by a taxonomist for positive identification. In addition, males have two rows of combs on each fore tarsus which are absent in females.

Credit: Martin Hauser, California Department of Food and Agriculture

Credit: Martin Hauser, California Department of Food and Agriculture
Credit: Martin Hauser, California Department of Food and Agriculture

Credit: Martin Hauser, California Department of Food and Agriculture
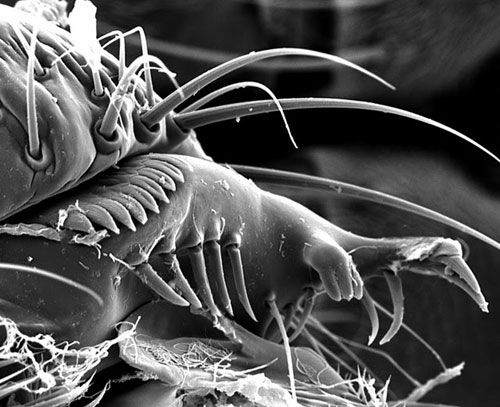
The females have a serrated ovipositor with which they penetrate the fruit skin. The serrations are much darker than the rest of ovipositor (Kanzawa 1939; Walsh et al. 2011).

Credit: Martin Hauser, California Department of Food and Agriculture

Credit: Martin Hauser, California Department of Food and Agriculture
Credit: Martin Hauser, California Department of Food and Agriculture
Credit: Martin Hauser, California Department of Food and Agriculture
Eggs
The eggs are translucent, milky-white, and glossy. The eggs develop and hatch within the fruit in which they are laid (Walsh et al. 2011).
Larvae
The larvae are milky-white and cylindrical with black mouthparts. The body is tapered anteriorly with elevated posterior spiracles. First instar larvae are approximately 0.07 mm in length. Internal organs of larvae are visible after it has consumed some fruit. Larval development occurs inside the fruit and develops through three instars before pupation. Mature larvae may grow up to 6 mm in length (Kanzawa 1939; Walsh et al. 2011).

Credit: Whitney Cranshaw, Colorado State University, Bugwood.org
Pupae
The pupae are cylindrical, reddish brown with two small projections at the end. They are approximately 2–3 mm long. Pupation can occur either inside or on the exterior of fruit.

Credit: Whitney Cranshaw, Colorado State University, Bugwood.org
Life Cycle
Drosophila suzukii prefers a moderate climate but can also survive in cold conditions (Kanzawa 1939). The flies are most active at 20°C (68°F). Activity becomes reduced at temperatures above 30°C (86°F) or below freezing. However, Drosophila suzukii is firmly established on the island of Hokkaido in Japan where winters average -4 to -12°C (Kimura 2004), suggesting the possibility of its establishment in cooler climates. Preliminary research from Oregon suggests that Drosophila suzukii larvae, pupae, and adults have the potential to survive fluctuating overwintering conditions for periods up to 60 days. Adults are able to withstand longer periods of cold conditions than larvae or pupae (Walsh et al. 2011). In general, Drosphila spp. are very sensitive to desiccation. However, sensitivity to desiccation depends upon climatic conditions and flies could develop desiccation resistance over time (Davidson 1990; Bradley et al. 1999).
Like most Drosophila spp., reproduction in Drosophila suzukii is rapid. The total life cycle may be completed within one to two weeks depending upon the climatic conditions (Kanzawa 1939). Adult life span is about three to nine weeks. However, flies emerging late in the season overwinter and may live longer. In Japan, the fly produces 10 to 13 generations (Kanzawa 1939). Flies could survive up to 10 generations per year under California climate conditions (Walsh et al. 2011). The egg, larval and pupal stages last from 1–3, 3–13, and 4–5 days, respectively (Kanzawa 1939). Degree-day models on Drosophila suzukii suggest that the entire life cycle (egg to egg laying female) can be completed within 12 to 15 days at 18.3°C (65°F) or a little more than a week at 21.1°C (70°F) (Walsh et al. 2011).
A single female can lay one to 60 eggs per day and 200–600 eggs in her lifetime. A female lays approximately one to three eggs per oviposition site. The eggs are laid singly and are randomly distributed on fruits. Multiple clutches of larvae may be present on the same fruit because females may oviposit on the same fruit (Kanzawa 1939; Mitsui et al. 2006, Walsh et al. 2011).
Hosts
Drosophila suzukii has a wide host range, including:
-
Cornus kousa, dogwood
-
Eugenia uniflora, Surinam cherry
-
Fragaria ananassa, strawberries
-
Morus spp., mulberry
-
Murraya paniculata, orange jasmine
-
Myrica rubra, Chinese bayberry
-
Prunus spp.—P. avium, sweet cherries; P. domestica, plums; P. persica, peaches
-
Pyrus pyrifolia, Asian pears
-
Ribes spp., currants
-
Rubus spp.—R. armeniacus, Himalayan blackberries; R. loganobaccus, loganberries; R. idaeus, raspberries; R. laciniatus, evergreen blackberries; R. ursinus, marionberries
-
Vaccinium spp., blueberry, cranberry
-
Vitis vinifera, wine grape
The following hard fruits may be attacked if the skin is already broken:
-
Actinidia spp., kiwi
-
Diospyros kaki, persimmons
-
Eriobotrya japonica, loquat
-
Ficus carica, fig
-
Lycopersicon spp., tomato
-
Malus domestica, apple
-
Pyrus spp., pear
Damage
Fruit infestation is initially manifested by scars on the fruit surface left by ovipositing females (stinging). The initial oviposition site becomes sunken. Damage is caused primarily by larvae feeding on fruit pulp turning the fruit flesh brown and soft. The larger larvae cut breathing holes in fruit. Infested fruit collapse around the feeding site very rapidly and rot due to mold or secondary infections (Beers et al. 2010).

