Introduction
Understanding how non-native species escape or are accidentally released helps producers better design and operate aquaculture facilities to reduce or prevent escape. Active management of critical points where escape is possible will help achieve regulatory compliance. This document is the second in a four-part series devoted to educating industry and other stakeholders on the importance of preventing the escape of non-native species from aquaculture facilities as well as strategies for non-native species containment and regulatory compliance (Tuckett et al. 2016a; Tuckett et al. 2016b; Tuckett et al. 2016c).
Series Contents
- Part 1: General Considerations and Regulations—introduces series, explains why non-native species containment is important, provides information on regulations, including the Florida Aquaculture Best Management Practices (BMPs) rule, describes the BMP inspection process, and provides advice on achieving compliance
- Part 2: Facility Evaluation Strategies—describes farm layouts, how fish escape, and a process that aquaculturists can complete to identify potential escape points on their farms
- Part 3: Structural Strategies—provides information on structures and barriers that can prevent escape
- Part 4: Operational Strategies—describes operational and management strategies to prevent escape
Farm Layout
Farm layout plays an important role in reducing the escape of non-native species. Many farms have a combination of culture and holding systems located inside buildings or greenhouses and production ponds outdoors (Figure 1). Production ponds may or may not be covered with bird netting or plastic covers (winter). Most farms have a water detention system consisting of interior and perimeter ditches and, in some cases, ponds or constructed wetlands. Ditches receive water pumped from ponds or flowing from buildings. Water in these ditches may percolate into the soil or may flow into detention/retention ponds or offsite. Culverts or control structures, often with riser boards and screens, typically separate ditches into segments or occur at farm road crossings, ditch/pond connections, and points of effluent discharge. Culverts and control structures are discussed and shown in Part 3 of this series (Tuckett et al. 2016b).
Water flow through ditches may be fairly constant, often associated with flow-through discharge from indoor systems. Flow in other ditches may be highly intermittent and related to periodic pond dewatering or heavy rainfall. Numerous farms have a low base flow that is periodically increased during pond pumping cycles or seasonal rainy periods. Management of escape is important both for lower base flows and during periodic high-flow events.
Detention ponds and retention ponds, particularly those stocked with native predatory fishes such as largemouth bass Micropterus salmoides, bluegill Lepomis macrochirus, and eastern mosquitofish Gambusia holbrooki, can be very effective at preventing the escape of non-natives and helping producers to comply with Florida Aquaculture Best Management Practices (BMPs; FDACS 2015). Research at the UF/IFAS Tropical Aquaculture Laboratory (TAL) showed that farms with detention ponds stocked with native predatory fishes were less than one sixth as likely to have a BMP violation as farms without (Tuckett et al. 2016d). In contrast, multiple points where effluent leaves farms make management of non-native species escape more difficult.
Several other features can reduce the escape or loss of non-natives from aquaculture facilities. Berms around ponds or around the facility reduce the chance that heavy rain will lead to release due to flooding. Possibly most important is the location of the facility away from offsite streams and wetlands, but security fences and locked gates, security lighting, guard dogs, and on-site staff are helpful as well, reducing the potential for theft and unauthorized releases by human or animal trespassers.
Some facilities vary from the layout shown in Figure 1. For example, indoor, low- or zero-discharge facilities are becoming more common. Non-native species are unlikely to escape from facilities housing all production indoors and producing no offsite discharge. Our research showed no loss of non-native fish at four such facilities in west-central Florida (Tuckett et al. 2014).
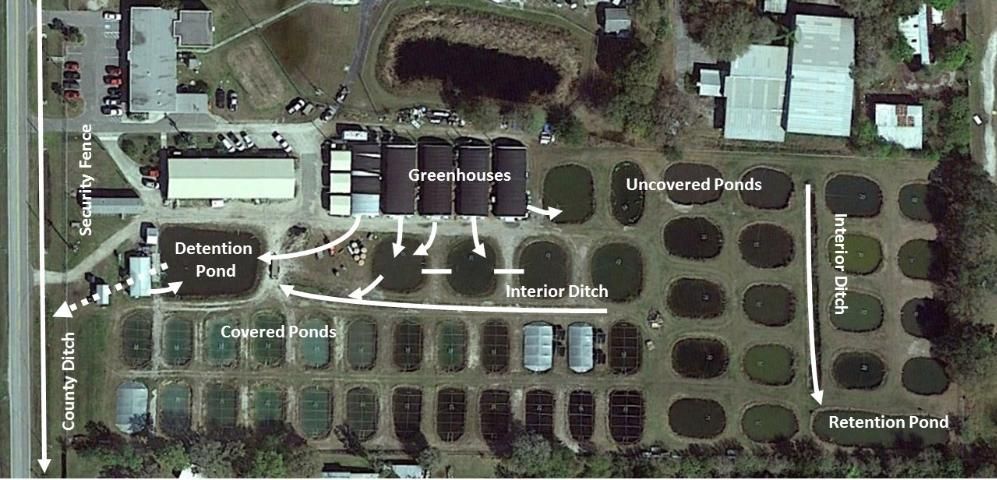
Credit: Reprinted from Hill et al. (2016)
How Fish Escape
Fish and other cultured organisms generally escape from aquaculture facilities through effluent discharged offsite to surface waters. Fish are also thought to escape because of vandalism, losses during fish transfer and transportation, and through carry-off by birds or other animals. However, our research shows that these pathways are unimportant for most aquaculture producers in Florida (Tuckett et al. 2014). Potentially, fish can escape during several life stages, including as fertilized eggs, fry, juveniles, and adults. In practice, free-swimming stages (juveniles and adults) are far more likely to escape.
