The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids, and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Several species of entomopathogenic nematodes in the genus Thripinema are known to naturally parasitize thrips (Thysanoptera). Thripinema fuscum Tipping and Nguyen is the most common species in Florida (Figure 1). Thripinema fuscum is economically important because it is a natural enemy of the insect pest, the tobacco thrips, Frankliniella fusca (Hinds).
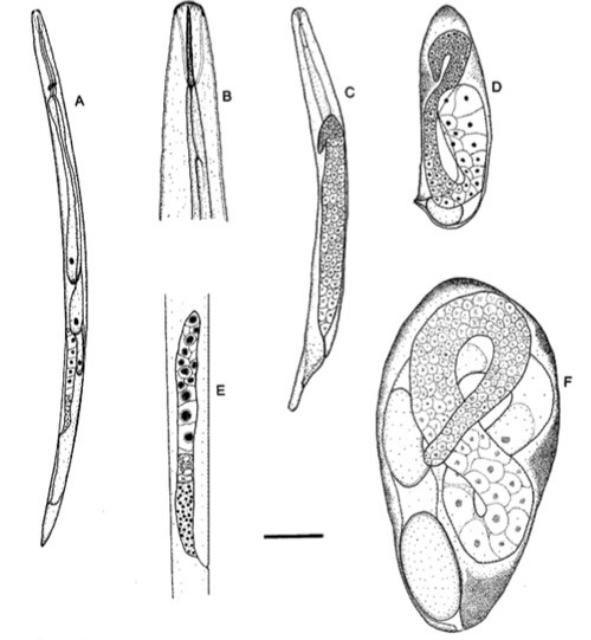
Credits: Journal of Nematology 30: 232–236 (used with permission)
Taxonomy
The first observation of parasitic nematodes of thrips was made by Uzel (1895) in Europe when an unnamed nematode was reported in the body cavity of Thrips physapus L. A nematode inhabiting bean thrips, Heliothrips fasciatus L., was reported in California by Russell (1912), but not described.
The first description of parasitic nematodes of thrips was not made until 1932 by Sharga, who described the nematode Tylenchus aptini from Aptinothrips rufus Gmelin in England. Following that, Lysaught (1936) proposed the name Anguillulina aptini for this species (Tipping 1998).
In 1986, the genus Thripinema was erected by Siddiqi during a taxonomic revision of the species, Howardula (Mason and Heinz 2012). The genus revision included renaming the nematode species described by Sharga (1932) as Thripinema aptini.
Currently, there are five species in the genus Thripinema: Thripinema aptini (Sharga 1932), Thripinema nicklewoodi (Nickle and Wood 1964), Thripinema reniraoi (Reddy et al. 1982), Thripinema khrustalevi (Chizhov et al. 1995), and Thripinema fuscum (Tipping et al. 1998).
Host Range and Distribution
Nematodes in the genus Thripinema are host specific. Currently, the five described species of Thripinema have been recovered from twelve species among eight genera of thrips: Thrips physapus L., Aptinothrips rufus Gmelin, Frankliniella vaccinia Morgan, Frankliniella occidentalis Pergande, Frankliniella fusca Hinds, Taeniothrips vaccinophilus Hood, Stenothrips graminium Uzel, Catinathrips vaccinophilus Hood, Heliothrips species, and Megaluriothrips species (Arthurs 2005).
The distribution of Thripinema most likely reflects where surveys have been conducted, not necessarily the global distribution of the nematodes. Today, Thripinema species are known to occur in Canada, Germany, India, Russia, the United Kingdom, and the United States (Arthurs 2005).
Life Cycle and Biology
Thripinema species are obligate parasites but unlike other entomopathogenic nematodes in the families Steinernematidae and Heterorhabditidae, Thripinema species must keep their host alive for their survival (Funderburk and Latsha 2005). The current understanding of Thripinema biology suggests that the life cycle of each species is similar (Mason and Heinz 2002).
The total development time from egg to adult is 9 days upon entering the host (Sims et al. 2012). The infective female nematode enters the thrips host and becomes vermiform within the thrips hemocoel (Sims et al. 2012). Within 4–5 days of entering the hemocoel, female nematodes oviposit eggs. Eggs hatch into juveniles within 6–8 days of parasitization and feed on fluids within the host's abdominal cavity (Figure 1) (Mason and Heinz 2002, Sims et al. 2012). Fourth-stage juveniles migrate to the hindgut, where males inseminate females before emergence from the anus or ovipositor (Sims et al. 2012).
