Introduction
Roses are one of the most popular ornamental flowering shrubs grown worldwide. In the United States, the total wholesale production of roses accounts for $204 million annually (USDA 2015). Rose mosaic virus disease is one of the most economically important diseases among viral diseases affecting roses, because a single symptomatic leaf can result in the rejection of complete shipments for wholesale or retail rose producers and can increase the cost due to the need for re-planting with healthy roses for landscapers and homeowners (Paret et al. 2014). First described by R. P. White in 1928 as a "chlorosis of roses" (White 1928), rose mosaic virus disease continues to be a problem in nursery production and landscapes.
Causal Agent(s)
Rose mosaic virus disease is generally associated with mixed infections of viruses that belong to two taxa: Ilarvirus and Nepovirus. Within the genus Ilarvirus, the most common are Prunus necrotic ringspot virus (PNRSV) and Apple mosaic virus (ApMV). PNRSV has been isolated worldwide and is the most commonly found in the United States (Horst and Cloyd 2007). ApMV has been isolated mainly in the US. Among the genus Nepovirus, Arabis mosaic virus (ArMV) and Strawberry latent ringspot virus (SLRSV) have also been found, alone or in complexes with Ilaviruses, but are not believed to exist in US-grown roses (Manners 1997). Other nepoviruses, specifically Tobacco ringspot virus (TRSV) and Tomato ringspot virus (ToRSV), have also been isolated from symptomatic roses in the US (Horst and Cloyd 2007).
Symptoms
The symptoms associated with rose mosaic are highly variable and depend primarily upon the rose cultivar, virus complex, and the environment (Manners 1985; Porter and Tan 2012) (Figures 1‒12). Some of the more common foliar symptoms include chlorotic line patterns (Figure 1, 2, 3 and 9), ring spots (Figure 4), yellow vein banding and puckering (Figure 5), severe distortion of leaves (Figure 6), mild mottling of the leaves (Figure 7 and 8), mottling symptoms (Figure 10 and 11), and intense yellow spot symptom (Figure 12). The leaf blade around the chlorotic area may be puckered or distorted and may appear as an oak-leaf pattern, and the chlorotic sections can occasionally appear red or orange as well.

