Introduction
Attempts to develop and successfully operate facilities that commercially exploit crocodilians have always faced considerable challenges. Huge ongoing investments in money, time, and labor are required with scant returns for several years (Elsey et al. 1994; Lever 1994). Even then, profits are not guaranteed in an unforgiving industry with a volatile meat and hide export market (Cox and Rahman 1994; David 1994; Kelly 1994). The difficulties in running a crocodilian facility are further compounded in developing countries where the basics, like reliable sources of electricity, running water, appropriate land, and cheap, fresh crocodile food, are difficult to obtain (Child 1987; Cox and Rahman 1994). In developing countries, inability to acquire the appropriate technology and licenses to propagate intensively and market crocodilians are also prohibitive factors to success (Magnusson 1984; Cox and Rahman 1994). All of these aspects, along with the misleading beliefs that crocodiles are simple to keep in captivity and will provide substantial profits, have resulted in very few successful crocodilian businesses in the developing world (Mazzotti 1987; David 1994).
The three types of consumptive uses involving crocodilians, recognized by the Convention on International Trade in Endangered Species (CITES) and the Crocodile Specialist Group (CSG), are ranching, wild harvesting, and captive breeding (Cox and Rahman 1994). Ranching involves collecting eggs or juveniles from the wild and rearing them to market size in grow-out facilities. A certain percentage of the eggs or juveniles collected in ranching operations can then be returned to the wild at a size that should ensure a higher survival rate (Mazzotti 1987; Cox and Rahman 1994; Elsey et al. 1994). Adults and sub-adults can also be harvested directly from the wild (Mazzotti 1987; Cox and Rahman 1994). These methods are considered "open" cycle as they are ultimately concerned with the health and viability of wild populations and habitat in order to maintain success (Thorbjarnarson 1992; Cox and Rahman 1994). On the other hand, captive breeding that relies solely on rearing captive-born animals to produce skins and meat for market is considered "closed" cycle (Thorbjarnarson 1992; Cox and Rahman 1994). Captive breeding enterprises can also include a tourism element to supplement their income, with crocodiles placed on display for viewing (Cox and Rahman 1994).
For "open cycled" programs to be successful requires an ongoing commitment of funding for research and monitoring of wild crocodile populations and ecosystems (Hines and Abercrombie 1987; David 1994). This will ensure that harvesting or ranching can be effectively managed and strategies adapted to prevent any detrimental impact on the wild population (Hines and Abercrombie 1987; Hollands 1987; David 1994). In the majority of places, crocodile populations can usually withstand high levels of egg ranching (Cox and Rahman 1994). However, intensive harvesting of adults cannot always be sustained at the high levels required to ensure economic profitability for commercial operations (Elsey et al. 1994). One other element to be considered when engaging in ranching or harvesting is local drought and flood patterns. Prolonged and severe drought or flood can significantly reduce the productivity and availability of wild crocodilians (Hollands 1987; David 1994).
The success of ranching or harvest operations in places such as Australia and Zimbabwe has relied upon years of research into wild populations, stability of wild populations, availability of resources, and adequate logistical and financial support (Bolton 1990). Failures in these areas have led to the demise of a number of ranching programs in the developing world (Bolton 1990; Hines 2002). Although it is important to be economically viable, the primary goal of programs should be conservation-based to ensure overall long-term viability and success (Joanen and McNease 1987; Mkanda 1992).
"Closed-cycle" programs provide no incentive to protect or preserve native populations of crocodilians and have proven largely unsuccessful and difficult to get off the ground in developing countries (Magnusson 1984; Elsey et al. 1994). The costs of feeding and maintaining crocodiles from hatchlings to marketable slaughter sizes usually outweigh the returns gained from skins, meat, and other products (Suvanakorn and Youngprapakorn 1987). Many unforeseen problems are commonly encountered on crocodilian farms. The most devastating problems include outbreaks of disease, mass mortalities, and a depressed market that has led to the collapse and abandonment of many farms (Webb et al. 1987; Kelly 1994). However, captive breeding and rearing is important to the long-term economic viability of any crocodile enterprise (Davis 1994). If it is suitable, a tourism aspect can also be included to gain extra revenue. This can significantly reduce overhead costs and accommodate for down cycles in the hide and meat market by providing a regular income, thus improving the viability of a facility (Child 1987; Cox and Rahman 1994; Kelly 1994).
Despite the difficulties associated with crocodilian enterprises, there are guidelines that, if implemented, can increase chance of success and reduce mortality. Combining captive breeding with ranching and tourism may be the best overall approach for linking conservation of crocodiles with economic benefits to local communities and governments (Mazzotti 1987; Thorbjarnarson 1992).
Facility Planning
There are a number of options for housing captive crocodilians that can be adapted to suit the individual goals of any facility, such as acres of lake and marsh habitat enclosed to house many animals or as small pens used for one or two individuals (Figure 1 and Figure 2). Adult crocodilians require a great deal of space, and, considering their longevity (often 50 years or more) and large adult size (Thorbjarnarson 1992), careful planning for future housing needs of the facility should be at the forefront of enclosure design. Careful and comprehensive planning becomes even more important when the facility is intended to house "nuisance" crocodiles. In these cases the number of animals requiring space may increase significantly and unpredictably over several years.

Credit: Michael Cherkiss (2006)

Credit: Matthew Brien, 2006
Enclosures can be designed for exhibit. Exhibit enclosures are those intended to provide ample viewing opportunities for public observation of the animals. They require the most careful planning and construction. Enclosures that are used to house animals not intended for public display (e.g., breeding, rearing, and holding) can often be constructed with less financial investment. Both enclosure types share similar minimal requirements of providing safe and secure confinement in a manner that also provides for the health and well-being of crocodiles (QLD 1997; Ziegler 2001). Exhibit enclosures must also account for public safety and be aesthetically pleasing (Figure 3). Off-exhibit enclosures can often be far more utilitarian in terms of landscape design and orientation (Figure 4). Off-exhibit enclosures can generally be designed to accommodate more individuals while requiring lower construction and maintenance costs (Ziegler 2001).

Credit: Michael Cherkiss, 2006

Credit: Matthew Brien, 2006
Regardless of enclosure type, it is important to realize that the success of any enclosure relies on how effectively it can be cleaned on a regular basis with minimal disturbance to the crocodiles (Hutton and Webb 1994; Vernon 1994; Ziegler 2001).
