Introduction
Global trade and travel transport plants and animals from native ranges to new ecosystems. About 10–20% of nonnative (exotic, alien) species that arrive in new locales become invasive, meaning they are likely to harm the environment, economy, or public health. Preventing the introduction of invasive species is the most effective way to protect native biodiversity and ecosystem integrity. Once an invader begins to establish and spread, its control costs increase rapidly (Figure 1).
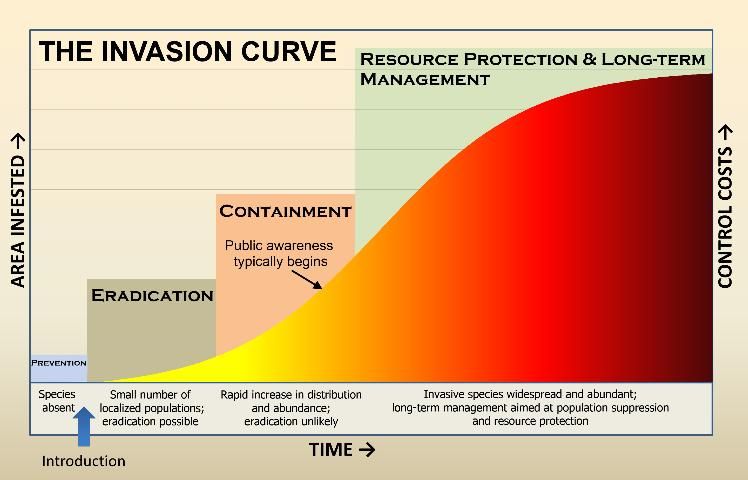
Credit: Adapted from Invasive Plants and Animals Policy Framework, State of Victoria, Department of Primary Industries, 2010
The United States and most US states lack adequate regulation and implementation of laws for cross-border species trade (Jenkins et al. 2007). Despite some progress in recent years, most nonnative species still enter the country without sufficient screening.Florida ports are the entry points for about half of the reptiles, arachnids, insects, and crustaceans imported into the United States (Romagosa 2011). These arrivals, coupled with the state's hospitable climate and habitats, have made Florida home to more invasive species than any other state but Hawaii. While it is too late to prevent the invasion of Burmese pythons and Argentine black and white tegus (Figure 2), action is needed to prevent other potentially destructive species from establishing (Figure 3).

Credit: Thomas A. Rahill; Liz Barraco, Florida Fish and Wildlife Conservation Commission

Credit: © 2007 Wolfgang Wüster; © 2006 Henk Wallays
Ecological risk assessment estimates the probability of an ecological event occurring and evaluates subsequent consequences. For invasive species, a risk assessment addresses the questions "How likely is a species to become invasive?" and "What can go wrong if it does become invasive?" Scientists explore a species' characteristics to determine its potential to invade new areas and cause negative impacts.
Risk assessment can be applied at various stages of the invasion process, most notably prevention and eradication (Figure 1). In the prevention phase, risk assessment is essential to develop screening procedures and regulate importation. After species are introduced, risk assessment remains critical to identify priority species for early detection and rapid response (EDRR). This fact sheet focuses on the development of risk screening tools for both prevention and EDRR.
How to Assess Invasion Risk
There are three different approaches to assess a species' invasiveness:
- Qualitative risk assessment uses professional judgments to assign species to categories based on biological characteristics and climate information. Experts assign a numerical value for each parameter, then total the scores to categorize species as low, medium, or high risk.
- Quantitative risk assessment uses multivariate statistical analyses and/or model simulations to predict species spread and associated effects. Techniques may include, for example, discriminant analysis, decision trees, population modeling, and niche modeling.
- Semi-quantitative risk assessment uses both a scoring system and statistical analyses. This combined approach may be the most accurate way to capture a species' dynamic interaction with its environment.
No single factor adequately predicts a species' introduction and establishment, and factors vary across taxa and environments. Therefore, all approaches must consider multiple biological and ecological parameters (Table 1). Researchers also should acknowledge any uncertainties in the data and consider the implications of uncertainties for risk estimates (Bartell and Nair 2003).
Risk Assessment Examples
Weed Risk Assessment
One of the earliest ecological risk assessments focused on the introduction of plants into Australia and New Zealand. A 49-item questionnaire was developed to assess a species' biological attributes, its weed status in other locations, and its climate and environmental preferences. This tool was validated against local experts' "weediness" scores for 370 Australian taxa (Pheloung et al. 1999) and has had an impact on plant imports. The Weed Risk Assessment system (WRA) has been validated for use in Florida and elsewhere in the United States (e.g., Gordon et al. 2011).
The UF/IFAS Assessment of Non-native Plants in Florida's Natural Areas (assessment.ifas.ufl.edu) uses the WRA to evaluate invasion risk of introduced and proposed nonnative plant species in Florida.

