Expanding production methods of marine fish is critical to the advancement of the food, bait, and ornamental aquaculture industries. Currently, only a limited number of marine fish species are being produced and with variable success. The major impediment to commercial production of currently grown species and success with candidate species is the utilization of an appropriate live feed during the first feeding phase of the larval cycle. This period is extremely crucial for the optimal development of marine fish larvae. A live feed with the proper nutritional composition, constituting a suitable size range, and stimulating a feeding response is necessary to expand the number of species of marine fish produced. In this Extension publication, we will discuss the pros and cons of using rotifers, brine shrimp, and copepods as live food for marine fish larvae.
Marine Fish Larviculture Requirements
Fatty Acid Nutrition
Marine fish larvae require live feeds that contain essential nutrients at appropriate concentrations. One group of essential nutrients are the fatty acids, organic acids found in animal and vegetable fats and oils. Fatty acids are mainly composed of long chains of hydrocarbons (molecules containing carbon and hydrogen) that end with a carboxyl group (comprised of carbon, two oxygen atoms, and hydrogen). Fatty acids are considered saturated when the bonds between carbon atoms are all single bonds and are unsaturated when some of these bonds are double bonds. Fatty acids have double bonds that start at carbon number 0, 3, 6, or 9. The process of increasing the number of carbons in a fatty acid is termed elongation; increasing the number of double bonds is termed desaturation. As an example, the fatty acid eicosapentaenoic acid (EPA, 20:5n-3) has 20 carbons and 5 double bonds, and the first double bond is on the third carbon atom. Elongation will increase the number of carbons to greater than 20 and desaturation will increase the number of double bonds to more than 5. Most organisms cannot efficiently change the location of the first double bond so n-3 fatty acids cannot be converted to n-6. (The "n-3 fatty acids" are also known as "ω-3" or "omega-3" fatty acids, and the "n-6 fatty acids" are also known as "ω-6" or "omega-6" fatty acids.) The n-3 highly unsaturated fatty acids (HUFAs) docosahexaenoic acid (DHA, 22:6n-3) and eicosapentaenoic acid (EPA, 20:5n-3) are essential for marine fish (Watanabe 1993). The ratio of DHA to EPA significantly affects the survival of marine fish larvae. The yolk of many wild marine fish eggs contain a DHA:EPA ratio of about 2.0, which suggests at least a 2:1 ratio of DHA:EPA in first-feeding larvae (Parrish et al. 1994).
The ability to synthesize EPA, and subsequently DHA, through elongation of linolenic acid (LNA, 18:3n-3) is absent in most tropical and subtropical marine fish. Therefore, they must rely on their diet to receive these essential nutrients. Marine fish contain large amounts of DHA and EPA in the phospholipids of their cellular membranes, specifically in the neural and visual membranes (Sargent et al. 1999). A lack of these essential fatty acids can result in retarded physiological development and altered behavior, such as impaired pigmentation and poor vision in low light intensities, resulting in increased vulnerability to predation and reduced hunting capability (Bell et al. 1995; Estevez et al. 1999; Sargent et al. 1999).
A similar situation exists for marine fish larvae and the n-6 fatty acids. Arachidonic acid (ARA, 20:4n-6) cannot be synthesized from linoleic acid (LA, 18:2n-6) by many marine fish species. ARA is a precursor to the eicosanoids, an important group of immunological compounds, which includes prostaglandins and leukotrienes. Without these compounds, the fish immune system is compromised. However, over-enrichment of ARA could have deleterious effects, so an optimal ratio for the species of interest should be maintained (Bessonart et al. 1999; Estevez et al. 1999).
Physical Characteristics of Prey
The size of live feed organisms and their ability to elicit a feeding response from fish larvae are important considerations in marine fish larviculture. The small mouth gape of many marine fish larvae limits the size of food it can consume and prevents the initial use of larger live food organisms such as brine shrimp. As fish larvae have evolved to feed on natural congregations of zooplankton, the stimuli produced by the movement of live feed organisms is needed for many marine fish larvae to elicit a feeding response. Larval mouth gape and feeding response to various live feeds are species specific; both should be established for the species to be cultured since they will determine which live food to use.