Many factors influence decisions that facilities make when constructing crocodile enclosures. The two most restrictive are space and budget. Carefully weighing options and having a solid plan for the future of the facility will save a great deal of money and time while accomplishing the minimum goal of safe and humane confinement.
Crocodilian Species—Central America and the Caribbean
In most commercial situations it is recommended that local species of crocodilians are utilized over non-native. This will avoid the undesirable situation whereby an exotic species escapes into local waterways and breeds. Here we focus on two species native to Central America and the Caribbean, the American crocodile (Crocodylus acutus) and the Morelet's crocodile (Crocodylus moreletti) (Figure 5 and Figure 6). While the American crocodile (Crocodylus acutus) can reach lengths of up to 4 m, the Morelets crocodile (Crocodylus moreletti) is a slightly smaller species attaining lengths up to 3 m (Brazaitis 1973). Each species possesses unique physical characteristics. The American crocodile has a long, slender snout and the most irregular dorsal armor of any crocodilian (Thorbjarnarson 1992). The Morelet,s crocodile typically has a shorter, broader snout and more regular dorsal armor (Thorbjarnarson 1992). However, dependent upon location, the two species can often be almost indistinguishable. Although their ranges overlap, C. acutus tend to inhabit coastal or brackish environments whereas C. moreletti are found primarily in fresh water and low-salinity coastal habitats (Thorbjarnarson 1992). Both C. acutus and C. moreletti have been successfully housed and bred in captivity (Groombridge 1987; Lang 1987; Ojeda et al. 1998). Both species possess a commercially high-quality skin, are relatively non-aggressive, and have been found to demonstrate a high tolerance of conspecifics (Lang 1987a; Thorbjarnarson 1992). Due to the high quality of their skin, both species have been targeted extensively by hunters, leading to their classification under Appendix 1 (CITES), and they are considered endangered (Thorbjarnarson 1992).

Credit: Michael Cherkiss, 2006

Credit: Wayne King
Housing of Crocodilians
Elevated stress levels in captive crocodilians can lead to poor growth and survivorship, disease, poor skin quality, and reproductive failure (Elsey et al. 1994; Lance 1994; Smith and Marais 1994). The overall success of any facility holding crocodilians relies upon reducing the amount of stress to individuals by providing and maintaining high water quality, adequate temperatures, appropriate cover, and a high standard of food quality and hygiene (Elsey et al. 1994; Hutton and Webb 1994; Smith and Marais 1994). As the well-being of the crocodiles and the economic success of the facility are inextricably linked, the following sections should be carefully considered when housing crocodiles.
Enclosure Size
The guidelines given here are the minimum standards generally accepted by zoological facilities for all species of crocodilian. Ideally, space and funds permitting, enclosures should be larger than the examples given here. If crocodiles are kept in small exhibits where movement is restricted, they will soon become lethargic and obese. This will increase their susceptibility to cardiac and other diseases (Pooley 1990; Elsey et al. 1994). Cramped housing of multiple animals will lead to undue aggressive behavior, greater injuries, and increased stress (Elsey et al. 1990; Elsey et al. 1994; Smith and Marais 1994). It is important to establish appropriate and safe stocking densities in the enclosures (Pooley 1990).
Individuals should also be housed according to similar size class to reduce bullying or fighting as a result of territoriality (Pooley 1990). Wild crocodilians will be far less adapted to captive conditions. It is recommended that they be stocked at lower densities and provided with more space than captive-reared individuals (Lang 1987a; Hutton and Webb 1994).
The total area of a basic enclosure is 15 square meters and an accompanying pond should be approximately 7 m long and 10 m wide (Ziegler 2001). However, the area should be based on either the adult size of the intended species or the maximum size of the individual that will be housed in that particular enclosure (Ziegler 2001). The minimum area required to house a single crocodile is twice the length of the average adult of this species, in both width and depth (Ziegler 2001). For example, C. acutus has an average adult size of 4 m and would therefore require an enclosure at least 8 m wide by 8 m deep for a total minimum area of 64 square meters. The enclosure size must increase for each additional animal that will be housed. As a general rule, area should be increased 30% (20% more water and 10% more land) per additional animal (Ziegler 2001). To house one adult male and two adult females, the above enclosure example would increase to a minimum size of approximately 10 x 10 m (100 square meters). Remember that the examples given are minimum size requirements. The actual number of animals that can be safely housed in any one enclosure can vary greatly depending on many factors, some of which will be discussed in the following sections.
Physical Construction
There are several suitable options for the enclosure's outer fence. Consider benefits and costs of each, along with the overall appearance of the enclosure, when deciding what materials to use. Fences can be 5.08-cm chain link fencing (at least 11.5-gauge or stronger), poured cement, cement blocks, or brick (Ziegler 2001). Chain link is usually the most affordable option, but it is often best to use a combination of materials for different sides (Ziegler 2001). For example, solid cement or block wall may be used at the back of an enclosure while chain link is used at the viewing areas (Figure 4). Another excellent option for relatively small enclosures is to build a short block wall with chain link extending above to provide easy viewing and improved safety (Ziegler 2001) (Figure 7). Using chain link with a green vinyl coating, if available, can be more attractive.

Credit: Matthew Brien, 2006
Regardless of materials used, the fence of the enclosure must be properly constructed to prevent escape. This requires extension above and below ground, as crocodiles can be excellent diggers and climbers (Pooley 1990). Fence height should be equal to the total length of the largest animal being housed, or have a turn-back at the top to force a climbing animal to fall (Ziegler 2001).
The best option, in terms of cost and aesthetics with large species such as C. acutus, is to utilize a vertical fence with an inward-facing turn at the top (Ziegler 2001). Independent of height or material, any fence will need to be buried to prevent animals from digging out (Pooley 1990). This is especially important for both C. acutus and C. moreletti, as both species are recognized diggers. C. acutus are known to dig into mud banks up to 9 m (Webb and Manolis 1989). Another consideration is to ensure effective drainage within the enclosure (Ziegler 2001). This is particularly important if the facility is located in an area that is subjected to seasonal precipitation. It is also necessary to ensure that, if the enclosure does become flooded, the animals cannot swim out or become trapped and drown.
