Dog Flea Ctenocephalides canis (Curtis) (Insecta: Siphonaptera: Pulicidae)
Introduction
Over 2,500 species and subspecies of fleas (Order: Siphonaptera) exist worldwide (Durden and Hinkle 2009). Approximately 94% of all flea species are reported to feed on mammals. These insects are known to vector the pathogens causing plague, murine typhus and feline leukemia. Most of the medically important fleas, including those in the genus Ctenocephalides, are found within the family Pulicidae.
The dog flea, Ctenocephalides canis (Curtis), is an important ectoparasite of both wild and domestic canids around the world (Durden et al. 2005). It is similar in appearance to the ubiquitous cat flea, Ctenocephalides felis (Bouché), but is encountered less frequently and thus has not been studied as thoroughly. The dog flea has been reported on many mammalian hosts, including dogs, cats, rabbits, rats, gray foxes, red foxes, woodchucks, and humans (Fox 1940). Ctenocephalides canis can act as intermediate hosts for parasitic worms, including the double-pored tapeworm, Dipylidium caninum, and the nematode, Acanthocheilonema reconditum (Durden and Hinkle 2009).

Credit: Krista Seraydar, University of Florida
Synonymy
Pulex canis Curtis, 1826
Pulex serraticeps Baker, 1895
Pulex serraticeps Osborn, 1896
Pulex canis Rothschild, 1901
Ctenocephalus canis Baker, 1904
Pulex canis Rothschild, 1905
Ctenocephalus canis C. Fox, 1910
Ctenocephalus canis C. Fox, 1925
Ctenocephalus canis Stewart, 1928
Ctenocephalus canis Ewing, 1929
Ctenocephalus canis Jordan, 1929
Ctenocephalides canis Stiles and Collins, 1930
Ctenocephalides canis Stewart, 1933
Ctenocephalides canis Shaftesbury, 1934
(summarized in Fox 1940)
Distribution
Members of the genus Ctenocephalides are thought to have originated in Africa, but have been introduced to countries around the world. Ctenocephalides canis occurs worldwide and is the predominant flea found on dogs in Ireland (Wall et al. 1997), Greece (Koutinas et al. 1995), Albania (Xhaxhui et al. 2009), Hungary (Farkas et al. 2009), Korea (Chee et al. 2008), Argentina (González et al. 2004), and rural areas of Chile (Alcaíno et al. 2002).
Although comprehensive surveys have not been conducted in the United States for over half a century, Ctenocephalides canis had been reported from 31 states and the District of Columbia (Ewing and Fox 1943). While Ctenocephalides canis may not be the predominant flea species found on domestic dogs in the United States, it is still an important ectoparasite of both wild and domestic animals. In a study performed by Durden et al. (2005), over 21% of the fleas recovered from dogs in the state of Georgia in the United States were Ctenocephalides canis. These fleas also are found relatively frequently on wild canids such as wolves, foxes, and coyotes throughout the United States, but especially in northern areas (Durden et al. 2005).
Description
Fleas are holometabolous and thus progress through the following life stages: egg, larva (three larval instars), pupa, and adult. The life cycle of Ctenocephalides canis can be completed in two to four weeks, which can lead to multiple generations in a given year (Ewing and Fox 1943). Adults occur throughout the year on host animals, with population numbers peaking in late summer or early autumn (Durden et al. 2005).
Eggs
A blood meal is required for a mated adult female flea to produce and lay eggs (Durden and Hinkle 2009). A single female flea can produce hundreds of eggs in her lifetime (Bayer Environmental Science 2007). Flea eggs range from 0.1 to 0.5 mm in length, are pearl-white in color, oval shaped, smooth, and non-sticky. Females lay eggs continuously while on the host, and eggs fall from the host, landing on the host's sleeping or resting area. According to a study performed by Baker and Elharam (1992), egg hatch was most successful at 25°C and 75% relative humidity. Under these conditions, egg hatch occurred within three to four days; however, eggs can take up to a week to hatch.
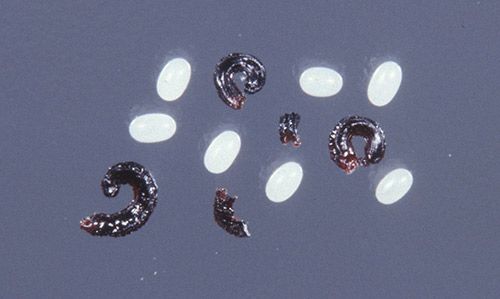
Credit: James Castner, University of Florida
Larvae
Ctenocephalides canis larvae are white, elongate, contain numerous setae, and lack legs and eyes (Durden and Hinkle 2009). Chewing mandibles and silk glands are housed within a well-developed head capsule. The larvae use these mandibles to ingest blood-rich feces, often referred to as "flea dirt," deposited by adult female fleas as they feed. At 25°C and 75% relative humidity, it takes 10 days for larvae to progress through three larval instars and develop into pupae (Baker and Elharam 1992); however, the larval period usually lasts two to three weeks. The first instar larva is about 1.5 mm in length, while the third instar is about 5 mm long (Bayer Environmental Science 2007). This last instar larva will use its silk glands to spin a cocoon that serves to protect the immobile pupal stage (Baker and Elharam 1992).