Credit: Susannah da Silva

Credit: Binoy Babu

Credit: Binoy Babu

Credit: Binoy Babu

Credit: Binoy Babu

Credit: Binoy Babu

Credit: Binoy Babu

Credit: Susannah da Silva

Credit: Susannah da Silva

Credit: Hank Dankers

Credit: Susannah da Silva

Credit: Susannah da Silva
Color-breaking of flowers has also been reported in some cultivars. Sometimes, only a portion of the plant appears to be symptomatic, and, in some cases, the infected plants never express symptoms. Foliar symptoms usually appear in the spring and remain throughout the growing season, although symptom expression can vary radically over time.
Previous researchers have shown that infected plants have decreased vigour, reduced blossom quality and quantity, reductions in transplant survival rates, early autumn leaf drop, and are more susceptible to winter-kill (Cochran 1972, 1982, 1984; Secor et al. 1977; Thomas 1982, 1984). The symptoms associated with the disease are highly variable depending on the cultivar and are influenced by weather and growing conditions. The infected plants may appear to be quite healthy for much of the year. These long-term weaknesses of rose mosaic-infected stock, combined with any number of the short-term, visually apparent symptoms discussed above, may potentially make the plants unsaleable by the rose growers and nurseries, leading to major limitations in marketability.
Mode of Transmission
The major source of rose mosaic virus disease transmission occurs through the budding or grafting of infected buds or scions onto healthy plants (Figure 13) (Manners 1985; Porter and Tan 2012). Healthy buds or scions propagated on rose mosaic infected rootstock will also result in an infected plant. Cuttings from infected plants, as well as budded plants produced from infected scions, will have greater chance of infection. Spread of rose mosaic virus disease has been demonstrated on very closely spaced plants through rare natural root grafts (Golino 2007). Such a spread has never been observed in a garden; however, in the past, transmission of rose mosaic virus disease through aphids, thrips, pruning shears, contaminated soil, and root contact have all been suggested (Cochran 1988; Davidson 1988; Manners 1988). In more recent studies, there has been no scientific evidence to support the transmission of rose mosaic through these methods (Golino et al. 2007; Manners 1997).
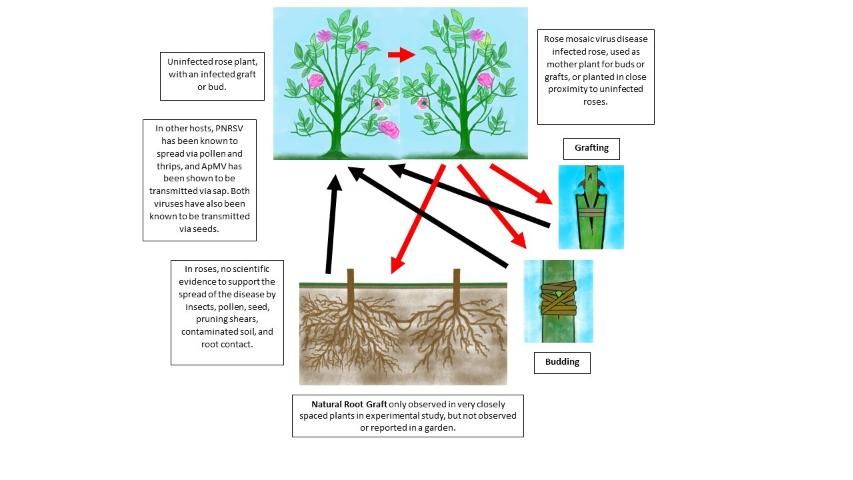
Credit: Susannah da Silva, Mathews Paret
Management
There is no known resistance to rose mosaic in commercial cultivars. Once the plant is infected, there is no easy cure. When purchasing new roses, buy only certified disease-free roses and carefully check the leaves for any characteristic symptoms of rose mosaic, which will be most apparent in the spring season. Application of heat therapy has been efficiently used in the propagation of rose mosaic-free plants (Manners 1985). Axillary buds from infected plants are heated at a temperature of 38°C for 28‒35 days, then budded onto index plants to determine if they are disease-free; these buds can be used for the budding and subsequent propagation of rose mosaic free plants. However, success and survival rates of the buds vary. Infected portions of the plant can be pruned; however, this only removes the symptoms: the plant is systemically infected and may eventually develop symptoms on other leaves. Infected plants should be removed and destroyed if they are not performing up to desired levels, but if they are far from other plants, they are not a risk for spreading the disease to healthy plants based on current scientific information. While there is no proof of the transmission of the viruses causing rose mosaic through pruning or propagation tools, it is still important to sanitize tools consistently when working with roses to minimize any potential risks.
References
Cochran, L. C. 1972. "Virus diseases of roses." In Amer. Rose Ann. Shreveport, LA: Amer. Rose Soc.
Cochran, L. C. 1982. "Rose mosaic: it can be controlled." In Amer. Rose Ann. Shreveport, LA: Amer. Rose Soc.
Cochran, L. C. 1984. "Let's get rid of rose mosaic." Amer. Rose Magazine.
Cochran, L.C. 1988. "Letter to the editor." The Amer. Rose Magazine.
Davidson H. 1988. "Letter to the editor." The Amer. Rose Magazine.
Golino, D. A., S. T. Sim., M. Cunningham and A. Rowhani 2007. "Transmission of rose mosaic viruses." ISHS Acta Horticulturae. 751: 217‒224.
Horst, R. K., and R. A. Cloyd. 2007. Compendium of Rose Diseases and Pests. 2nd ed. St. Paul, MN: APS Press. 32‒33.
Manners, M. M. 1985. "The rose mosaic heat therapy program at Florida Southern College", Proceedings of the Florida State Horticultural Society. 98: 344‒347.
Manners, M. M. 1988. "Letter to the editor." The Amer. Rose Magazine.
Manners, M. M. 1997. "Effects of rose mosaic disease on performance of hybrid tea roses in Florida", in Proceedings of the Florida State Horticultural Society: 118-121.
Secor, G. A., M. Kong, and G. Nyland. 1977. "Rose virus and virus-like diseases." California Agr. 31(3): 4‒7.
Thomas, B. J. 1982. "The effect of prunus necrotic ringspot virus on field-grown roses." Ann. Appl. Biol. 100: 129‒134.
Thomas, B. J. 1984. "Epidemiology of three viruses infecting the rose in the United Kingdom." Ann. Appl. Biol. 105: 213‒222.
USDA Census of Agriculture. 2015. "2012 Census of Agriculture: Census of Horticultural Specialties (2014), Volume 3." Accessed June 23, 2022. https://agcensus.library.cornell.edu/wp-content/uploads/2012-Census-of-Horticultural-Specialties-HORTIC.pdf
White, R. P. 1928. "An infectious chlorosis of roses." https://babel.hathitrust.org/cgi/pt?id=uc1.31175007201984;view=1up;seq=49