Fence Materials and Design
We recommend that the fence extend beneath the ground level in an L shape, both vertically (1.0 m) and horizontally (0.75 m) (Ziegler 2001). The most basic and least expensive design recommends including heavy (10-gauge) chain link, rising 2 m above ground with the final 50-60 cm making a 45° turn back into the enclosure (Ziegler 2001) (Figure 8). This fencing should also extend 1 m below ground, turn 90°, and continue 0.75 m into the enclosure (Ziegler 2001). Fence posts/poles should be sturdy enough to support the weight of a climbing animal (7.8 cm, or 3 in. outer diameter) and pole spacing should be no more than 3-4 m apart (Ziegler 2001). At least every other pole should be set in cement. Further strength and support for the fence can be accomplished by using horizontal pipe (4.3 cm) connecting the vertical poles. This should be done where the turn-back starts near the top of the fence (the construction contractor can make final design recommendations depending on the base materials you choose) (Ziegler 2001).
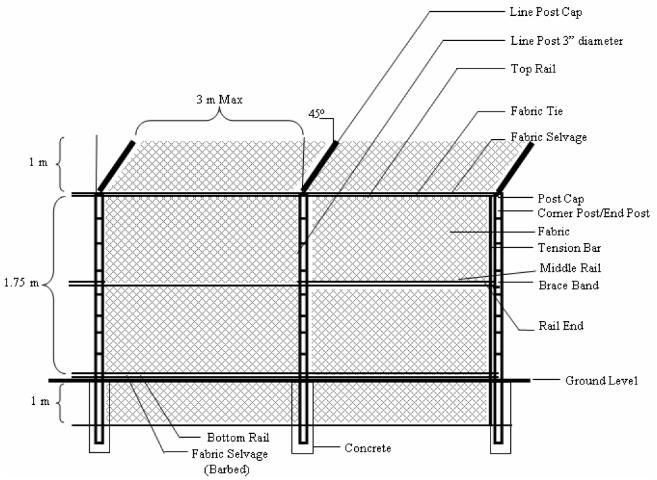
Credit: Ziegler, 2001
It is advisable to include 8-gauge or heavier tension wire at ground level (Ziegler 2001). If the ground is hard enough that animals cannot dig deeply, it will not be necessary to have the buried fence material extend into the enclosure's interior. Some facilities prefer to extend the fencing further below the ground and eliminate the turn-back.
Gates
Keeper safety requires at least one gate for any enclosure housing large crocodiles (Ziegler 2001). Gates must open inward to prevent animals escaping by pushing out against the door (Ziegler 2001). Locks should be sturdy and accessible from inside and outside of the enclosure.
Gates should be placed well away from the water's edge, be accessible, be lockable, and be large enough to safely move animals, keepers, and equipment through them (QLD 1997; Ziegler 2001). Gates must also incorporate the same escape-proof features as the outer fence, with a turn-back at the top, and either cement (75 cm deep and 20 cm wide) or chain link footer extending below ground (Ziegler 2001). The gate will be the weakest point in your enclosure, so supports should be set in cement.
Pool Design and Construction
The design of the water feature within the enclosure must be carefully considered, since feeding, thermoregulation, and breeding and other social interactions in crocodilians are all focused on the aquatic environment (Lang 1987a). The shape, size, and clarity of the pool will determine the level of safety for keepers, the ease of maintenance, and the quality of public viewing. The water area should cover 50% of the total enclosure, leaving enough dry land area for all specimens to haul out while at least half the available dry land remains unoccupied (Vernon 1994; Ziegler 2001) (Figure 9). Keep in mind that landscape features such as logs, tree clumps, or other impassable vegetation reduce the amount of land area available for use by the animals, and enclosure size should be adjusted to compensate for this (Britton 2001; Ziegler 2001).

Credit: Matthew Brien, 2006
While natural ponds (or engineered excavations below the water table) are sometimes incorporated into captive enclosures, they present many challenges to the facility. Locating and monitoring the health of individuals kept in earthen ponds can be difficult due to irregular depths, lack of water clarity, and crocodiles excavating tunnels in the banks (which may lead to escape) (Child 1987; Pooley 1990). Large natural enclosures are commonly used for adults and may be quite suitable as long as water quality is maintained (Figure 2). Natural ponds are not normally recommended for raising stock due to the difficulty in maintaining a clean, safe, and sanitary environment (Child 1987; Pooley 1990). If outbreaks of disease occur, earthen ponds have proven almost impossible to treat (Webb et al. 1987). A shallow (1 m) poured, concrete pool, with either an integrated drain system or mechanical pump for changing water, is generally a better long-term choice for crocodile grow-out enclosures (Pooley 1990; Britton 2001) (Figure 10). For smaller individuals and sub-adult groups, it is often best to use two or more cement ponds in an enclosure (Hutton and Jaarsveldt 1987; Pooley 1990). This will enable each pond to be cleaned alternately to avoid displacing all crocodiles from the water each time, thus reducing stress (Hutton and Jaarsveldt 1987; Pooley 1990). They can then also be cleaned more thoroughly (Child 1987). The pond(s) should typically be around 10-15 m long by 1.5 m wide and 60-100 cm deep (Ziegler 2001).

Credit: Matthew Brien, 2006
The drain system requires a greater initial investment but can be linked to other ponds at the facility and will allow for easy cleaning of pools (or water level adjustment) by simply opening a valve outside the enclosure (Ziegler 2001). The drain must be located at the bottom of the pool, with the pool floor sloping towards it to ensure that feces and uneaten food that collect will be removed (Pooley 1990). Water from all pools can then be routed away from the enclosures and public areas to a proper treatment and disposal site (Ziegler 2001). The second option is a portable mechanical pump (gas or electric) that can be moved between enclosures and that uses hoses to pump the water from the pond to a treatment site (Ziegler 2001). Pool shape greatly influences the number and size of animals that can be housed together, the safety of keepers, and the aesthetics of the enclosure. Creating breaks in the line of sight between areas of the pool creates visual isolation between animals and helps to reduce stress and aggression (Lang 1987a; Morpurgo 1992). Irregular pool shapes can best create these "visual barriers" in addition to physical barriers (land and vegetation) (Lang 1987a; Morpurgo 1992) (Figure 9 and Figure 11). Although U- or V-shaped pools are recommended over circular or rectangular pools, S- or Z-shaped pools can usually accommodate more animals with less conflict (and often less total area per animal) (Ziegler 2001). If circular or rectangular pools must be used, barriers should be incorporated in the form of partially submerged logs, boulders, or islands, which will create some degree of separation between animals (Vernon 1994; Ziegler 2001) (Figure 11). More complicated shapes usually increase construction costs.