Little is known about Thripinema species after they emerge from the host. However, free-living Thripinema appear to attack thrips in the moist microclimate within flowers, leaf galls, and the terminal ends of developing foliage (Loomans et al. 1997).
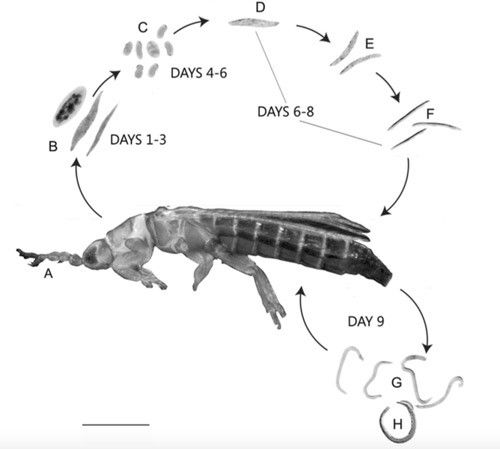
Credits: Journal of Natural History 46: 1111–1128 (used with permission)
Thripinema species do not kill their host and infected thrips do not show obvious signs of parasitism (Arthurs 2005). However, adult female thrips parasitized as larvae do not produce eggs and female thrips parasitized as adults stop laying eggs within 2-3 days (Sims et al. 2005). Parasitism also reduces the feeding of the thrips host (Arthurs and Heinz 2003) and ability of thrips to transmit plant viruses (Sims et al. 2005, 2009).
All stages of thrips are susceptible to parasitism by Thripinema species. Sims et al. (2005) reported that the larval and adult stages are the most susceptible to parasitism whereas Mason and Heinz (2002) found the pupal stage to be the most susceptible. Both male and female thrips may be parasitized by Thripinema species; however, Thripinema show a preference for female thrips (Tipping et al. 1998, Mason and Heinz 2002).
Economic Importance
The Western flower thrips, Frankliniella occidentalis and the tobacco thrips, Frankliniella fusca have the potential to cause significant yield losses in agronomic, fruit, vegetable, and ornamental crops in the United States, including Florida. Both species cause injury to crops through feeding and the transmission of tospoviruses such as Tomato spotted wilt virus. Thripinema nicklewoodi parasitizes Frankliniella occidentalis and Thripinema fuscum parasitizes Frankliniella fusca.
A study conducted by Mason and Heinz (2002) in Texas found that Thripinema nicklewoodi suppressed populations of Frankliniella occidentalis on infested greenhouse chrysanthemum plants. They concluded that augmentative applications of Thripinema nicklewoodi may provide an alternative to current control strategies. Arthur and Heinz (2003) reported that Frankliniella occidentalis parasitized by Thripinema nicklewoodi were less active transmitters of Tomato spotted wilt virus and disease transmission was reduced by 50%.
In Florida peanut production, populations of Frankliniella fusca are naturally suppressed by Thripinema fuscum. Tipping et al. (1998) reported that parasitism of Frankliniella fusca by Thripinema fuscum was 51% in 1995 and 68% in 1996. Additional studies found that parasitism of Frankliniella fusca reduced feeding by 65% and disease transmission by 50% (Sims et al. 2009). To maximize biological control of thrips in peanut production, planting dates may be delayed as flowers are primary sites of parasitism and peanuts flower for the remainder of the growing season (Funderburk and Latsha 2005).
Although Thripinema species have not been commercially mass-produced, they have potential as biological control agents. They are important natural enemies capable of regulating field populations of thrips (Funderburk et al. 2002a, b).
Sampling
Thrips may be collected from the field by selecting flowers, then either striking flowers against a board or exhaling on each flower to extract thrips (Funderburk et al. 2019). Thripinema may be extracted from thrips by placing thrips in a dish of distilled water with one drop of dish liquid and gently swirling them (Tipping 1998) or by cutting open the thrips' abdomen (Funderburk et al. 2002a; 2002b).
Selected References
Arthurs S, Heinz KM. 2003. Thrips parasitic nematode Thripinema nicklewoodi (Tylenchida: Allantonematidae) reduces feeding, reproductive fitness, and Tospovirus transmission by its host, Frankliniella occidentalis (Thysanoptera: Thripidae). Environmental Entomology 32: 853-858.