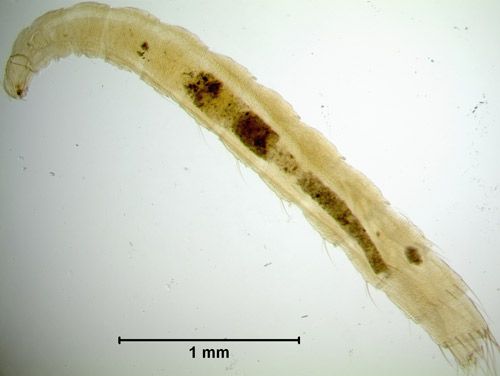
Credit: Krista Seraydar, University of Florida
Pupae
The larval silk used to build the cocoon is sticky and adheres to any debris in the environment; as a result, cocoons are camouflaged. The debris may help protect the developing pupa from predators such as ants (Silverman and Appel 1984). Inside the cocoon, the larva becomes immobile and molts to the pupal stage after several days. Pupae are off-white in color, eventually becoming dark brown as they mature. Adults will emerge from pupae after seven days at 25°C and 75% relative humidity (Baker and Elharam 1992). At lower-than-ideal temperatures and humidity levels, the pupal period lasts one to two weeks (Hill and MacDonald 2007). Adult fleas can remain inactive inside the cocoon as pre-emerged adults for extended periods of time, which enhances their survival during periods when no host animals are available. The closely related cat flea, Ctenocephalides felis, has been shown to survive inside cocoons for up to five months and become active upon detection of potential host vibrations (Durden and Hinkle 2009).

Credit: James Castner, University of Florida
Adults
Adult dog fleas are small (2.0–3.25 mm), wingless, bilaterally compressed, and heavily chitinized (Bayer Environmental Science 2007, Durden and Hinkle 2009). Members of the genus Ctenocephalides have genal and pronotal combs, large black eyes, and 5-segmented labial palps (Ewing and Fox 1943).
Although the dog and cat flea are very similar in appearance, the comb on the ventral margin of the head, the genal ctenidium, is used to help distinguish between Ctenocephalides canis and the cat flea, Ctenocephalides felis. The size of the first two genal spines also differentiates the two species. The first (or outer) genal spine of Ctenocephalides canis is much shorter than the second. In Ctenocephalides felis, the first genal spine is as long as, or barely shorter than, the second (Fox 1940).
Ctenocephalides canis have a head that is comparatively more rounded and about one and a half times as long as it is wide, while the head of Ctenocephalides felis is about twice as long as it is wide (Fox 1940). The cat flea has four to five "teeth" on the tibia of all six legs, whereas the dog flea has seven to eight teeth on the tibia of all six legs (Kramer and Mencke 2001).

Credit: Krista Seraydar, University of Florida

Credit: Krista Seraydar, University of Florida

Credit: Lyle Buss, University of Florida
Hosts
Once a suitable host is secured, adult dog fleas usually remain with that host animal for the remainder of the flea's life. Many animal species have been reported to successfully sustain dog fleas ,including coyotes, wolves, dogs, woodchucks, cats, rabbits, rats, gray foxes, and red foxes (Fox 1940, Durden and Hinkle 2009). Ctenocephalides canis is more likely to infest dogs that reside outdoors, in kennels, or in rural areas (Boushira et al. 2011) than in indoor or more urban areas.
Medical and Economic Significance
Ctenocephalides canis can serve as an intermediate host for the double-pored or dog tapeworm (Dipylidium caninum), rodent tapeworm (Hymenolepis diminuta), and dwarf tapeworm (Hymenolepis nana) (Durden and Hinkle 2009). Larval fleas ingest tapeworm eggs in the environment, which hatch and develop inside the flea gut. Tapeworms continue to inhabit the insect as the flea progresses to the adult stage. An animal or human can obtain a tapeworm infection upon ingestion of a flea containing a tapeworm cyst. Accidental ingestion usually occurs when animals groom themselves or when children play with pets (Durden and Hinkle 2009).
