Tomatoes are a major commodity in Florida, with an estimated production value of $453 million and accounting for 18% of Florida's fresh market vegetable acreage, and 35% of the fresh market tomato acreage in the US (USDA/NASS, 2021). Among the many pests and diseases that affect tomatoes, nematodes are one of the major problems. Since the ban on methyl bromide, these ubiquitous soil pests have become much more difficult to manage. This article will discuss the use of nematode-resistant tomato cultivars as a tool to help manage root-knot nematodes in Florida.
Root-knot disease in tomato is a serious and growing problem in Florida and is mainly due to three major tropical root-knot nematode (RKN) species, i.e., Meloidogyne javanica, M. incognita and M. arenaria. RKNs (Meloidogyne spp.) are broadly distributed and are generally considered to be the most important plant-parasitic nematodes worldwide. RKNs are sedentary, obligate parasites and are particularly difficult to control due to wide host ranges, short generation times, and high reproductive rates. Among a range of vegetable crops affected by RKNs, tomato is a very good host for all three major tropical root-knot nematode species in Florida. Common symptoms exhibited by tomato plants due to severe RKN infection can be categorized as:
- Above-ground symptoms
Symptoms resemble nutrient deficiencies such as yellowing of leaves and stunted growth.

Credit: J. Desaeger, UF/IFAS
- Below-ground symptoms
Galled roots are the typical symptom caused by root-knot nematode.

Credit: J. Desaeger, UF/IFAS
Fortunately, tomato is one of the few vegetable commodities in which host resistance against root-knot nematode is well established. Resistance in tomato against root knot nematode is generally governed by a single dominant gene Mi, which confers resistance to M. incognita, M. arenaria, and M. javanica (Gilbert and McGuire 1956). The Mi gene was identified during the 1940s in a wild relative of tomato Solanum peruvianum and was later introgressed into cultivated tomato (Smith 1944).
Tomato varieties carrying the Mi gene are effective in controlling Meloidogyne spp. and can be cultivated in RKN-infested fields without significant yield loss (Figure 2).

