House Dust Mites, Dermatophagoides spp. (Arachnida: Acari: Pyroglyphididae)
The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The term "house dust mites" has been applied to a large number of mites found in association with dust in dwellings. The American house dust mite, Dermatophagoides farinae Hughes, and the European house dust mite, Dermatophagoides pteronyssinus (Trouessart), are discussed here. The first permanent structures for houses date back to 6,000 to 5,000 BCE, but it was not until the late 1600s that scientists became interested in the dust of houses. The pyroglyphids are parasites associated with birds and/or mammals. Kern (1921) found house dust to give positive cutaneous reactions in sensitive patients. Cook (1922) and Coa (1922) also found that dust extracts gave positive skin reactions in over 30% of the individuals tested. Voorhorst et al. (1964) found that mites contribute to the house dust allergy problem.
Credit: Division of Plant Industry
Distribution
House dust mites are nearly cosmopolitan in distribution as they are associated with house dust and bird nests.
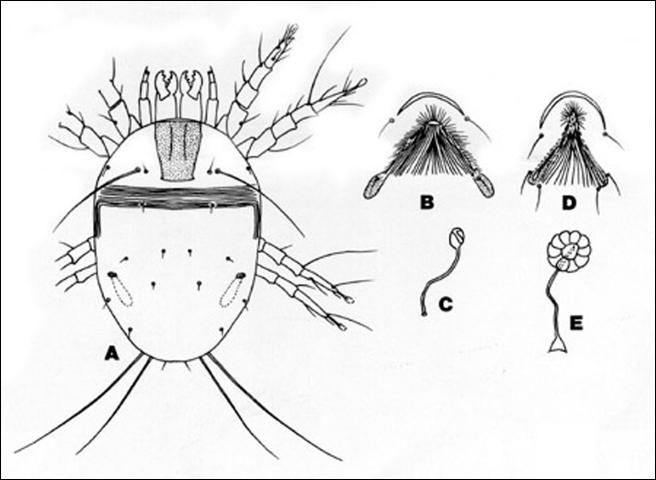
Description
Both male and female adult house dust mites are globular in shape, creamy white and have a striated cuticle. The female measures approximately 420 microns in length and 320 microns in width. The male is approximately 420 microns long and 245 microns wide. A pair of suckers on the ventral posterior idiosoma of the male is used to grasp the female during copulation. Males are more sclerotized with enlarged legs I and III. The male aedeagus is located between the apodemes of leg IV. The female has a well-defined genital opening anterior to the bursa copulatrix located near the anus (Suggars 1987).
Life Stages and Biology
The life cycles of these two mite species include egg, active larva, resting larva (pharate tritonymph), active tritonymph, resting tritonymph (pharate adult), and active adult stages. Between 19 and 30 days are needed to complete a life cycle depending upon the temperature and humidity (Furumizo 1973). Mated females live about two months. A male may attach itself to a tritonymph female and mate when she reaches the adult stage. Dermatophagoides farinae lays eggs over a 30-day period, producing about an egg a day, while Dermatophagoides pteronyssinus lays about 80 eggs over a 45-day period. There is a general agreement that house dust mites in the home feed on shed skin of humans. The average individual sheds 0.5 to 1.0 gram (~0.02 – 0.04 oz) of skin daily. Spieksma et al. (1971) reported that the mites were sensitive to relative humidity and at 60% or lower the mite population stops growing and dies out.

Credit: G. W. Wharton
Hosts
House dust mite hosts include mammals, particularly humans. They can also be found in bird nests, and occasionally in bee hives.
Economic Importance
Because of the medical implications, house dust and mites associated with house dust have been tested for the source of the house dust allergen. Mites and insects that inhabit buildings as well as bacteria, fungi, and algae appear to have allergenic properties. The influence of house dust mites on the overall problem of dust allergies is not clearly known. In dust samples taken by the senior author and those taken by others, the American and European house dust mite are usually found in high numbers (Yoshikawa and Bennett 1979). Davies (1958) reported the highest house dust allergen activity was found in dust samples stored at 85% relative humidity. Mite analyses were not included in Davies' report. The age of the dust also appears to be a factor in the degree of allergen activity. Mite allergens are mainly present in feces of house dust mites and may become airborne and inhaled by patients, giving rise to asthma, rhinitis, or atopic dermatitis (eczema) (Van Bronswijk 1981). For a complete review of house dust mites and the related problems see Wharton (1976) and van Bronswijk (1981).
Survey and Detection
A number of complicated procedures have been published for separating mites from dust collected from vacuuming mattresses, carpets, sofas, and chairs. A simple method for detecting the presence of mites is to put a small amount of dust on the surface of water and examine it under 20X magnification. Live and dead mites will remain on the surface of the water. Furumizo (1973) reported a sieving and flotation technique. Shamiyeh et al. (1973) placed a 0.05 g sample of dust in 30 mL of a saturated NaCl solution and added 5 drops of detergent. The dust was teased apart and subjected to ultrasonic treatment for 20 minutes. The suspension was rinsed through a 45-micron mesh sieve, stained with crystal violet and examined under a stereo microscope. Hirschmann (1986) reported that dust mites were attracted to white DIN-A4 sheets of typewriter paper placed where the dust mites are suspected. Paper is examined in the mornings and evenings by holding the paper over a lamp. The mites cast a shadow.