Credit: Matthew Brien, 2006
However, they may provide greater economy in terms of space requirements for individuals and total number of enclosures required to meet all housing needs (Ziegler 2001). Creating multiple haul-out areas (gradual slopes where animals can easily enter and exit the water) around the pool also limits unnecessary interaction with enclosure mates (Ziegler 2001) (Figure 9). Slope of haul-outs should be between 10–20o. Steeper grades often cause animals to slip backwards, damaging their feet and bellies (Pooley 1990; Ziegler 2001). Crocodiles are very susceptible to repeated abrasions of the feet and ventral surface which can lead to infection and, if untreated, death (Pooley 1990). The finished texture of concrete used to construct pools must be very smooth (Pooley 1990). This can be achieved with either a fiberglass or paint finish, which will also aid in cleaning (Pooley 1990; Britton 2001). Particular attention should be paid to the shallow areas of the pool (haul-outs), where crocodiles will be expected to enter and exit the water. Extending the concrete approximately 1 m or more around the entire edge of the pool will reduce debris entering the pool and maintain a safe obstruction-free zone for maintenance personnel (Suvanakorn and Youngprapakorn 1987) (Figure 9).
Water depth only needs to be enough for an adult animal to fully submerge (1 m is plenty for even the largest crocodiles) (Hutton and Webb 1994; Ziegler 2001). There should be sufficient area of deep water for all animals in the enclosure to be submerged at the same time (Ziegler 2001). If the enclosure is to be used for breeding it should incorporate a large area of deeper water (> 1 m) to promote successfully this behavior (Lang 1987a). Deeper pools may be used but have several disadvantages. Deep pools are more difficult to keep clean, make uneaten food harder to recover, and require more water at scheduled drain and refill intervals (Hutton and Webb 1994; Ziegler 2001). They can also present a safety hazard by allowing crocodiles to approach keepers unseen, although this can be reduced by constructing deeper areas farther from the edge (QLD 1997; Ziegler 2001).
Landscape Design
Landscape design can be simple or complex as long as it meets the basic needs of crocodiles. These needs include well-drained ground substrate, areas of sun and shade for thermoregulation, and barriers to allow visual and physical separation between enclosure mates (Ziegler 2001). Landscape design should also be created with keeper safety in mind. Areas near the water should be clear in order to allow keepers to maintain visual contact with the animals. There should also be several easily accessible escape routes available (Ziegler 2001). Generally it is suggested that furnishings be avoided. If they are to be used in exhibits, they should be secured to avoid their being displaced and potentially causing injury to the animals (Ziegler 2001).
Suitable options for ground cover include grass or fine sand (Britton 2001; Ziegler 2001) (Figure 7 and Figure 12). These substrates will drain well and provide a non-abrasive surface for the crocodiles. Many types of soil provide adequate results as long as they drain and dry quickly (Britton 2001). Animals kept in a constantly damp, poorly drained environment are subject to a high possibility of bacterial infection (Ziegler 2001). Substrates such as rock and woodchips can cause intestinal blockage if ingested and provide a breeding ground for bacteria (Ziegler 2001). Enclosures can be sparsely or densely planted so long as adequate shade is provided for all animals (Britton 2001) (Figure 7 and Figure 12).

Credit: Matthew Brien, 2006
A more densely planted ground area will help to reduce aggression by creating physical and visual barriers between individuals, but requires more dry land (Ziegler 2001). Be sure that plant species used in the enclosure are non-toxic. Consider animal size when planning your landscape. Woody plants and tall sturdy grasses tend to be hardier and better withstand repeated abuse by large crocodilians (Britton 2001; Ziegler 2001). Species of short clumping palms make excellent landscape barriers and are sturdy and aesthetically pleasing. Enclosures housing sub-adults can often be much more ornate than those intended for mature specimens (Britton 2001). If sparse landscaping is desired, large logs and boulders can be used to create necessary barriers, and plywood shelters to provide shade (Ziegler 2001). Exhibits intended for public observation should have plantings oriented perpendicular to the viewing areas to provide a quality viewing experience (Ziegler 2001).
Staff and General Public Safety
All the guidelines presented in this paper, particularly regarding construction, have been designed to achieve a high level of safety and minimize risk of negative humancrocodile interactions within a facility. However, crocodiles are large, unpredictable, fast, powerfully strong predators, so it is the responsibility of the facility to ensure that all staff are appropriately trained (QLD 1997). Caution and safe practices should always be executed and reinforced with staff to prevent laxness. If the facility offers an exhibition section, inevitably children will be present at times. Staff should always be vigilant about monitoring visitor behavior and ensuring rules regarding safety are strictly adhered to (Table 1 and Figure 13). It is also essential that the construction of the "exhibit" enclosure ensure that visitors can not come in to direct contact with the crocodiles (QLD 1997). This can be assured by including an extra "safety" barrier or fence around the actual fence of the enclosure (Whitaker and Andrews 1998).

Credit: Matthew Brien, 2006
Water Quality and Maintenance
General Maintenance
Maintenance requirements will vary greatly depending on the overall size and landscape design of the enclosure, size and number of crocodiles housed, water quality desired, and the chosen substrate for ground cover. It will also be important to balance frequency of cleaning with minimizing stress to individuals (Pooley 1990; Hutton and Webb 1994). Land area of enclosures should be kept dry and free of loose debris, feces, and uneaten food (Pooley 1990). Also, land areas should be scrubbed occasionally with dilute disinfectant (Hutton and Jaarsveldt 1987; Hutton and Webb 1994). However, the most important aspect of cleaning will be associated with the water and pond area, as this is where most bacteria and disease generally occur (Hutton and Webb 1994).
Water Quality
As enclosures typically contain a water body encompassing 50% of the total area, it is crucial that good water quality and optimum temperature are achieved (Vernon 1994; Britton 2001). Water used in the enclosure should be fresh water (Hutton and Webb 1994; Britton 2001). If the water is to be chlorinated, it should be maintained at very low levels (2–3 ppm). Any uneaten food should be removed from the water daily to avoid dangerous bacterial levels (Pooley 1990). To maintain high water quality, it is recommended that pools be drained, rinsed, and refilled with clean water on a regular basis to ensure good water clarity and to prevent disease (Elsey et al. 1994; Hutton and Webb 1994). The frequency of cleaning and water changes will be dependent upon pool volume, crocodile density, size of animals, and other variables, such as food type and environmental conditions. Dilute disinfectant should also be used to scrub out the pond bases on occasion (Hutton and Webb 1994). However, it is crucial that all disinfectant is thoroughly washed out before crocodiles are allowed to return to the water (Hutton and Webb 1994). While cleaner water is never detrimental and better visibility increases keeper safety and visitor viewing enjoyment, cost and availability of clean water may be prohibitive to frequent change intervals (Pooley 1990). Algal growth usually affects water clarity well before bacterial conditions become dangerous for crocodiles, and some facilities change water as infrequently as every 2–3 months (Ziegler 2001). If cleaning of the water occurs too frequently, it may also be counterproductive by causing undue stress through keeper presence and rapid changes in water temperature (Smith and Marais 1994). Consider the priorities of your facility and experiment with different full and partial water change schedules to achieve the desired results.