Effluent is by far the dominant pathway for non-native fish to escape from aquaculture farms in Florida (Tuckett et al. 2014). Fish may exit down drainpipes or jump from tanks, ending up in building trench drains and making their way to the farm's detention system (ditches or ponds). Fish may pass through riser board structures, over standpipes, and through culverts to move through the detention system and eventually offsite. This movement can occur during normal operations where there is a base flow of water or during periodic high-flow periods when ponds are pumped or when rainfall is heavy. Heavy rainfall can also cause ponds and ditches to flood, allowing fish to move to other production ponds or into the detention system. An overtaxed detention system can then discharge fish offsite. Flooding can connect ponds or ditches to offsite streams or wetlands if rainfall is extremely heavy and berms/ditches are not properly maintained.
Fish escape from ornamental aquaculture facilities is low relative to the very large number of fish present on each farm. Further, the diversity of fish released is low compared to the variety of fish on the farms and in the ornamental fish trade (Hill and Yanong 2010). However, compliance with Florida Aquaculture BMPs requires producers to address fish escape. Escape and survival (in near-farm ditches, canals, and streams) of non-native organisms will also differ among the various types of organisms; our research at 23 ornamental aquaculture facilities showed that cichlids and livebearers are the two groups most likely to be captured near the effluent (Figure 2) (Tuckett et al. 2014). Figure 2 is an approximation of fish escape, which will vary within the broad groups shown.

Credit: UF/IFAS Extension
Different structures vary as well, especially in their ability to contain various species of fish. Livebearers, for example, escape from unscreened tank standpipes and through ditch structures with surface flow (e.g., standpipes or riser boards; Figure 3). This may be due to the fact that many livebearers are surface-oriented and will follow water flow. Research into the effectiveness of different types of barriers on different fish groups would help producers choose appropriate barriers for their facilities.

Credit: UF/IFAS Extension
Other mechanisms of escape or release were investigated for Florida aquaculture producers. Vandalism has been shown to be an issue in cage culture operations in public waters in other parts of the world (e.g., salmon production in coastal waters or tilapia and carp production in lakes). Of 23 ornamental fish producers, none had experienced vandalism or theft that resulted in the release of cultured fishes. Such an occurrence is unlikely for most types of cultured products and most farm configurations in Florida. Escape during transport or contamination by hitchhiking species is a concern for some aquaculture and live aquatic species industries. However, our research shows that loss during transport is highly unlikely for ornamental fishes produced on Florida farms (Zajicek et al. 2009, Tuckett et al. 2014). Escape for other Florida aquaculture finfish and shellfish commodities also should be low during transport (Zajicek et al. 2009). Movement of non-native fish by waterfowl or wading birds is a common concern but has had little documentation. Our research showed that bird carry-off is extremely unlikely, even for aquaculture farms with numerous open ponds visited by fish-eating birds (Tuckett et al. 2014). Generally, fish-eating birds move fish only short distances and the fish that escape from birds are heavily damaged.
Analysis of Escape on the Farm—Critical Points
Producers are responsible for prevention of the escape of non-native species under the Florida Aquaculture BMPs. To comply, producers can analyze their facilities and operational procedures to determine how non-natives might escape. There are several ways that a producer might go about such an analysis, but a good framework is known as Hazard Analysis Critical Control Point (HACCP; pronounced "has-sip"). HACCP was developed in the food safety industry to minimize the health risks associated with food production and processing. Through this process, the hazards are identified and critical control points are established to effectively manage the hazards.