Live Feeds
Rotifers
Rotifers are small metazoans with over 2000 species described; most inhabit freshwater lakes and ponds (Lubzens and Zmora 2003). Two marine species, Brachionus plicatilis and B. rotundiformis have been used to culture over 60 species of marine fish larvae and 18 species of crustacean larvae (Dhert 1996). Rotifers produce dormant cysts following sexual reproduction. These cysts can be collected and purchased to start a new population or a portion of an existing live population can be used to initiate a new population.
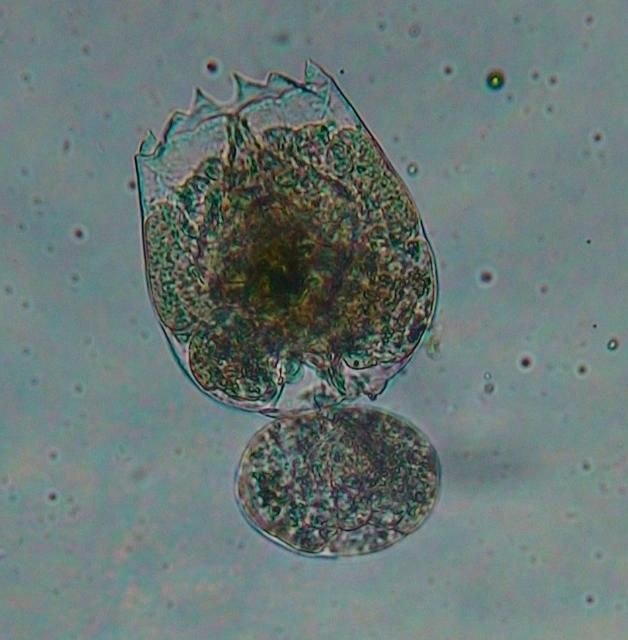
Credit: Cortney Ohs, UF/IFAS
Rotifers propagate quickly under suitable conditions, with populations doubling over a few days. Cultures can become quite dense and commonly exceed 1000 rotifers/mL. This is an advantage for fish hatcheries with a large demand for live feeds during the larval phase. On average, 20,000 to 100,000 rotifers will be fed to each fish larvae during a 20–30 day larval period (Lubzens and Zmora 2003). Rotifers are small, with a body width (BW) of 90–350 microns (1000 micron = 1 millimeter), but are nevertheless larger than the mouth gape size range of many first feeding marine fish species. Recent studies have examined the potential for culture of a much smaller marine rotifer, Proales similis, which has a BW of ~40 microns (Wuller et al. 2009). So, smaller rotifers may be commercially available in the future.
Rotifers are commercially available and can be cultured in sufficient numbers to satisfy the needs of a marine fish hatchery. Rotifers can be cultured with live algae or algal paste which may simplify the process and decrease cost of production. However, there are some disadvantages to using rotifers as a live feed. Rotifers do not have the proper nutritional profile required by marine fish larvae and lack DHA, EPA, and ARA. Rotifers lack the ability to elongate shorter chain fatty acids and, therefore, must be enriched to satisfy the HUFA requirement before they are fed to marine fish larvae (Sargent et al. 1997). Currently, commercially available enrichments are fed to rotifers, and they acquire and retain potentially adequate levels of HUFAs for several hours, provided they are kept at 10°C (50°F) to reduce their metabolic rate. Once rotifers are placed in a larval culture tank, metabolism resumes and the nutrients they acquired from the enrichments are metabolized or leach: as a result the nutrients are only available to the fish larvae for a short period of time. Furthermore, while rotifers can be enriched to increase their EPA, DHA, and ARA content, the delivery mechanism is not ideal. HUFAs delivered through phospholipids are more easily absorbed by fish larvae than those available as triacylglycerols (Sargent et al. 1999). However, most enrichments use triacylglycerols instead of phospholipids to deliver HUFAs because of the ease of encapsulation and reduction in leaching.
The quantity of rotifers within a larval system must be constantly monitored to ensure rotifers do not propagate beyond the grazing pressure of fish larvae. If this occurs, fish larvae will consume nutritionally inadequate rotifers and the water quality can quickly deteriorate, subsequently decreasing survival of the fish larvae. Further evidence suggests that rotifers are not easily digested (Schipp et al. 1999) and their steady, random motion, controlled by their ciliated crown, does not induce a feeding response in all marine fish larvae (Chesney 2005).
