- Skunkvine is fast-growing and can adapt to a wide range of light, soil, and salt conditions.
- It invades disturbed areas and undisturbed native plant communities, including sandhill, rockland hammock, upland mixed forest, swamps, and floodplains.
- This weed creates dense canopies, leading to damage or death of native vegetation (Figure 1).
- It has smothered out one of the remaining populations of the endemic, federally endangered Cooley's water willow.
- Skunkvine spreads by transport of rooted fragments and seeds.
- This weed is also found in landscapes.
Native to eastern and southern Asia, skunkvine is an invasive plant species introduced to the USDA Field Station near Brooksville (Hernando County, FL) before 1897 (Morton 1976). By 1916, it was already reported as a troublesome weed in that area. It was known to escape to thickets and fencerows in peninsular Florida in 1933 (Small 1933). By 1977, it was considered an economically important weed (Reed 1977). In 1993, it was included on the Florida Exotic Pest Plant Council List of Invasive Species as a Category I, defined as "species that are invading and disrupting native plant communities in Florida." It was added to the Florida Noxious Weed List (5B-57.007 F.A.C.) in 1999, making it illegal to possess, move, or release in Florida.
Skunkvine is most common in west-central Florida, but it has also been documented northward into Georgia (Wunderlin et al. 2022). It has also been reported in North Carolina, Louisiana, Texas, South Carolina, and the Hawaiian Islands (https://www.eddmaps.org/distribution/) and in natural areas in over 25 counties throughout north, central, and south Florida (Langeland et al., 2008; Wunderlin et al., 2022).

Credit: UF/IFAS Center for Aquatic and Invasive Plants
How to Identify Skunkvine
- It has perennial twining vine from a woody rootstock.
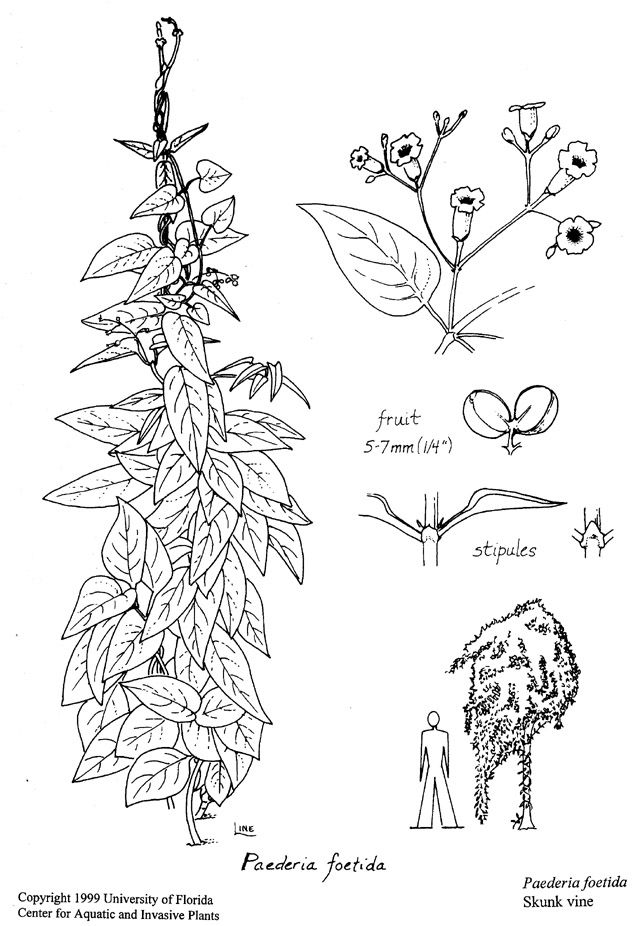
- Leaves are opposite (rarely in whorls of 3), oval to lance-shaped, and often lobed at the base. They grow 2–11 cm (1–4.3 in) long with conspicuous stipules (appendages at bases of leaves), and have leaf margins without teeth. The leaf surfaces can be hairy or non-hairy (Figure 2 and Figure 5).
- Leaf stalks commonly grow to 6 cm (2.4 in) long.
- Leaves and stems have a disagreeable odor, especially when crushed.
- Flowers (appearing mostly in late summer and fall) are small and grayish pink or lilac in color. The flowers form in broad or long, "leafy," curving clusters. Petals join to form a tube (corolla), with usually five spreading lobes (the corolla is dense and hairy) (Figure 3 and Figure 5).
- Fruit (persisting through winter) are shiny brown and nearly round, growing to 0.7 cm (0.3 in) wide. Each fruit has two seeds that are black, roundish, and often dotted with white, needle-shaped crystals (Figure 4 and Figure 5).
- Sewer vine (P. cruddasiana), a serious (but less widespread) pest, is similar to skunkvine but has oval, flattened fruits and distinctly winged seeds.

Credit: UF/IFAS Center for Aquatic and Invasive Plants

Credit: UF/IFAS Center for Aquatic and Invasive Plants

Credit: UF/IFAS Center for Aquatic and Invasive Plants
Credit: UF/IFAS Center for Aquatic and Invasive Plants
Control
Some hand removal of skunkvine in landscape situations will be necessary, but large-scale hand removal in natural areas has proven ineffective. Cut stems will die above, but regrow from below. Always be careful not to dispose of skunkvine where seeds can germinate or where stem fragments can take root. Flooding decreases vigor, but plants have survived submersion for at least 192 days. Herbicides that contain the active ingredients aminopyralid, glyphosate, imazapic, triclopyr amine, and triclopyr ester provide control (Table 1). Complete control may not be achieved with a single application; follow-up applications may be necessary. Avoid contact of herbicides with sensitive nontarget vegetation. Use herbicides according to all directions on the label.
References
Langeland, K. A., H. M. Cherry, C. M. McCormick, and K. A. Craddock Burks. 2008. Identification and Biology of Nonnative Plants in Natural Areas of Florida, 2nd edition. SP-257. Gainesville: University of Florida Institute of Food and Agricultural Sciences.
Morton, J. F. 1976. "Pestiferous Spread of Many Ornamental and Fruit Species in South Florida." Proc. Fla. State Hort. Soc. 89:348-53.
Reed, C. F. 1977. Economically Important Foreign Weeds: Potential Problems in the United States. Ag. Handbook No. 498. Washington, D.C.: APHIS, USDA.
Small, J. K. 1933. Manual of the Southeastern Flora, Parts one and two. Chapel Hill, NC: University of North Carolina Press. Facsimile reprint 1972, New York: Hafner Publishing.
Wunderlin, R. P., B. F. Hansen, A. R. Franck, and F. B. Essig. 2022. Atlas of Florida Plants (http://florida.plantatlas.usf.edu/).