Credit: R. R. Smith, USDA
Zebra Mussel
Research on zebra mussels (Dreissena polymorpha) in the Great Lakes of the United States highlighted invasion pathways, identified other high-risk species and vulnerable areas, and recommended preventative measures (Kolar and Lodge 2002). Impacts were assessed using quantitative models based on regression analysis of data from multiple invaded sites (Ricciardi 2003). This pioneering work has led to development of risk-assessment models for other taxa.

Credit: D. Jude, University of Michigan
Cane Toad
Establishment risk of the cane toad (Rhinella marina, formerly Bufo marinus) in Australia was quantitatively assessed using taxonomic scores, climate match scores, history of successful introductions, and geographic range sizes. This tool identified the cane toad as an extremely risky species. Had it been applied prior to the cane toad's introduction, impacts of this invasive species could have been avoided (Bomford et al. 2005).

Credit: USGS
Arabian Camel
Camels (Camelus dromedarius) were assessed prior to their introduction into New Zealand for a tourism venture. A quantitative risk assessment of exotic vertebrates considered propagule pressure, climate match, history of establishment elsewhere, and taxonomic group (Bomford 2008). Camels were deemed unlikely to establish wild populations but posed risks to native vegetation, human culture, and health. The analysis prevented mass importation of camels. Only a small number of camels have since been imported with special permits and regulations.

Credit: Norbert Nagel
Invasiveness Screening Kits for Florida
The Weed Risk Assessment (WRA) of Australia and New Zealand provided a foundation for many invasive species risk assessments used today (Pheloung et al. 1999). The Fish Invasiveness Screening Kit (FISK) applies the WRA's peer-reviewed approach to assess species based on their biogeography, ecology, history, and potential invasiveness. FISK initially focused on freshwater fish in the British Isles and other temperate-zone countries and has been modified for use in Florida and other warm regions (Lawson et al. 2013; Figure 5). The FISK tool has been used to support regulatory decisions in Florida (Hill and Lawson 2015). The kit has also been adapted for amphibians (AmphISK), marine fish (MFISK), and marine and freshwater invertebrates (MI-ISK and FI-ISK).

Credit: Melanie L. J. Stiassny; Iris Stern
Literature Cited
Bartell, S.M., and S.K. Nair. 2003. "Establishment risks for invasive species." Risk Analysis 24(4): 833–845.
Bomford, M. 2008. Risk assessment models for establishment of exotic vertebrates in Australia and New Zealand. Invasive Animals Cooperative Research Centre. Canberra, Australian Commonwealth Territory. 198pp.
Bomford, M., F. Kraus, M. Braysher, L. Walter, and L. Brown. 2005. Risk assessment model for the import and keeping of exotic reptiles and amphibians. Bureau of Rural Sciences. 110pp.
Fujisaki, I., K.M. Hart, F.J. Mazzotti, K.G. Rice, S. Snow, and M. Rochford. 2009. "Risk assessment of potential invasiveness of exotic reptiles imported to south Florida." Biological Invasions 12(8): 2585–2596.
Gordon, D.R., K.J. Tancig, D.A. Onderdonk, and C.A. Gantz. 2011. "Assessing the invasive potential of biofuel species proposed for Florida and the United States using the Australian Weed Risk Assessment." Biomass and Bioenergy 35(1): 74–79.
Hill, J.E., and K.M. Lawson. 2015. "Risk screening of Arapaima, a new species proposed for aquaculture in Florida." North American Journal of Fisheries Management. 35(5): 885–894.
Jenkins, P.T., K. Genovese, and H. Ruffler. 2007. Broken screens: the regulation of live animal imports in the United States. Defenders of Wildlife, Washington, D.C. 56pp.
Kolar, C.S., and D.M. Lodge. 2002. "Ecological predictions and risk assessment for alien fishes in North America." Science 298: 1233–1236.
Lawson, L.L., Jr, J.E. Hill, L. Vilizzi, S. Hardin, and G.H. Copp. 2013. "Revisions of the Fish Invasiveness Screening Kit (FISK) for its application in warmer climatic zones, with particular reference to peninsular Florida." Risk Analysis 33:1414–1431.
Pheloung, P.C., P.A. Williams, and S.R. Halloy. 1999. "A weed risk assessment model for use as a biosecurity tool evaluating plant introductions." J. Environ. Manag. 57: 239–251.
Reed, R.N., J.D. Willson, G.H. Rodda, and M.E. Dorcas. 2012. "Ecological correlates of invasion impact for Burmese pythons in Florida." Integrative Zoology 7(3): 254-270.
Ricciardi, A. 2003. "Predicting the impacts of an introduced species from its invasion history: an empirical approach applied to Zebra Mussel invasions." Freshwater Biology 48: 972-981.
Romagosa, C.M. 2011. "A summary of live animal importation by the United States." http://www.evergladescisma.org/SummaryofUSliveanimalimports.pdf.
For University of Florida's complete report summarizing invasive species risk assessments, including references, see http://crocdoc.ifas.ufl.edu/publications/reports/riskassessment.pdf.