HACCP is now commonly used to minimize the potential to introduce or spread non-native aquatic species. Federal fish hatcheries nationally and many commercial baitfish and aquaculture operations in the upper Midwest develop and implement HACCP plans (e.g., USFWS 2015). While formal HACCP plan development, adoption, and implementation are not necessary for Florida producers to effectively limit non-native species escape, understanding basic HACCP principles will assist producers in effective management of escape. HACCP is a framework, a way of thinking about limiting specific hazards, and it is adaptable to a producer's needs. In essence, a producer can do the analysis as formally or informally as wanted. Our research on BMP effectiveness and non-native fish escape used the HACCP framework in an informal way to help determine likely locations and procedures where escape was a potential occurrence (Tuckett et al. 2014). Information and training on HACCP as well as specific HACCP plans can be found online at the US Fish and Wildlife Service HACCP website (USFWS 2015) and from Minnesota Sea Grant (Gunderson and Kinnunen 2004).
The seven HACCP steps or principles include:
- Conduct a hazard analysis. Prepare a list of steps in the process where significant hazards occur and describe the control measures.
- Identify the critical control points (CCP) in the process.
- Establish controls for each CCP identified.
- Establish CCP monitoring requirements. Establish procedures for using monitoring results to adjust the process and maintain control.
- Establish corrective actions to be taken when monitoring indicates that there is a deviation from an established critical limit.
- Establish procedures to verify that the HACCP system is working correctly.
- Establish effective record-keeping procedures that document the HACCP system.
Principle 1: Hazard Analysis
Hazards in this analysis would be the escape of non-native species from the aquaculture facility. The farm layout and operational procedures determine where fish are likely to escape. Our research showed that through the effluent is the main pathway, but producers should consider vandalism/theft, transport, and bird carry-off if applicable to their facilities. Once locations and procedures are identified, control measures can be determined. For the facility in Figure 1, control points related to water flow occur between:
- tank systems (recirculating or flow-through) inside each greenhouse or building and any trench/floor drains,
- the trench/floor drains and outlet pipes from the greenhouses and buildings,
- the outlet pipes and the receiving ponds,
- the receiving ponds and the interior ditch,
- the interior ditch and the detention pond,
- and the detention pond and the county ditch.
Each of these locations can be fitted with physical barriers such as screens to prevent movement of fish through the control points (Tuckett et al. 2016b). Operational procedures that can be identified as control points might include:
- siphoning of tanks into trench/floor drains,
- pumping of ponds into ditches,
- and movement of seines, traps, waders, or other items from pond to pond.
Procedures can be put in place to ensure that these activities do not increase the chance that non-native species may escape (Tuckett et al. 2016c). For example, screening can be placed on pump intakes coupled with appropriate placement of the outlet hose in the detention system (e.g., at least 100 feet up the ditch from the detention pond) during pond pumping.
Principle 2: Determine the Critical Control Points
Although many control points and associated control measures may be identified during hazard analysis, relatively few control points may be critical control points (CCPs), that is, points where the hazard can be effectively controlled. For our example facility (Figure 1), three CCPs might be identified as the control points between (1) the buildings/greenhouses and the outdoor detention system of ponds, ditches, and the detention pond, (2) the interior ditch system and the detention pond (perhaps also retention pond), and (3) the detention pond and the county ditch (Figure 3). Considering these as CCPs would ensure redundancy in barriers and make it more difficult for non-native species to escape the facility. Since this facility has only a single point for the release of effluents, the control point between the detention pond and county ditch could be designated as the sole CCP and managed accordingly. In practice, many farms will have a more mixed approach where some internal control points are managed as critical points but the main emphasis will be on the effluent points where water leaves the farm.
Principle 3: Establish Controls
Effective management controls can be determined once the CCPs are located and defined. Control measures are chosen to effectively address potential escape but also to be cost-effective (i.e., not too costly to install or maintain). For example, a detention pond is highly effective but may be too expensive and difficult to install for an existing facility. For the example facility (Figure 1) where three CCPs were identified, the use of screens on tanks and building/greenhouse drains, a control structure separating the interior ditch from the detention pond and the detention pond from the county ditch (especially with screens), and predatory fish stocked into the detention pond (Figure 1) would be effective control measures (Figure 3).
Principle 4: Critical Control Point Monitoring
Monitoring the CCPs will help ensure that the control measures used to prevent escape are effective and working as intended. Monitoring should include more than the normal BMP compliance inspections done by FDACS staff and include more frequent inspections by facility staff. Conditions may change and effectiveness of control measures may decline over time. Monitoring frequency is an important consideration, and any monitoring schedule should cover both normal operation and periodic events that may change the potential for species to escape (e.g., during heavy pumping or rainy periods). Adjustments can be made in response to deficiencies identified during monitoring. For example, over time the population of predatory fishes in a detention pond can decline and allow some non-natives to survive long enough to escape from the pond. Excessive growth of aquatic or marginal vegetation also might reduce the efficiency of predatory fishes. Monitor periodically to identify these issues and correct them before non-native species escape.