Symptoms of an infection with Dipylidium caninum in humans, a condition called dipylidiasis, include irritability, restlessness, weight loss, insomnia, and abdominal pain (Hamrick et al. 1983). A person or animal infected with a tapeworm will pass segments of the organism in the stool, which is an important indication of infection. The dog flea is also a principal host of the nematode, Acanthocheilonema reconditum. If bitten by a flea infected with Acanthocheilonema reconditum, a dog may develop a relatively benign condition called canine filariasis, the infestation of tissues with filarial nematodes.
In addition to obtaining parasites from fleas, host animals can develop allergic reactions to the saliva of the adult flea and develop a condition called flea-bite dermatitis (Durden and Hinkle 2009). In hypersensitive dogs and cats, this condition typically presents itself as hairless patches of purple papules or swellings, concentrated on the hindquarters of the animal (Werner 2008). Flea-bite dermatitis usually is treated with a corticosteroid injection (Durden and Hinkle 2009). Domestic dogs and cats often overcome hypersensitivity to the flea's saliva after repeated infestations.
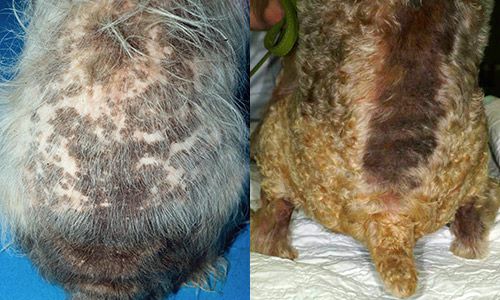
Credit: Rosanna Marsella, University of Florida
To learn more about flea-bite dermatitis, please visit the website of Dr. Alexander Werner, DVM Animal Dermatology Center.
Management
Prior to taking pest treatment steps, it is important to be sure that you have a flea infestation. Dogs, like many animals, itch for a variety of reasons, only one of which is the result of a flea infestation. Fleas on your pet or flea bites around one's ankles are both hallmark indications of flea presence in your home (Richman and Koehler 1997). Also, look for flea "dirt," which may appear as black specks on your carpet or pet's bedding. You can monitor the severity of a flea infestation by combing your pet or wearing white socks around the house.
A successful flea management program in and around the home should consist of a coordinated approach that includes both the host and environment. Dogs and cats can be bathed to remove flea dirt, and then combed thoroughly and repeatedly with a metal flea-comb to remove adults. An EPA-approved topical flea treatment available in stores or from a veterinarian can be applied to control any adults missed during combing or new infestations. Prior to using any insecticide on your pet, you should consult with your veterinarian regarding your dog's health status and breed sensitivities.
Sanitation is critical in areas where the animal sleeps or rests. Indoors, carpet should be vacuumed and bedding should be washed. Outdoors, dog houses, bedding, and other materials should be washed and placed in the sun. If desired or if the adult flea infestation is significantly large, consult with a pest control professional for additional insecticide treatments. Once a flea infestation has become established, management efforts both on the host and in the environment must be made simultaneously.
Prevention
Prior to taking your pet to another residence, veterinary clinic, dog kennel, or other place frequented by other dogs, have a plan in place. Consider pre-treatment of your pet, or provide one of the fast-acting insecticides after picking up your pet but before returning home. Many of these products are capable of killing adult fleas within 30 minutes. These efforts must be preplanned and your veterinarian should be consulted prior to the administration of any insecticide to your pet.
Mechanical Control
The most important form of mechanical control of the dog flea involves maintaining a hygienic environment. Mechanical control is as simple as vacuuming or steam cleaning your pet's bedding and/or carpet on a regular basis (Durden and Hinkle 2009). Bathing your pet regularly can remove some of the fleas and debris that would otherwise cause irritation. Flea combs also remove adult fleas and debris from pets. In case of an infestation, contaminated bedding, nesting material, or clothing should be either disposed of or exhaustively cleaned (Bayer Environmental Science 2007).
Chemical Control
Chemical control can be employed through direct use on pets or on areas infested with fleas. Pulicidal (flea specific) shampoos and sprays, topical insecticides or insect growth regulators (IGRs), and oral insecticides can be used directly on pets to control fleas (Durden and Hinkle 2009). Oral insecticides are absorbed into the host's bloodstream and kill adult fleas that attempt to feed on the animal. IGRs are dispersed in the larval fleas' habitat, either through spraying or as a part of an on-animal application to hinder proper larval growth and therefore prevent larvae from developing into viable adults (Durden and Hinkle 2009).
Topical, or spot-on, treatments for pets are available in many formulations. Ensure you are using the appropriate topical treatments for your pet (Durden and Hinkle 2009). Proceed with caution when using topical treatments, and carefully read information regarding proper administration of the product. Always consult with your veterinarian before using any method of chemical control on a pet. Cats and many breeds of dogs are sensitive to some of the chemicals available, and certain health issues or medications that animals may be on could have unfortunate and unforeseen negative consequences.