Credit: Martin Hauser, California Department of Food and Agriculture
Management
Monitoring and Trapping
Management practices for spotted-wing drosophila are similar to those used to manage common Drosophila flies. Monitor adult fly populations before fruit begin ripening and before flies begin laying eggs. Bucket-style traps or quart containers used for monitoring other Drosophila flies can be effectively used for monitoring Drosophila suzukii. Mixtures of yeast, sugar, and water; fruit purees, distillates from apple cider vinegar or wine; ethanol, acetic acid, and phenylethanol in 1:22:5 ratios are potent baits for monitoring Drosophila suzukii populations. However, traps baited with yeast-sugar-water mixtures facilitate easy identification of flies because of the clear color of the bait material. Addition of a small drop of dish soap as a surfactant or placement of a sticky card within the traps improves trap efficiency by retaining the flies which have already entered the traps. The traps perform best when deployed under cool and shady areas in the field (Walsh et al. 2011).

Credit: Whitney Cranshaw, Colorado State University, Bugwood.org
Cultural Control
Good field sanitation is critical to prevent further spread of flies. Ripe fruit should be picked frequently to minimize population buildup. All damaged fruit should be removed from the field and destroyed, either by burial or disposal in closed containers. Drosophila flies are weak fliers but spread easily through infested fruits or by wind. Therefore, all nearby sources of fruit should be managed to eliminate flies (Ministry of Agriculture and Lands, British Columbia 2009).
Biological Control
Parasitoids from the families Braconidae and Cynipidae are potential biocontrol agents of Drosophila suzukii. Orius insidiosus has been reported to feed exclusively on Drosophila suzukii. (Kanzawa 1939; Dubuffett et al. 2009; and Walsh et al. 2011). However, the role of beneficial organisms in management of Drosophila suzukii has not been fully ascertained (Walsh et al. 2011).
Chemical Control
Insecticide sprays effective against other Drosophila flies are also effective against Drosophila suzukii adults. However, Drosophila suzukii's preference for ripening fruit necessitates use of chemicals with shorter pre-harvest intervals (Walsh et al. 2011).
Selected References
Beers EH, Smith TJ, Walsh DB. (August 2010). Spotted wing drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilida). Orchard Pest Management Online. http://treefruit.wsu.edu/crop-protection/opm/spotted-wing-drosophila/ (18 January 2023).
Bradley TJ, Williams AE, Rose MR. 1999. Physiological responses to selection for desiccation resistance in Drosophila melanogaster. Integrative and Comparative Biology 39: 337–345.
Bolda MP, Goodhue RE, Zalom FG. (February 2010). Spotted wing drosophila: potential economic impact of a newly established pest. Agriculture and Resource Economics Update. https://s.giannini.ucop.edu/uploads/giannini_public/81/fe/81feb5c9-f722-4018-85ec-64519d1bbc95/v13n3_2.pdf (18 January 2023).
Davidson JK. 1990. Nonparallel geographic patterns for tolerance to cold and desiccation in Drosophila melanogaster and Drosophila simulans. Australian Journal of Zoology 38: 155–161.