Heating and Thermoregulation Requirements
Ensuring that constant, warm optimum temperatures are maintained within the enclosure is essential to the health and growth of crocodiles (David 1994; Smith and Marais 1994). Fluctuating temperatures ultimately result in decreased growth, high mortality, and instances of disease (David 1994). If dramatic shifts in temperature occur in the area, such as those associated with frosts and cold fronts, constantly heated water or an indoor retreat with heat need to be provided (Whitaker 1987). As individuals may seek refuge within the water, temperatures should always be stable to counter fluctuating air temperatures (Hutton and Webb 1994; Vernon 1994). It is therefore necessary that a heating element is installed within the water (Hutton and Webb 1994; Vernon 1994). Optimal growth in crocodilians has been found to occur around 31°C, with appetites and effective digestion diminishing below 29°C (Coulson and Hernandez 1964; Coulson and Coulson 1986). Ensuring an appropriate thermal gradient within the enclosure will also enable individuals to thermoregulate effectively (Lang 1987b; Smith and Marais 1994). This can be achieved by positioning the enclosure so that it maximizes natural heating and cooling processes. As certain times of the day will become either too hot or cold, heating sources and adequate shade should also be provided (Hutton and Jaarsveldt 1987) (Figure 7 and Figure 12).
Diet and Feeding
Providing individuals with enough food to ensure optimal growth is important, but overfeeding should be avoided as it can lead to obesity and associated disease (Pooley 1990). Growth rates of crocodilians are rapid during the first few months of life and require feeding at least 6-7 times a week (Whitaker and Andrews 1998). Once individuals reach sub-adult size they only need to consume 8-10% of their body weight a week (Whitaker and Andrews 1998). However, size, body condition, species, age, sexual maturity, breeding condition, and season can all influence individual requirements. Determining the optimum rate of feeding for individuals in a facility requires some trial and error (Pooley 1990; Whitaker and Andrews 1998). Generally, crocodilians will increase their level of consumption during warmer months of the year, while decreasing it during the cooler months (Pooley 1990).
Despite accounts of different species in the wild feeding on carrion and "storing" food until it is rancid, crocodilians prefer fresh food (Webb and Manolis 1989). Crocodilians also fare better when fed a diet that is high in protein, as research suggests that crocodilians utilize protein over fat as an energy source (Hutton and Webb 1994; Smith and Marais 1994). If the food source does not already include high levels of calcium, in the form of bones, it is important that a calcium supplement be included (Hutton and Webb 1994). Crocodilian skin contains bones known as osteoderms. Providing calcium-rich food not only eliminates bone disorders and abnormalities but can improve the quality of skins (Webb and Manolis 1989; Pooley 1990). Vitamin supplements should also be provided to further boost immune systems and minimize mortality (Hutton and Webb 1994; Smith and Marais 1994). It is believed that providing individuals with the appropriate amounts of amino acids, vitamins, and minerals can significantly reduce the detrimental effects of stress in captive crocodilians (Smith and Marais 1994). This is important as, despite the best husbandry and management practices, crocodilians will still inevitably suffer certain levels of stress under captive conditions (Elsey et al. 1994). It is recommended that food be presented in bite-sized portions to reduce the mess caused by individuals when feeding (Webb and Manolis 1989; Pooley 1990). If meat is to be frozen, it should be fresh and sealed in plastic. Meat should never be refrozen once thawed, as this will result in high levels of bacteria (Pooley 1990). Red meat is commonly the preferred food source for crocodilians (Elsey et al. 1994; Hutton and Webb 1994). However, minced meat and meat high in fat should be avoided as it will only foul the water which will create an environment for disease (Lang 1987a; Elsey et al. 1994; Hutton and Webb 1994). Fresh or fresh-frozen chicken heads, carcasses, and fish are also other options. In the wild, fish constitute the main prey item of American crocodiles (Britton 2001). Regardless, it is important to provide crocodilians with a varied diet for optimum results (Pooley 1990). Feeding captive crocodilians a mono-diet of fish has been correlated with an inability to produce fertile eggs in Morelet's crocodiles (Hunt 1980).
Any savings achieved as a result of providing lower quality food will ultimately be offset by negative implications on the crocodiles (Pooley 1990; Elsey et al. 1994). A poor diet will lead to decreased fertility, stunted growth, bone and teeth disorders, and reduced immunity to disease (Pooley 1990). It is especially important to strictly adhere to a high protein and calcium-rich diet for hatchlings and juveniles less than one year, as they are highly susceptible to disease and mortality (Hutton and Jaarsveldt 1987; Webb and Manolis 1989).
Captive Breeding and Rearing of Crocodilians
The ability of individuals to mate successfully and produce viable offspring in captivity presents an important indicator as to how effectively husbandry and management practices are being employed within a facility (Elsey et al. 1994). If practices are inadequate, they will be demonstrated by infertility and failed hatching (Elsey et al. 1994). It is imperative that the standard of husbandry practices be at its highest in the breeding, incubation, and hatchling sections of a facility housing crocodilians.
Breeding
As with most aspects of housing crocodilians, successfully breeding individuals can be difficult at best with a number of variables that need to be taken into consideration. As a general rule, males should always be larger than females, and the male-to-female ratio of individuals in an enclosure skewed heavily towards females (Child 1987; Lang 1987a). One-to-one sex ratios may be preferred, especially in highly aggressive, territorial species and for individuals introduced from the wild (Joanen and McNease 1971; Hutton and Webb 1994). However, this ratio is not always economically or practically feasible. Therefore, other stocking options of one male to several females in a several hectare pond can also be employed. Optimal stocking rates will be ultimately dependent upon size and species (Elsey et al. 1990; Elsey et al. 1994).