There are many pros and cons to feeding rotifers to marine fish larvae. Rotifers have been successfully used to culture many species of marine fish and are used for commercial production of several species, but their nutritional composition must be enriched for success, and their size and movement do not meet the needs of all species of marine fish. For further information on the culture of rotifers see Lubzens and Zmora (2003) and Dhert (1996).
Brine Shrimp
Brine shrimp (Artemia spp.) are the most widely used form of live feed to culture larvae in the world. Brine shrimp are wild harvested from natural hypersaline lakes as dormant cysts, which are easily collected, stored dry, and marketed. About 24 hours after cysts are introduced into 28°C saline water they hatch and nauplii can be collected. The optimal salinity for hatching is 15–35 g/L, but they can hatch from 1–80 g/L. The ability of cysts to be stored for long periods of time and relative predictability of hatching success make brine shrimp an attractive live feed for culture of many marine fish species.

Credit: Cortney Ohs, UF/IFAS
Newly hatched brine shrimp nauplii are about 450 microns in body width, which is usually too large for most first feeding marine fish larvae to consume. Therefore, they are typically fed after the rotifer feeding phase and up to the transition to an artificial diet; only a few larger marine fish larvae can be fed brine shrimp nauplii at first feeding.
Brine shrimp are a nutritionally deficient live feed for most developing marine fish larvae. Like rotifers, they have insufficient levels of DHA, EPA, and ARA for marine fish larvae. Nauplii of the instar I and II stages are the most common stages fed to larval fish and develop within 24 and 36 hours after hatching, respectively. The instar I, a non-feeding stage, cannot be enriched but all other life stages should be enriched before they are fed to marine fish larvae. Enrichment of instar II can temporarily improve their nutritional composition; however, the fatty acid concentrations attained can be inconsistent because brine shrimp do not uniformly consume the enrichments and some of what the brine shrimp ingest they will metabolize before they themselves are consumed. Additionally, brine shrimp have the disadvantage of catabolizing DHA back to EPA. Therefore, the ability to increase the DHA:EPA ratio by enriching brine shrimp may be limited.
The hatching and growth characteristics of brine shrimp can also impede the success of larval culture. Decapsulation and hatching of cysts, and molting of nauplii produce shells and exoskeletons which, if not removed from the culture system, can deteriorate water quality. Recent advancements in artificial larval diets and the variable harvest and supply of brine shrimp from hypersaline lakes may decrease the aquaculture industry's use of brine shrimp in the future. However, brine shrimp are still the most commonly used food for marine larvae and serve as the primary food organism between early larval stages and weaning to dry diets. For further information on the culture of brine shrimp see Lavens and Sorgeloos (1999).
Copepods
Copepods are tiny crustaceans, and are one of the most ubiquitous marine organisms with over 21,000 species currently described. Copepods are a major component of the marine zooplankton community (Smithsonian Institution 2008). It is well documented that, in the wild, copepods constitute a major link in the nutrient pathway from primary producers to marine fish larvae. Marine fish larvae eat copepod nauplii, and juvenile fish consume adult copepods. The role of copepods in the marine trophic system is essential to the survival of many marine fish species. Research investigating the efficacy of using copepods to culture various fish species is at the forefront of advancing marine aquaculture. Studies focused on feeding copepods to marine fish larvae have documented improvements in growth and survival of many species worldwide.