Mi resistance against RKNs is characterized by a hypersensitive response of the plant causing localized cell death of host tissue near the invading nematodes (Williamson and Kumar 2006). A typical root-knot nematode life cycle (Figure 3) involves root invasion by the second-stage juvenile (J2), and formation of giant cells (specialized feeding cells within the root galls) that allow the nematodes to develop to adult females with eggs. Newly emerging juveniles then move through the soil to invade more roots. In roots of Mi tomato cultivars, the formation of giant cells required by the invading nematode, is largely prevented. Root-knot nematode juveniles may still penetrate and migrate through roots of nematode-resistant cultivars (although in lower numbers), but most of them will not be able to feed and reproduce. They either leave or starve within the root.
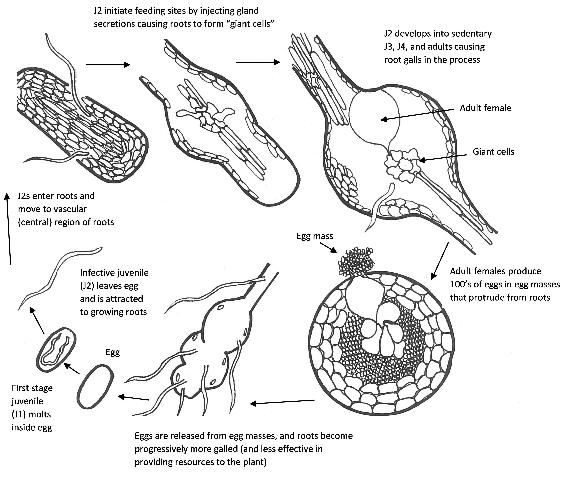
Credit: H. Regier, adapted from G. Abawi and V. Brewster
Mi gene in tomato has also induced resistance to some piercing and sucking insects such as aphids and whiteflies (Kaloshian et.al. 1995; Nombela et.al. 2003), although this effect is generally insufficient for providing field-level resistance against such pests.
The level of RKN resistance in Mi-gene-bearing tomatoes can vary from good to intermediate among cultivars. For example, Sanibel, Daytona, and Mariana exhibit good resistance to RKNs while Skyway 687, Southern Ripes, SV 7631TD, and Felicity possess intermediate resistance against the same nematodes.
Although Mi-nematode-resistant tomato cultivars may be valuable to Florida tomato growers, there are certain risks associated with these:
- To date, Mi is the only nematode resistance gene that has been introgressed into tomato. Although several other genes have been reported, none of these have been successfully incorporated in commercial tomato cultivars.
- Resistance breakdown - There is always a chance that RKNs become insensitive to the Mi gene. This is more likely when RKNs are continuously exposed to root-knot-resistant cultivars, especially in monoculture systems. Resistance-breaking populations of M. incognita have been reported since the 1990s, and these have become very widespread in California.
- Heat instability – Although the Mi gene is highly effective, resistance is compromised at high soil temperatures (=32°C or 90°F). Some researchers have reported a complete loss of function of Mi at high soil temperatures of (Dropkin 1969; Williamson 1998), while others have found that the gene regains function once the soil temperature lowers (de Carvalho 2015). In Florida, trials conducted by D. W. Dickson (personal communication) in Citra, FL, and by ourselves in Wimauma, FL, showed no breakdown of resistance in Mi tomato cultivars—neither in spring- nor fall-planted tomatoes—suggesting that soil temperatures may not often exceed the limits for maintaining Mi-based resistance.
- Target specificity - The Mi gene is effective against the three major tropical and sub-tropical RKNs, but it does not work against another tropical RKN, M. enterolobii. M. enterolobii, the guava root-knot nematode, was first reported in the United States in Florida (Brito et al. 2007), but has now been found in several other southeastern states. Mi is also ineffective against M. hapla, the northern root-knot nematode, which is common in the northern United States and Canada. This species has been reported in several strawberry farms in Florida where the seedlings are imported from Canada and more northern parts of United States, but the nematode does not appear to be common in Florida tomato fields.
Final note: The Mi gene in tomato provides reasonable protection against major tropical and sub-tropical root-knot nematodes, but soil fumigation may still be needed for management of other pests. Besides nematodes, soilborne pathogens are endemic to Florida tomato fields, and major disease challenges include fusarium wilt, caused by Fusarium oxysporum f. sp. lycopersici; fusarium crown and root rot, caused by Fusarium oxysporum f. sp. radicus-lycopersici; Pythium, Phytophthora; southern blight caused by Sclerotium rolfsii; and bacterial wilt caused by Ralstonia solanacearum. Additionally, there are many weeds in tomato fields that can compete with the crop, and not all of these can be controlled through the use of herbicides. Soil fumigation may therefore be needed to simultaneously manage all these different soilborne pests and diseases. Thus, although tomato cultivars with the Mi gene may not be a replacement for soil fumigants in Florida, they may be a valuable tool for those tomato growers who choose not to fumigate, or for growers to use in combination with soil fumigation for fields with high root-knot-nematode pressure.
References
Brito, J. A., J. D. Stanley, R. Kaur, R. Cetintas, M. Di Vito, J. A. Thies, and D. W. Dickson. 2007. "Effects of the Mi-1, N and Tabasco genes on infection and reproduction of Meloidogyne mayaguensis on tomato and pepper genotypes." Journal of nematology 39(4): 327–332.
Castagnone-Sereno, P. 2012. "Meloidogyne enterolobii (= M. mayaguensis): Profile of an emerging, highly pathogenic, root-knot nematode species." Nematology 14(2): 133–138. https://doi.org/10.1163/156854111X601650
Crop protection: Crop Nematodes of Vegetables. Retrieved from https://agritech.tnau.ac.in/crop_protection/nematology/crop_prot_nematode_veg_root_nemo.html
de Carvalho, L. M., N. D. Benda, M. M. Vaughan, A. R.Cabrera, K. Hung, T. Cox, Z. Abdo, L. H. Allen, and P. E. A.Teal. 2015. "Mi-1-Mediated Nematode Resistance in Tomatoes is Broken by Short-Term Heat Stress but Recovers Over Time." Journal of Nematology. 47(2): 133–140. https://pubmed.ncbi.nlm.nih.gov/26170475/
Dropkin, V. H. 1969. "The necrotic reaction of tomatoes and other hosts resistant to Meloidogyne: reversal by temperature." Phytopathology 59: 1632–1637.
Eugene M., and M. Ozores-Hampton. 2017. "Tomato Varieties for Florida." The Florida Tomato Proceeding. pp 32–33.
Freeman, J. H., E. J. Mcavoy, N. S. Boyd, R. Kanissery, M. Ozores-Hampton, H. A. Smith, and G. E. Vallad. 2018. Tomato Production. HS1189. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/hs1189
Gilbert, J. C., and D. C. McGuire. 1956. Inheritance of resistance to severe root-knot from Meloidogyne incognita in commercial type tomatoes. Proc. Am. Soc. Hortic. Sci. 68:437–42.
Kaloshian, I., W. H. Lange, and V. M. Williamson. 1995. "An aphid-resistance locus is tightly linked to the nematode-resistance gene, Mi, in tomato."Proceedings of the National Academy of Sciences of the United States. 92: 622–625. Retrieved from https://www.pnas.org/content/pnas/92/2/622.full.pdf
Milligan, S. B., J. Bodeau, J. Yaghoobi, I. Kaloshian, P. Zabel, and V. M. Williamson. 1998. "The root knot nematode resistance gene Mi from tomato is a member of the leucine zipper, nucleotide binding, leucine-rich repeat family of plant genes." The Plant Cell 10(8): 1307-1319.
Nombela, G., V. M. Williamson, and M. Muñiz. 2003. "The Root-Knot Nematode Resistance Gene Mi-1.2 of Tomato Is Responsible for Resistance Against the Whitefly Bemisia tabaci." 16(7): 645–649. Retrieved from https://apsjournals.apsnet.org/doi/pdfplus/10.1094/MPMI.2003.16.7.645
Smith, P. G. 1944. "Embryo culture of a tomato species hybrid." Proceedings of the American Society for Horticultural Science. 44: 413–6.
USDA National Agricultural Statistics Service (USDA NASS). 2021. "Quick Stats." Accessed 8 December 2021. https://quickstats.nass.usda.gov/
Verdejo-Lucas, S., M. Blanco, L. Cortada, and F. Javier Sorribas. 2013. "Resistance of tomato rootstocks to Meloidogyne arenaria and Meloidogyne javanica under intermittent elevated soil temperatures above 28°C." Crop Protection 46: 57–62.
Williamson, V. M. 1998. "Root-knot nematode resistance genes in tomato and their potential for future use." Annual Review of Phytopathology. 36: 277–293.
Williamson, V. M., and A. Kumar. 2006. "Nematode resistance in plants: The battle underground." Trends in Genetics. 22:396–403.