As both C. acutus and C. moreletti demonstrate a high degree of tolerance for conspecifics in captivity, they may be stocked at higher densities. If physical barriers and an irregular-shaped pond are provided (see Pool Design and Construction), more individuals can be stocked in a smaller area and bred with considerable success (Lang 1987a; Morpurgo 1992). This has been demonstrated for species such as the Nile crocodile (Crocodylus niloticus), in which all males were known to establish a small territory and successfully mate when housed in an irregular-shaped pond (Morpurgo 1992). Smaller females that usually have their reproductive activity suppressed by larger, more mature, females have also been found to breed and nest more successfully in such an enclosure (Morpurgo 1992).
It is strongly recommended that breeding stock be obtained from captive bred animals if possible. They will tolerate captive conditions and breed more successfully and at an earlier age (Joanen and McNease 1971; Lang 1987a; Hutton and Webb 1994). Female and male crocodiles will generally take between 4-15 years to reach maturity, or at between 2–3 m in length depending on the species and quality of husbandry (Lang 1987a). Maturity is effectively size dependent and can be achieved at earlier ages if growth and development is increased under captive conditions (Lang 1987a). The number of eggs in a clutch and the fertility of the embryos will generally increase with age and maturity (Lang 1987a). A decrease in fertility and clutch size will occur at about 20 years of age, depending on the species (Lang 1987a). Female C. acutus become sexually mature at 2–2.25 m in length and have a courtship period that can last up to 2–3 months (Kushlan and Mazzotti 1989). C. moreletti reach sexual maturity at 7–8 years of age, or 1.5 m in length (Platt and Thorbjarnarson 2000).
Nesting
Adequate areas for nesting should be provided to ensure that every reproductively mature female can access a site, with each site appropriately spaced (Lang 1987a). This will reduce fighting over nest sites and during nesting. The sites of nests can be manipulated by strategically placing nesting materials within the enclosure (Lang 1987a).
American crocodiles are extremely adaptable in their nesting strategy, and while they are mainly recognized as hole-nesters, individuals will readily build mound nests if suitable materials are provided (Kushlan and Mazzotti 1989; Thorbjarnarson 1992). Morelet's crocodiles are predominantly mound-nesters (Thorbjarnarson 1992). The females of both species have been known to lay eggs within the same nests as conspecifics, suggesting a more gregarious and tolerant demeanor (Kushlan and Mazzotti 1989; Britton 2001).
It is important to select nesting sites that are open, easily accessible, and away from deep water. Once eggs have been laid, they will need to be gathered for artificial incubation (Hutton and Webb 1994). Collecting eggs from nests is very hazardous, especially involving species in which the females are highly protective (QLD 1997). This process requires people to gather the eggs, protect the gatherers, and keep lookout or oversee the whole situation to prevent an accident (QLD 1997). It should always involve a coordinated effort from an experienced team of at least 2-3 people, with more people advisable depending on the situation (QLD 1997).
There have been a wide range of nest defense behaviors noted for C. acutus, from non-existent to highly aggressive (Groombridge 1987; Kushlan and Mazzotti 1989; Thorbjarnarson 1992). Females of this species have also been known to abandon nests entirely if disturbed (Kushlan and Mazzotti 1989). C. moreletti on the other hand will aggressively defend their nests against any intruders with males even participating in the protection of young (Rainwater et al. 2000). Therefore, great care should be exercised when removing eggs from nests of species that are protective.
Incubation
Ideally, crocodilian eggs should be placed in an artificial, climatically controlled incubation chamber with a thermostat (Hutton and Webb 1994; Ojeda et al. 1998) (Figure 14). Once eggs are collected, the top orientation of the egg in the nest should be marked and placed in the incubator in approximately the same position to prevent embryo mortality (Hutton and Webb 1994; Ojeda et al. 1998). Eggs should be cleaned with fresh water and possibly disinfected with an antifungal treatment before being placed in the incubator (Hibberd 1994). This has been found to increase fertility by up to 80–85% for C. moreletti in a farm situation (Ojeda et al. 1998). Handling of eggs should be kept to a minimum, and initial washing should be done through irrigation rather than by hand (Hibberd 1994). The recommended substrate is either vermiculite or sand that should be kept moist, but not wet, to avoid both desiccation and saturation of the eggs (Hibberd 1994; Hutton and Webb 1994). However, eggs do not require a substrate if precisely controlled environmental conditions can be achieved (Webb and Manolis 1989).

Credit: Matthew Brien, 2006
For C. acutus, clutch sizes tend to be between 30–60 eggs (Thorbjarnarson 1989) while C. moreletti lay around 20–40 eggs (Thorbjarnarson 1992). Incubation times are generally between 80–90 days for both species, but this is essentially temperature-dependent (Britton 2001). Incubation times for C. moreletti have been achieved at around 69 days in captivity at higher temperatures (Ojeda et al. 1998).
From a captive study in Mexico, C. moreletti appear to incubate successfully at a temperature of 32°C, with a relative humidity of greater than 90%, and a high gas exchange (Ojeda et al. 1998). This is similar for most other species of crocodilians, with recommended incubation temperatures at between 31–33°C, almost 100% relative humidity, and good air flow, ideally bubbled through water (Webb and Manolis 1989; Ojeda et al. 1998). It is crucial that temperatures remain constant or at least within a few degrees (31–33°C). Large fluctuations outside of the normal range (>34°C and <29°C) can result in abnormalities or mortality (Webb and Manolis 1989; Hutton and Webb 1994).
Hatchlings and Grow Out
Crocodilians are most vulnerable and susceptible to disease, stress, and mortality in the first year of life (Hutton and Jaarsveldt 1987; Webb and Manolis 1989). In the wild, survivorship can be typically as low as 1% (Mayer et al. 1998). Although survivorship of hatchlings in captive-bred situations is significantly higher, it has still been found to be as low as 70% for species such as C. porosus, even when provided with ideal conditions (Mayer et al. 1998).
Hatchlings generally emerge at a length of 12–30 cm depending on the species (Lang 1987a; Webb and Manolis 1989). They should be placed in a darkened rearing enclosure with shallow, heated water at between 30–33°C, with the option to increase their body temperature to 35°C (Webb and Manolis 1989; Smith and Marais 1994) (Figure 10 and Figure 15). Temperatures above 36°C or below 28°C have been found to significantly increase stress levels and mortality in hatchlings and should be avoided (Turton 1994). Tanks can be concrete or plastic with simple and effective drainage and a constant warm temperature (Elsey et al. 1994; Mayer et al. 1998). Hide boards can also be used to provide cover and help reduce stress.