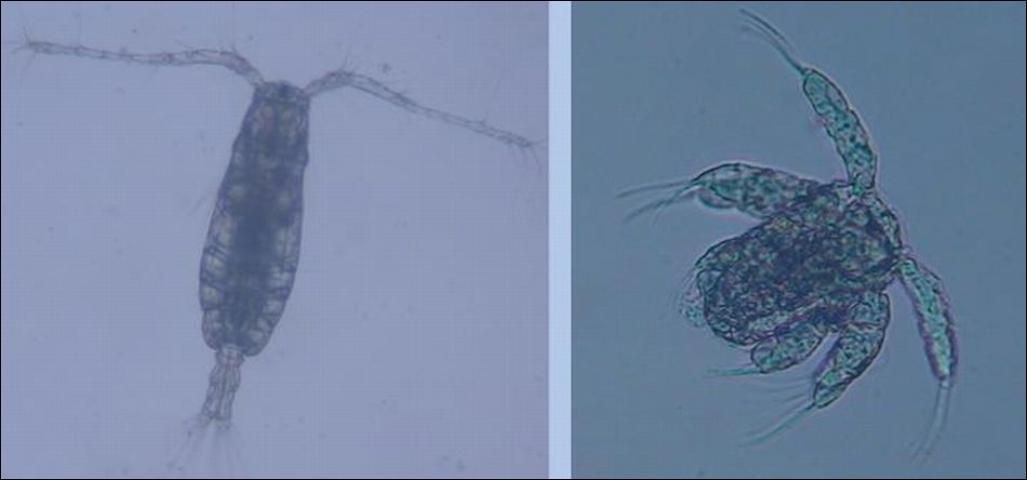
Credit: Cortney Ohs, UF/IFAS
Copepods are able to synthesize essential HUFAs, without enrichment, and maintain appropriate DHA:EPA and EPA:ARA ratios required by marine fish larvae. In addition, as much as 90% of the total fatty acids present in copepods are in the more easily used form of phospholipids. Therefore, unlike rotifers and brine shrimp, copepods do not need to be enriched and will not lose their nutritional value quickly because of leaching or excretion.
Fish larvae consume marine copepods from three main orders: Calanoida, Harpacticoida, and Cyclopoida. Copepods have life stages including nauplii, copepodites, and adults; each stage is progressively larger in size. Nauplii are normally fed to marine fish larvae. Several species of calanoid and harpacticoid copepod nauplii have been fed to fish larvae in aquaculture. The size range of nauplii varies among species and ranges from 38–220 microns in body width. Copepod locomotion is controlled by their swimming legs and is intermittent with periods of jerky forward motion and other periods of quiescence. This movement pattern allows fish larvae to identify copepods as prey and elicits a feeding response.
Although copepods are the preferred prey of wild marine fish larvae, their benefits to larviculture are not well documented and their use in commercial aquaculture has been slow. This is largely due to inconsistent production of substantial numbers of nauplii and species-specific culture methods which can vary greatly. The culture techniques involved with other live feeds, such as rotifers, are similar regardless of the species of rotifer.
Development of culture protocols for native copepods within your region should also be investigated to avoid the possible accidental escape of non-native species. Best management practices of an aquaculture facility should prevent direct discharge of culture water into local saline waters. Research must continue to be conducted to define appropriate culture methods for various species of copepods. In the future, copepods may allow for commercial production of many new species of marine fish larvae because they provide for survival and growth through the critical first feeding stage. Additionally, copepods may increase efficiency of the larval phase of species currently fed rotifers. However, there are some cons for use of copepods. Commercial sources of copepods are not common compared to rotifers and brine shrimp. Only a few species of calanoid copepods in the genus Acartia produce resting and/or subitaneous (long-term dormancy) eggs, which are being investigated for storage and marketing of eggs. Identification of species-specific culture methods is critical for commercial availability of copepods. Additionally, copepods have not been successfully cultured using any diet other than live algae. Prepared diets and algal paste have not been successful but warrant further investigation. Therefore, the culture of copepods requires more space, equipment, and time to culture the live algae, which is not required for culturing rotifers or hatching brine shrimp. For further information on the culture of copepods see Lee et al. (2005).
Conclusion
The decision whether to use rotifers, brine shrimp, and/or copepods should be based on the species of fish larvae being produced and the best way to deliver nutrients to accommodate the feeding capabilities of the fish species. Rotifers and brine shrimp have been successfully used to culture many species of marine fish through the larval phase. Well documented culture protocols, improvements in nutritional enrichments, and readily available cysts make these live feeds appealing. Species of copepods have been investigated and appropriate culture protocols are being developed. Currently, there are many research projects occurring worldwide evaluating the culture methods for copepods and feeding them to new species of marine fish. Determining economic costs and benefits of live food organisms should include any improvements in survival, growth rate, and stress resistance gained by the marine fish being cultured to fully account for the benefits.
Further Reading
Lee, C. S., P. J. O'Bryen, N. H. Marcus, 2005. Copepods in Aquaculture. Blackwell Publishing, Ames, Iowa. 269 pp.
