This publication is part of a series titled Soil Phosphorus Storage Capacity (SPSC) for Phosphorus Risk Assessment and Management. The series is intended for use by soil scientists, environmental consultants, state agency personnel, Extension faculty, and others who are interested in management practices and policies that minimize the risk of phosphorus loss from soils.
Introduction
Phosphorus (P) loss from agricultural fields results in environmental degradation and waste of P as a fertilizer resource. The purpose of this publication is to describe a practical means of determining when a soil has reached a level of P loading that constitutes an environmental risk. It is intended for use by soil scientists, environmental consultants, state agency personnel, Extension faculty, and others with an interest in the potential of P loss from soils.
Why Is Soil Test Phosphorus (STP) Not a Suitable Indicator of Environmental P Risk?
Soil test P (STP) procedures such as Mehlich 1-P fail to precisely indicate whether a given soil is capable of retaining additional P (P "sink") as opposed to functioning as a P source that would pose an environmental risk. By using the same STP extract, such as the Mehlich 1 (M1) extract, and analyzing it for iron (Fe) and aluminum (Al), a P saturation ratio (PSR) can be calculated, as follows:
PSR M1= (Mehlich 1-P/31) / ((Mehlich 1-Fe/56) + (Mehlich 1-Al/27)) Equation 1
This ratio of sorbed P (P held by the soil) to the sum of the components (Fe and Al) to which it is mainly sorbed is a useful indicator of the "change point" at which a soil becomes a P source to the environment. In other words, the risk of phosphorus moving from agricultural fields or lawns greatly increases when the soil reaches the change point, which can be estimated using this calculation based upon soil testing.
The Change Point
Since P concentrations in the soil solution are related to P losses via surface and subsurface flow, the PSR can be used to determine the point of P loading at which P release begins to abruptly increase and become an environmental concern. The amount of P lost from a soil after application of a P-rich amendment (e.g., inorganic fertilizer, biosolids, poultry manure, dairy manure, etc.) will depend on the solubility of the material and sorption capacity of the soil—the net effects of which can best be determined by extraction with water. When soils are extracted with water and plotted against PSR calculated using the Mehlich 1 extractant (PSRM1), we get a "change point", also referred to as the "Threshold PSR" (Figure 1). The change point is defined as that point where P concentrations in the soil solution abruptly increase. In Figure 1, the change point is 0.1.
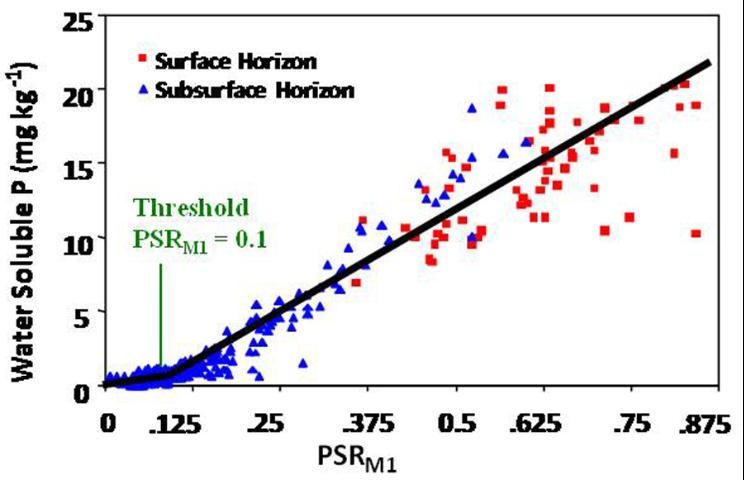
Change Points for Flatwood Soils Containing a Bh Horizon
Excess P can move vertically through the soil profile and come into contact with the spodic (Bh) horizon of Spodosols. It is therefore important to know the change point for Bh horizons (Figure 2).
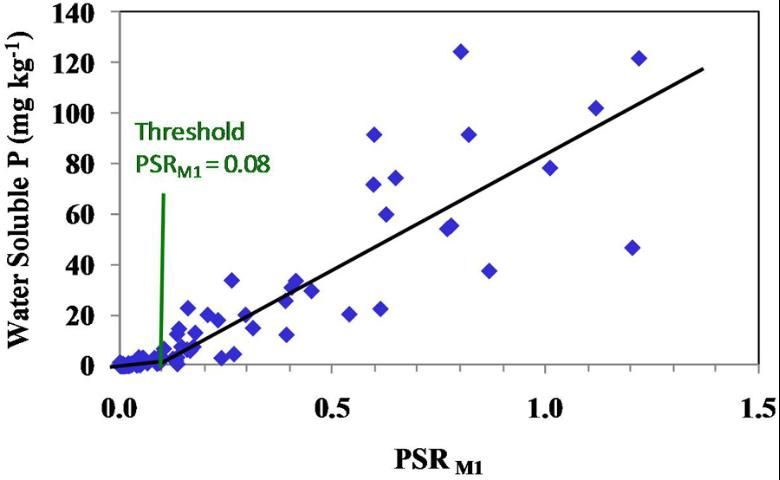
We can now use the change point of Bh horizons as an indicator of P release that would be environmentally detrimental. The change point for Bh horizons of Florida Spodosols has been determined to be 0.08 (Figure 2).