Other forms of chemical control include IGR applications to carpets or beddings and treating flea-infested homes with insecticides (Durden and Hinkle 2009). Traditional insecticides and other naturally-occurring materials are commercially available; however, some cat flea populations have expressed insecticide resistance, and it is likely that this may occur with the dog flea. Professional pest control services are another option for controlling dog flea populations in the home. With all pesticide use, you should consult with your state's cooperative Extension service for approved materials and your veterinarian regarding your animals' sensitivity, and you should read and understand each insecticide label prior to its use. These steps will help to preserve your pets' health.
The following sites provide additional information on methods of flea control:
Fleas: What They Are, What To Do, University of Florida
Fleas, UF/IFAS Extension (lists of FL approved insecticides)
Selected References
Alcaíno HA, Gorman TR, Alcaíno R. 2002. Flea species from dogs in three cities of Chile. Veterinary Parasitology 105: 261–265.
Baker KP, Elharam S. 1992. The biology of Ctenocephalides canis in Ireland. Veterinary Parasitology 45: 141–146.
Bayer Environmental Science. (2007). Fleas: Order Siphonaptera. Insect Guide. (12 February 2014)
Boushira E, Yoon SS, Roques M, Manavella C, Vermot S, Cramer LG, Ollagnier C, Franc M. 2011. Efficacy of fipronil, amitraz and (S)-methoprene combination spot-on for dogs against adult dog fleas (Ctenocephalides canis, Curtis, 1826). Veterinary Parasitology 179: 351–353.
Chee JH, Kwon JK, Cho HS, Cho KO, Lee YJ, Abdiel-Aty AM, Shin SS. 2008. A survey of ectoparasite infestations in stray dogs of Gwang-ju City, Republic of Korea. Korean Journal of Parasitology 46: 23–27.
Durden LA, Hinkle NC. 2009. Fleas (Siphonaptera). 115–135 pp. In Mullen G, Durden L. (editors). Medical and Veterinary Entomology, 2nd Ed. San Diego, CA. Academic Press.
Durden LA, Judy TN, Martin JE, Spedding LS. 2005. Fleas parasitizing domestic dogs in Georgia, USA: Species composition and seasonal abundance. Veterinary Parasitology 130: 157–162.
Ewing HE, Fox I. 1943. The Fleas of North America: Classification, Identification, and Geographic Distribution of these Injurious and Disease-spreading Insects. Washington, D.C. U.S. Government. 1–142 pp. (8 March 2014)
Farkas R, Gyurkovszky M, Solymosi N, Beugnet F. 2009. Prevalence of flea infestation in dogs and cats in Hungary combined with a survey of owner awareness. Medical and Veterinary Entomology 23: 187–194.
Fox I. 1940. Fleas of Eastern United States. Ames, IA. Collegiate Press.
González A, Castro DC, González S. 2004. Ectoparasitic species from Canis familiaris (Linné) in Buenos Aires province, Argentina. Veterinary Parasitology 120: 123–129.
Hamrick HJ, Drake Jr WR, Jones HM, Askew AP, Weatherly NF. 1983. Two cases of dipylidiasis (dog tapeworm infection) in children: update on an old problem. Pediatrics 72: 114–117.
Hill CA, MacDonald JF. (2007). Fleas: Biology and public health risk. Purdue Extension. (8 March 2014).
Kramer F, Mencke N. 2001. Flea Biology and Control: The Biology of the Cat Flea, Control and Prevention with Imidacloprid in Small Animals. Springer, Berlin, Germany.
Koutinas AF, Papazahariadou MG, Rallis TS, Tzivara NH, Himonas CA. 1995. Flea species from dogs and cats in northern Greece: environmental and clinical implications. Veterinary Parasitology 58: 109–115.
Richman DL, Koehler PG. (1997). Fleas: what they are, what to do. EDIS. (8 March 2014)
Silverman J, Appel AG. (1984). The pupal cocoon of the cat flea, Ctenocephalides felis (Bouché) (Siphonaptera: Pulicidae): a barrier to ant predation. Proceedings of the Entomological Society of Washington 86: 660–663.
Werner A. (2008). Flea Bite and Flea Allergic Dermatitis. Common Skin Diseases. (8 March 2014).
Wall R, Shaw SE, Penaliggon J. 1997. The prevalence of flea species on cats and dogs in Ireland. Medical and Veterinary Entomology 11: 404–406.
Xhaxhiu D, Kusi I, Rapti D, Visser M, Knaus M, Lindner T, Rehbein S. 2009. Ectoparasites of dogs and cats in Albania. Journal of Parasitology Research 105: 1577–1587.