Lavens, P., P. Sorgeloos, 1999. Manual on the production and use of live food for aquaculture. FAO Fisheries Technical Paper No. 361. FAO, Rome, Italy. 305 pp.
Stottrup, J.G., L. A. McEvoy, 2003. Live Feeds in Marine Aquaculture. Blackwell Scientific Publications Ltd, Oxford, United Kingdom. 318 pp.
References
Bell, J. G., J. D. Castell, D. R. Tocher, F. M. Macdonald, and J. R. Sargent. 1995. Effects of different dietary arachidonic acid:docosahexaenoic acid ratios on phospholipid fatty acid compositions and prostaglandin production in juvenile turbot (Scophthalmus maximus). Fish Physiology and Biochemistry 14: 139–151.
Bell, J. G., L. A. McEvoy, A. Estevez, R. J. Shields, and J. R. Sargent. 2003. Optimising lipid nutrition in first-feeding flatfish larvae. Aquaculture 227: 211–220.
Bell, M. V., R. J. Henderson, and J. R. Sargent. 1985. Changes in the fatty acid composition of phospholipids from turbot (Scophthalmus maximus) in relation to dietary poly-unsaturated fatty-acid deficiencies. Comparative Biochemistry and Physiology B-Biochemistry & Molecular Biology 81: 193–198.
Bessonart, M., M. S. Izquierdo, M. Salhi, C. M. Hernandez-Cruz, M. M. Gonzalez, and H. Fernandez-Palacios. 1999. Effect of dietary arachidonic acid levels on growth and survival of gilthead sea bream (Sparus aurata L.) larvae. Aquaculture 179: 265–275.
Chesney, E.J., 2005. Copepods as live prey: A review of factors that influence the feeding success of marine fish larvae. In: Lee, C.S., P. J. O'Bryen, N. H. Marcus (Eds.), Copepods in Aquaculture. Blackwell Publishing, Ames, pp. 133–150.
Dhert, P., 1996. Rotifers. In: Lavens, P., P. Sorgeloos (Eds.), Manual on the Production and Use of Live Food for Aquaculture. FAO Fisheries Technical Paper No. 361. Rome, FAO. pp. 49–78.
Estevez, A., L. A. McEvoy, J. G. Bell, and J. R. Sargent. 1999. Growth, survival, lipid composition and pigmentation of turbot (Scophthalmus maximus) larvae fed live-prey enriched in arachidonic and eicosapentaenoic acids. Aquaculture 180: 321–343.
Lubzens, E., O. Zmora, 2003. Production and nutritional value of rotifers. In: Stottrup, J. G., L. A. McEvoy (Eds.), Live Feeds in Marine Aquaculture. Blackwell Scientific Publications Ltd, Oxford, pp. 17–64.
Parrish, C. C., J. D. Castell, J. A. Brown, L. Boston, J. S. Strickland, and D. C. Somerton. 1994. Fatty acid composition of Atlantic halibut eggs in relation to fertilization. Bulletin of the Aquaculture Association of Canada 94: 36–38.
Sargent, J., G. Bell, L. Mcevoy, D. Tocher, and A. Estevez. 1999. Recent developments in the essential fatty acid nutrition of fish. Aquaculture 177: 191–199.
Sargent, J., L. McEvoy, J. G. Bell, 1997. Requirements, presentation and sources of polyunsaturated fatty acids in marine fish larval feeds. Aquaculture 155: 117–127.
Schipp, G. R., J. M. P. Bosmans, and A. J. Marshall. A method for hatchery culture of tropical calanoid copepods, Acartia spp. Aquaculture 174: 81–88.
Smithsonian Institution 2008. The World of Copepods. T. Chad Walter, database manager. National Museum of Natural History, Department of Invertebrate Zoology. Last accessed: May 2009. Available: http://invertebrates.si.edu/copepod/
Watanabe, T. 1993. Importance of docosahexaenoic acid in marine larval fish. Journal of World Aquaculture Society 24: 152–161.
Wuller, S., Y. Sakakura, and A. Hagiwara. 2009. The minute monogonont rotifer Proales similis de Beauchamp: Culture and feeding to small mouth marine fish larvae. Aquaculture 293: 62–67.