Arthurs S. 2005.Thrips-parasitic nematodes. Encyclopedia of Entomology. Springer 2004.
Chizhov VN, Subbotin SA, Zakharenkova NN. 1995. Thripinema khrustalevi sp. n. (Tylenchida: Allantonematidae), a parasite of thrips (Thysanoptera) in Moscow. Russian Journal of Nematology 3: 89-94.
Funderburk, J., Martini, X., Freeman, J., Strzyzewski, I., Traczyk, E., Skarlinsky, T., & Adkins, S. (2019). Sampling for estimating Frankliniella species Flower Thrips and Orius species predators in field experiments. JoVE (Journal of Visualized Experiments), (149), e59869.
Funderburk, JE, Ripa R, Espinosa F, Rodriguez F. 2002a. Parasitism of Frankliniella australis (Thysanopera: Thripidae) by Thripinema khrustalevi (Tylenchida: Allantonematidae) isolate Chile. Florida Entomologist 85: 645-649.
Funderburk JE, Stavisky J, Tipping C, Gorbet D, Momol T, Berger R. 2002. Infection of Frankliniella fusca (Thysanoptera: Thripidae) in peanut by the parasitic nematode Thripinema fuscum (Tylenchida: Allantonematidae). Environmental Entomology 31: 558-563.
Funderburk JE, Latsha KS. 2005. The entomophilic Thripinema. Nematodes as Biological Control Agents. CAB International, Wallingford, UK, pp. 401-410.
Loomans AJM, Murai T, Greene ID. 1997. Interactions with hymenopterous parasitoids and parasitic nematodes. In: Lewis, T. (ed.) Thrips as Crop Pests. CAB International, Wallingford, UK, pp. 355-397.
Lysaught AM. 1936. A note on the adult female of Anguillulina aptini (Sharga), a nematode parasitizing Aptinothrips rufus Gmelin. Parasitology 28: 290-292.
Mason JM, Heinz KM. 2002. Biology of Thripinema nicklewoodi (Tylenchida), and obligate Frankliniella occidentalis (Thysanoptera) parasite. Journal of Nematologists 34: 332-339.
Nickle WR, Wood GW. 1964. Howardula aptini (Sharga 1932) parasitic in blueberry thrips in New Brunswick. Canadian Journal of Zoology 42: 843-846.
Reddy YN, Nickle WR, Rao PN. 1982. Studies on Howardula aptini (Nematoda – Sphaerulariidae) parasitic in Megaluriothrips sp. in India. Indian Journal of Nematology 12 1-5.
Russell HM. 1912. The bean thrips (Heliothrips fasciatus Pergande). U.S. Department of Agriculture Entomology Bulletin 118: 1-45.
Sharga US. 1932. A new nematode, Tylenchus aptini n. sp., parasite of Thysanoptera (Insecta: Aptinothrips rufus Gmelin). Parasitology 24: 268-279.
Siddiqi MF. 1986. Tylenchida, parasites of plants and insects. London: CAB Commonwealth Institute of Parasitology.
Sims K, Funderburk JE, Boucias D. 2005. Host-parasite biology of Thripinema fuscum (Tylenchida: Allantonematidae) and Frankliniella fusca (Thysanoptera: Thripidae). Journal of Nematologists 37: 1-8.
Sims KR, Funderburk JE, Reitz SR, Boucias DG. 2009. The impact of a parasitic nematode Thripinema fuscum (Tylenchida: Allantonematidae) on the feeding behavior and vector competence of Frankliniella fusca (Thysanoptera: Thripidae). Entomologia Experimentalis et Applicata 132: 200-208.
Sims KR, Becnel JJ, Funderburk JE. 2012. The morphology and biology of the entomophilic Thripinema fuscum (Tylenchida: Allantonematidae), and the histopathological effects of parasitism on the host Frankliniella fusca (Thysanoptera: Thripidae). Journal of Natural History 46: 1111-1128.
Tipping C, Nguyen KB, Funderburk JE, Smart GC Jr. 1998. Thripinema fuscum n. sp. (Tylenchida: Allantonematidae), a parasite of the tobacco thrips, Frankliniella fusca (Thysanoptera). Journal of Nematology 30: 232-236.
Uzel H. 1895. Monographie der Ordnung Thysanoptera. Koniggratz, pp. 472.