Credit: Matthew Brien, 2006
As with all other size classes the temptation to stock hatchlings at high densities should be avoided (Pooley 1990). Optimal stocking rates can be achieved through trial and error. Individuals should also be segregated according to their respective size class (Pooley 1990). It is recommended that temperature fluctuations, loud noise, and changes in diet be avoided (Webb and Manolis 1989; Thuok 1998). They should be fed strictly a fresh, high quality diet (see Feeding and Diet). As a general rule, feeding should occur 6–7 days a week, and then reduced to 3–4 days a week after 3–4 months. After one year they should be fed every other day. It is important that the handling of hatchlings be kept to an absolute minimum, and that the design of the tanks enables regular and effective cleaning with minimal disturbance (Huchzermeyer 2002).
Recommended Readings
Crocodilian Husbandry
Britton, A. 2001. "Crocodilian Captive Care F.A.Q." http://crocodilian.com/crocfaq/
Morelets Crocodile (Crocodylus moreletti)
Alvarez del Toro, M. 1974. Los Crocodylia de Mexico. Instituto Mexicano de Recursos Naturales Renovables.
Hunt, H. 1975. "Maternal behavior in the Morelet's crocodile, Crocodylus moreletii." Copeia 1975: 763–764.
Hunt, H. 1977. "Aggressive behavior by adult Morelet's crocodiles, Crocodylus moreletii towards young." Herpetologica 33: 195–201.
Perez-Higareda, G. 1980. "Notes on nesting of Crocodylus moreletii in Southern Veracruz, Mexico." Bulletin Maryland Herpetological Society 16:52–53.
Platt, S. 1994. "Crocodylus moreletii and Crocodylus acutus in Belize." Crocodile Specialist Group Newsletter 13(4): 15–16.
American Crocodile (Crocodylus acutus)
Campbell, H. W. 1972. "Ecological or physiological interpretations of crocodilian nesting habits." Nature 238: 404–405.
Departamento de Vida Silvestre. 1993. Estudio y Protecion del Cocodrilo Americano (Crocodylus acutus) en la Republica Dominicana. Secretaria de Estado de Agricultura, Santo Domingo, Dominican Republic, 209.
Kushlan, J. A. and F. J. Mazzotti. 1989. "Historic and present distribution of the American crocodile in Florida." Journal of Herpetology 23(1): 1–7.
Kushlan, J. A. and F. J. Mazzotti. 1989. "Population biology of the American crocodile." Journal of Herpetology 23(1): 7–21.
Medem, F. 1981. Los Crocodylia de Sur America, Vol. 1, Los Crocodylia de Colombia. Bogota, Colciencias, 354.
Moler, P. and C. Abercrombie. 1992. "Growth and survival of Crocodylus acutus in south Florida, USA," In: Crocodiles. Proceedings of the 11th Working Meeting of the Crocodile Specialist Group. (Gland, Switzerland: IUCN), 14.
Schubert, A. 1994. "Conservation of American crocodile." Crocodile Specialist Group Newsletter 13(3): 14.
Thorbjarnarson, J. 1989. "Ecology of the American crocodile, Crocodylus acutus." In: Crocodiles. Their ecology, management and conservation. A Special Publication of the Crocodile Specialist Group. (Gland, Switzerland: IUCN), 228–258.
Literature Cited
Bolton, M. 1990. "The role of crocodile ranching in rural development." In: Crocodiles. Proceedings of the 9th Working Meeting of the Crocodile Specialist Group, Vol. 1, (Gland, Switzerland: IUCN), 89–97.
Brazaitis, P. 1973. "The identification of living crocodilians." Zoologica 58: 59–101.
Britton, A. 2001. "Crocodilian Captive Care F.A.Q." http://crocodilian.com/crocfaq/
Child, G. 1987. "The management of crocodiles in Zimbabwe." In: Wildlife Management: Crocodiles and Alligators. Edited by G. J. W. Webb, S. C. Manolis, and P. J. Whitehead, (Sydney: Surrey Beatty and Sons), 50–61.
Coulson, R.A, and T. D. Coulson. 1986. "Effect of temperature on the rates of digestion, amino acid absorption and assimilation of the alligator." Comparative Biochemistry Physiology 83A (3): 585–588.
Coulson, R.A. and T. Hernandez. 1964. "Biochemistry of the alligator. A study of metabolism in slow motion." Louisiana State University Press, Baton Rouge. pp. 2–33. In : Smith, G. A. and Marais, J. 1994. Stress in Crocodilians – The impact of nutrition.
Cox, J. H. and M. M. Rahman. 1994. "An assessment of crocodile resource potential in Bangladesh." In: Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 1.
David, D. N. 1994. "Harvesting wild crocodilians: Guidelines for developing a sustainable use program." pp. 274–292. In: Crocodiles. Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 1.
Davis, G. W. 1994. "Crocodile conservation: The benefits of farming and ranching." pp. 1–6. In: Crocodiles. Proceedings of the 2nd Regional (Eastern Asia, Oceania, Australasia) Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland.
Elsey, R. M., L. Joanen and L. McNease. 1990. "Stress and plasma corticosterone levels in the American alligator-relationships with stocking density and nesting success." Comparative Biochemistry Physiology 95A (1): 55-63.
Elsey, R. M., T. Joanen and L. McNease. 1994. "Captive breeding of alligators and other crocodilians." pp. 1–27. In: Crocodiles. Proceedings of the 2nd Regional (Eastern Asia, Oceania, Australasia) meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland.
Groombridge, B. 1987. "The distribution and status of world crocodilians." pp. 9–21. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Hibberd E. M. 1994. "Fungal disease in eggs and hatchlings of farmed Crocodylus porosus." pp. 39–47. In: Crocodiles. Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Hines, T. C. and C. L. Abercrombie. 1987. "The management of alligators in Florida, USA." pp. 44–46. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Hines, T. C. 2002. "Ecuador ranching project: Why did it fail?" pp. 19-22. In: Crocodiles. Proceedings of the 16th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Hollands, M. 1987. "The management of crocodiles in Papua New Guinea." pp. 75–87. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Huchzermeyer, F. W. 2002. pp. 173–175. In: Crocodiles. Proceedings of the 16th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Hunt, R. H. 1980. "Propagation of Morelets crocodile." pp. 161–165. In: Reproductive Biology and Diseases of Captive Reptiles. J. B. Murphy and J. T. Collins, Eds. Meseraull Printing Inc. Lawrence, Kansas, U.S.A.