How Can We Use the Change Point to Evaluate Environmental P Risk and Agronomic Needs Simultaneously?
Since P concentrations in the soil solution are related to P losses from a field via surface and subsurface flow, the PSR can be used to determine when P release from a soil reaches the point of environmental concern. Using this scenario, P could be applied to the soil at an agronomic rate based on the normal UF/IFAS recommendations unless the PSR is greater than 0.1 for surface soils. Depending on how much the PSR is greater than 0.1, a recommendation to apply P only at crop recovery quantities could be made. For surface horizons, depth of sampling could follow UF/IFAS protocols for soil testing. Deeper sampling may be called for where there is concern for subsurface release of P. The spodic horizon becomes an environmental P risk when PSR M1 is greater than 0.08.
Why Is the Use of a "Change Point" PSR Better Than STP?
Here's an example:
When two soils have the same STP value (e.g., 50 mg kg-1 Mehlich 1) but have different abilities to hold P (i.e., different Fe+Al values), then the soil (Soil 1) with the higher (Fe+Al) will have a lower PSR value compared to the soil (Soil 2) with a lower (Fe+Al) value. For example, if Soil 1 has a PSR value of 0.05 (below the threshold PSR of 0.1), and Soil 2 has a PSR of 0.2 (greater than the threshold PSR of 0.1), then Soil 1 will not be of an environmental P risk while Soil 2 would release P.
Can We Use Any Other Extractant for Determining PSR?
The original method of calculation of the PSR specified oxalate-extractable P, Fe, and Al (Breeuwsma and Silva 1992; Equation 2). Modifications are based on STPs used in various parts of the US Mehlich 1 extracts (Nair et al. 2004) and Mehlich 3 (Maguire and Sims 2002; Sims et al. 2002; Nair et al. 2004) extracts have been shown to be suitable to calculate PSR for sandy soils of the Southeastern US.
In addition to using oxalate P, Fe, and Al values (Equation 2), we can use Mehlich 1 P, Fe, and Al (Equation 1) or Mehlich 3 P, Fe, and Al (Equation 3) for PSR calculations.
PSROx = (Oxalate -P/31) / ((Oxalate -Fe/56) + (Oxalate -Al/27)) Equation 2
PSRM3 = (Mehlich 3-P/31) / ((Mehlich 3-Fe/56) + (Mehlich 3-Al/27)) Equation 3
Note that the "change point" PSR will be dependent on the method used in its calculations. Therefore, when reporting a PSR value, it is necessary to indicate the matrix [e.g., PSRM1 (PSR calculated using P, Fe, and Al in a Mehlich 1 solution), PSRM3 (PSR calculated using P, Fe, and Al in a Mehlich 3 solution), or PSROx (PSR calculated using P, Fe, and Al in an oxalate solution)]. For example, the change point using Mehlich 3-P, Fe, and Al is 0.08 and, when using oxalate extraction, is 0.10 for A and E horizons.
References
Breeuwsma, A., and S. Silva. 1992. "Phosphorus Fertilisation and Environmental Effects in The Netherlands and the Po Region (Italy)." Rep. 57. Agric. Res. Dep. The Winand Staring Centre for Integrated Land, Soil, and Water Research. Wageningen, The Netherlands.
Maguire, R. O., and J. T. Sims. 2002. "Soil Testing to Predict Phosphorus Leaching." J. Environ. Qual. 31:1601–1609.
Nair, V. D., K. M. Portier, D. A. Graetz, and M. L. Walker. 2004. "An Environmental Threshold for Degree of Phosphorus Saturation in Sandy Soils." J. Environ. Qual. 33:107–113
Sims, J. T., R. O. Maguire, A. B. Leytem, K. L. Gartley, and M. C. Paulter. 2002. "Evaluation of Mehlich 3 as an Agri-environmental Soil Phosphorus Test for the Mid-Atlantic United States of America." Soil Sci. Soc. Am. J. 66:2016–2032.
Acknowledgments
This study was supported in part by a grant from the Florida Department of Agriculture and Consumer Services. The authors thank Tom Obreza and Ed Hanlon for their valuable comments on the draft version of this publication.