Management
To manage mite populations, reduce the humidity below 70% and thoroughly vacuum mattresses, carpets, sofas, and chairs. Very sensitive individuals should encase their mattresses in plastic. Use drapes that can be washed and change bed clothing frequently. Vacuuming does not always remove all the live mites, and Korsgaard (1982) stresses reducing humidity over sanitation. No one method has been found for reducing mites and relieving allergy suffering. Immunotherapy, i.e., injections of mite extracts into the patients to increase antibody level, has had variable success (Munro-Ashman et al. 1976).
Selected References
Coa, A.F. 1922. Studies in specific hypersensitiveness. V. The preparation of fluid extracts and solutions for use in the diagnosis and treatment of allergies with notes on the collection of pollens. J. Immunol. 7:163–168.
Cook, R.A. 1922. Studies in specific hypersensitiveness. IV. New etiologic factors in bronchial asthma. J. Immunol. 7:147–162.
Davies, R.R. 1958. Moulds in dust and air. Thesis, Univ. of London.
De Saint-Georges-Gridelet, D. 1981. Mise au point de' une strategie de controle de l'acarien des poussieres (Dermatophagoides pteronyssinus) par utilisation de'un fongicide. Acta Oecologica. 2: 117–126.
Furumizo, R.T. 1973. The biology and ecology of the house dust mite Dermatophagoides farinae Hughes, 1961 (Acarina: Pyroglyphidae) Ph.D. dissertation, University of California, Riverside. 143 pp.
Hirschmann, W. 1986. Simple method for identifying live house dust mites. The Practical Pest Controller. 38:1–2.
Ker, R.A. 1921. Dust sensitization in bronchial asthma. Med. Clin. N. America. 5:751–758.
Korsgaard, J. 1982. Preventative measures in house dust allergy. Am. Rev. Respir. Dis. 125:80–84.
Leysen, M. Th., G. Reybrouck, and H. Van de Voorde. 1974. Susceptibility of the house dust mite to pesticides and disinfectants. Acta Allergologica. 29:455-461.
Mallea, M. and J. Charpin. 1977. Activities fongistatique et fongicide du paragerm forte ambiance sur les champignons. Allergologia et Immunopathologia. 5:536.
Mitchell, E.B., S. Wilkins, J. McCallum Deighton, and T.A.E. Platts-Mills. 1985. Reduction of house dust mite allergen levels in the home: Uses of the acaracide, pirimiphos methyl. Clinical Allergy. 15:235–240.
Munro-Ashman, D., and A.W. Frankland. 1976. Treatment of allergy to house dust with pyridine-extracted alum-precipitated extracts of the house dust mite. Annals of Allergy 36:95–98.
Oshima, S. 1964. Observations of floor mites collected in Yokokama. I. On the mites found in several schools in summer. Japanese Journal of Sanitary Zoolog. 15:233–244 (in Japanese with English summary).
Penaud. A., J. Mouirit, P. Autran, P. Timon-David, M. Jacquet-Francillon, and J. Charpin. 1973. Reflexions sur les moyens de lutte contre les acariens pyroglyphides des poussieres domestiques. Marseille-Medical. 110:377–380.
Shamiyeh, N.B., R.P. Hornsby, S.E. Bennett and N.L. Woodill. 1973. Distribution of house dust mites in the environs of a Tennessee River Valley. J. Econ. Entomol. 66:998.
Spieksma, F. Th. M., P. Zuidema and M.J. Leupen. 1971. High altitude and house-dust mites. British Med. J. 1:82–84.
Suggars, A.L. 1987. House Dust Mites: A Review. J. Entomol. Sci. Suppl. 1:3–15.
van Bronswijk, J.E.M.A. 1981. House dust biology. NIB Publishers, The Netherlands. 314 pp.
Van de Lustgraaf, B. 1977. Xerophilic fungi in mattress dust. Mykosen. 20:101–106.
Van de Lustgraaf, B. 1978. Ecological relationships between xerophilic fungi and house dust mites (Acarina: Pyroglyphidae). Oecologia (Berlin). 33:351–359.
Voorhorst, R., M.I.A. Spieksma-Boezeman and F. Th. M. Spieksma. 1964. Is a mite (Dermatophagoides sp.) the producer of the house-dust allergen? Allergie u. Astma. 10:329–334.
Wharton, G.W. 1976. House dust mites. J. Med. Entomol. 12:577–621.
Yoshikawa, M. and P.H. Bennett. 1979. House dust mites in Columbus, Ohio. Ohio J. Sci. 79:280–282