De Camargo R, Phaff HJ. 1957. Yeasts occurring in Drosophila flies and in fermenting tomato fruits in Northern California. Journal of Food Science 22: 367–372.
Dubuffet A, Colinet D, Anselme C, Dupas S, Carton Y, Poirié M. 2009. Variation of Leptopilina boulardi success in Drosophila hosts: what is inside the black box? Advances in Parasitology 70: 147–88.
Green MM. 2002. It really is not a fruit fly. Genetics 162: 1–3.
European and Mediterranean Plant Protection Organization. (February 2010). https://gd.eppo.int/taxon/DROSSU (18 January 2023).
Hauser M, Gaimari S, Damus M. (October 2009). Drosophila suzukii new to North America. North American Dipterists Society. http://www.nadsdiptera.org/News/FlyTimes/issue43.pdf (29 April 2011).
Kaneshiro KY. 1983. Drosophila (Sophophora) suzukii (Matsumura). Proceedings of Hawaiian Entomological Society 24: 179.
Kanzawa T. 1939. Studies on Drosophila suzukii Mats. Kofu. Review of Applied Entomology 29: 622.
Kimura MT. 1988. Adaptations to temperate climates and evolution of over-wintering strategies in the Drosophila melanogaster species group. Evolution 42: 1288–1297.
Markow TA, O'Grady PM. 2006. Drosophila: A Guide to Species Identification and Use. Academic Press, London. 272 pp.
Ministry of Agriculture and Lands, British Columbia. (December 2009). Spotted wing drosophila (Fruit fly) Pest alert. https://www2.gov.bc.ca/gov/content/industry/agriculture-seafood/animals-and-crops/plant-health/insects-and-plant-diseases/tree-fruits/spotted-wing-drosophila (18 January 2023).
Mitsui H, Achterberg VK, Nordlander G, Kimura MT. 2007. Geographical distributions and host associations of larval parasitoids of frugivorous Drosophilidae in Japan. Journal of Natural History 41: 1731–1738.
Molina JJ, Harisson MD, Brewer JW. 1974 Transmission of Erwinia carotovora var. atropeptica by Drosophila melanogaster Meig. American Potato Journal 51: 245–250.
Oku T. 2003. SWD: Drosophila suzukii (Matsumura) in Japan. Agricultural Pest Encyclopedia. Zenkoku Noson Kyoiku Kyokai 381 pp.
Price JF, Nagle CA. (November 2010). Spotted wing drosophila new in Florida berry culture. EDIS. https://edis.ifas.ufl.edu/publication/IN839 (29 April 2011).
Steck GJ, Dixon W, Dean D. (August 2009). Spotted wing drosophila, Drosophila suzukii (Matsumura) (Diptera, Drosopilidae), a fruit pest new to North America. FDOACS-Division of Plant Industry. https://www.fdacs.gov/content/download/66350/file/Pest_Alert_-_Spotted_Wing_Drosophila_-_Drosophila_suzukii.pdf (18 January 2023).
Toda MJ. 1987. Vertical microdistribution of Drosophilidae (Diptera) within various forests in Hokkaido. III. The Tomakomai Experiment Forest, Hokkaido University. Research Bulletin of College Experiment Forests. 44: 611–632.
Walsh D.B. (2009). Spotted wing drosophila could pose threat for Washington fruit growers. Washington State University Extension. http://sanjuan.wsu.edu/Documents/SWD11.09.pdf (29 April 2011).
Walsh DB, Bolda MP, Goodhue RE, Dreves AJ, Lee J, Bruck DJ, Walton VM, O'Neal SD, Frank G Z. 2011. Drosophila suzukii (Diptera: Drosophilidae): Invasive pest of ripening soft fruit expanding its geographic range and damage potential. Integrated Pest Management 106: 289–295.