Principle 5: Corrective Actions
If CCPs become compromised then corrective actions must be taken. This can be as minor as replacing a missing screen on a tank standpipe or as major as repairing a breech in a berm. Quick follow-up on noted issues will reduce the chance that non-natives will escape. Following correction, do an evaluation of the reasons for the CCP failure, and then take action to anticipate and remediate future problems. For example, experience might determine that pumping five ponds into a specific ditch in one day will overwhelm the capacity of the ditch and cause flooding. A corrective action would be to pump no more than four ponds per day into the ditch.
Principle 6: Verification Procedures
Verification is used where there is a formal requirement for HACCP plans due to regulation or policy. Verifiers would be an industry group, agricultural or natural resource agency, or other third-party organization. No official HACCP program exists for Florida aquaculture producers, so this step is not applicable.
Principle 7: Record-Keeping Procedures
Records of defining the CCPs, control methods, monitoring, and corrective procedures can help producers address long-term issues with their HACCP plans and help suggest ways to strengthen management of non-native species escape. Keeping records regarding HACCP is not a requirement of the Florida Aquaculture BMPs.
Implementation
Producers are encouraged to use HACCP as a framework to help implement management of escape of non-natives. HACCP is flexible, and portions can be used at the producer's discretion. Full-blown HACCP plans containing all seven principles may be more than is necessary or wanted, but HACCP contains many elements that are useful and can be adapted to more modest planning. Extension faculty and staff at the UF/IFAS Tropical Aquaculture Laboratory in Ruskin can assist producers in design and implementation of HACCP or HACCP-like plans.
Acknowledgements
First and foremost, we thank our aquaculture facility cooperators and the Florida Tropical Fish Farms Association. This research would not have been possible without their generous support. We are indebted to staff at the Florida Department of Agriculture and Consumer Services Division of Aquaculture, including Joe Clayton, Kal Knickerbocker, Serina Rocco, Portia Sapp and Paul Zajicek. Funding was provided by the UF/IFAS School of Forest Resources and Conservation, the UF/IFAS Tropical Aquaculture Laboratory (Craig Watson, director), and a grant from the Florida Department of Agriculture and Consumer Services' Division of Aquaculture.
References
Gunderson, J., and R. Kinnunen. 2004. Aquatic nuisance species: hazard analysis and critical control point training curriculum, 2nd ed. Minnesota Sea Grant Publication Number MN SG-F11.
Hill, J. E., and R. P. E. Yanong. 2010. Freshwater ornamental fish commonly cultured in Florida. CIR-54. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/fa054.
Tuckett, Q. M., C. V. Martinez, J. L. Ritch, K. M. Lawson, and J. E. Hill. 2016a. Preventing escape of non-native species from aquaculture facilities in Florida, part 1: general considerations and regulations. FA195. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/fa195.
Tuckett, Q. M., C. V. Martinez, J. L. Ritch, K. M. Lawson, and J. E. Hill. 2016b. Preventing escape of non-native species from aquaculture facilities in Florida, part 3: structural strategies. FA197. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/fa197.
Tuckett, Q. M., C. V. Martinez, J. L. Ritch, K. M. Lawson, and J. E. Hill. 2016c. Preventing escape of non-native species from aquaculture facilities in Florida, part 4: operational strategies. FA198. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/fa198.
Tuckett, Q. M., J. L. Ritch, K. M. Lawson, and J. E. Hill. 2014. Best management practices and escapement of non-native fish from aquaculture facilities in Florida. Final Report. Florida Department of Agriculture and Consumer Services. Tallahassee, Florida.
Tuckett, Q. M., J. L. Ritch, K. M. Lawson, and J. E. Hill. 2016d. "Implementation and Enforcement of Best Management Practices for Florida Ornamental Aquaculture with an Emphasis on Nonnative Species." North American Journal of Aquaculture 78:113–124.
USFWS. 2015. "Hazard analysis and critical control point (HACCP)." http://www.fws.gov/fisheries/ans/ans-haccp.html
Zajicek, P. W., J. E. Hill, N. Stone, H. Thomforde, C. Ohs, D. Cooper, G. Flimlin, B. Mclane, and W. D. Anderson. 2009. Preventing Hitchhiking Nonindigenous Species in Live Shipments. Southern Regional Aquaculture Center, Stoneville MS, Publication No. 3902.