Hutton, J. M., and K. R. V. Jaarsveldt. 1987. "Crocodile farming and ranching in Zimbabwe." pp. 323–327. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Hutton, J. M., and G. J. W. Webb. 1994. "The principles of farming crocodiles." pp. 1–38. In: Crocodiles. Proceedings of the 2nd Regional (Eastern Asia, Oceania, Australasia) meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland.
Joanen, T. and L. McNease. 1971. "Propagation of the American alligator in captivity." Proceedings of the 25th Annual Conference Southeast Association. Game and Fish Commission. 25: 106-116.
Joanen, T. and L. McNease. 1987. "Alligator farming research in Louisiana, USA." pp. 329–334. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Kelly, H. R. 1994. "A comparison between the breeding results of captive and wild Nile crocodiles and the conservation merits of closed cycle breeding in South Africa." pp. 182–186. In: Crocodiles. Proceedings of the 12th Working meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 1.
Kushlan, J. A. and F. J. Mazzotti. 1989. Population biology of the American crocodile. Journal of Herpetology 23: 7–21.
Lance, V. A. 1994. "An overview of stress in farmed crocodilians." p. 1. In: Crocodiles. Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Lang, J. W. 1987a. "Crocodilian behavior: Implications for management." pp. 273–94. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Lang, J. W. 1987b. "Crocodilian thermal selection." pp. 301–317. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Lever, J. 1994. "Captive breeding and conservation. Allies or enemies?" pp. 162-166. In: Crocodiles. Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 1.
Magnusson, W. E. 1984. "Economics, developing countries, and the captive propagation of crocodilians." Wildlife Sociological Bulletin 12: 194–197.
Mayer, R., S. Peucker, B. Davis and H. Stephenson. 1998. "Environmental conditions for rearing Crocodylus porosus on farms." pp. 326–332. In: Crocodiles. Proceedings of the 14th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Mazzotti, F. J. 1987. "Developing a management plan for Morelet's crocodile in the Usamacinta and Grijalva Delta, Tabasco, Mexico." pp. 569–574. Trans. Del Symp. Inter. Sobre la Ecol. Y Cons. Del Deltas Usamacinta y Grijalva, Tobasco, Mexico. INIREB, Tobasco, Mexico.
Mkanda, F. X. 1992. Ranching the Nile crocodile Crocodylus niloticus in Malawi. pp. 1–13. In: Crocodiles. Proceedings of the 11th Working Meeting of the Crocodile Specialist Group of the Species Survival Commission of the IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Morpurgo, B. 1992. "Pond structure: The relation to hierarchy and reproductive activity in male captive Nile crocodiles." p.17. In: Crocodiles. Proceedings of the 11th Working Meeting of the Crocodile Specialist Group of the Species Survival Commission of the IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Ojeda, F. J. L., R. Arredondo and M. C. R. Montijo. 1998. "Artificial incubation of eggs of Crocodylus moreletii under captive conditions." pp. 347–351. In: Crocodiles. Proceedings of the 14th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Platt, S. G. and J. B. Thorbjarnarson. 2000. "Nesting ecology of the American crocodile in the coastal zone of Belize." Copeia. 2000: 868–872.
Pooley, T. 1990. "Basic crocodile farming/ranching methods in remote areas." pp. 123–165. In: Crocodiles. Proceedings of the 10th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Queensland. "Division of Work place Health and Safety. 1997." pp. 1-28. Guide for the Crocodile Industry. http://www.detir.qld.gov.au/hs/guide/gde20.pdf.
Rainwater, T. R., S. T. McMurry and S.G. Platt. 2000. "Crocodylus moreletti (Morelet's crocodile) behavior." Herpetological Review. 31: 237.
Smith, G. A. and J. Marais. 1994. "Stress in crocodilians – The impact of nutrition." pp. 2–33. In: Crocodiles. Proceedings of the 12th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland. Volume 2.
Suvanakorn, P. and C.Youngprapakorn. 1987. "Crocodile farming in Thailand." pp. 341–343. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Thorbjarnarson, J. 1989. "Ecology of the American crocodile, Crocodylus acutus." pp. 228–258. In: Crocodiles. Their ecology, management and conservation. A Special Publication of the Crocodile Specialist Group. IUCN, Gland, Switzerland.
Thorbjarnarson, J. B. 1992. Crocodiles: An action plan for their conservation. pp. 90–135. Compiled by IUCN/SSC Crocodile Specialist Group. IUCN, Gland, Sitzerland.
Thuok, N. 1998. "Current status of crocodile in Cambodia in captivity and in the wild." pp. 141-150. In: Crocodiles. Proceedings of the 14th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Turton, J. 1994. "Preliminary report on stress in farmed Crocodylus porosus hatchlings." In: Proceedings of the 2nd Regional Meeting of the IUCN/SSC Crocodile Specialist Group. Darwin, 1994.
Vernon, B. P. 1994. "The economics of crocodile farming." In: Crocodiles. Proceedings of the 2nd Regional (Eastern Asia, Oceania, Australasia) meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland.
Webb, G. J. W. and S.C. Manolis. 1989. Australian Crocodiles: A Natural History. New Holland publishers: Sydney.
Webb, G. J. W., P. J. Whitehead and S. C. Manolis. 1987. "The management of crocodiles in the Northern Territory of Australia." pp. 115–121. In: Wildlife Management: Crocodiles and Alligators. Surrey Beatty and Sons: Sydney.
Whitaker, R. 1987. "Management of crocodilians in India." pp. 63–72. In: Wildlife Management: Crocodiles and Alligators. G. J. W. Webb, S. C. Manolis and P. J. Whitehead, Eds. Surrey Beatty and Sons: Sydney.
Whitaker, N. and H. Andrews. 1998. "Madras croc bank: an update." pp. 402–406. In: Crocodiles. Proceedings of the 14th Working Meeting of the Crocodile Specialist Group, IUCN – The World Conservation Union, Gland, Switzerland and Cambridge UK.
Zeigler, W. F. 2001. "Crocodilian exhibit design and general husbandry parameters." pp. 1–22. In: Crocodilian Biology and Captive management. American Zoo and Aquarium Association: Silver Spring, MD.