The following description of citrus fertilizer uptake, soil and leaf testing, and nutrient recommendations was taken from EDIS publication SL253, Nutrition of Florida Citrus Trees, 3rd Edition, edited by Kelly T. Morgan and Davie M. Kadyampakeni.
Fertilizer, Nutrition Uptake, and Yield Response
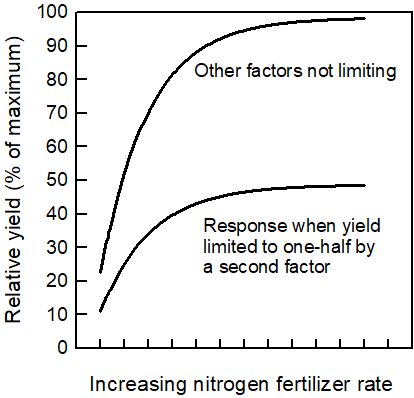
This section describes the typical citrus yield increase with added fertilizers. The increase in yield with increased fertilizer rates is called the yield response curve. The shape of this curve is similar for a range of crops and conditions (Figure 1).
Credit: UF/IFAS
Fertilizer nitrogen (N) is used in this example, but the nature of the response curve is similar for other nutrients. At very low N rates, there is a large increase in yield with each added unit of N. As yield increases, each additional unit of N results in a smaller increase in yield. This smaller response to increasing the fertilizer amount is also referred to as the law of diminishing returns. The two response curves in Figure 1 compare the effect of N rate for two situations: one where the amount of fertilizer nutrients in the soil limits or reduces yield, and another where the yield is limited to one-half by low concentrations of a second fertilizer nutrient. The shapes of the curves are similar, and the rate of N where the slope levels off is only slightly higher for the more productive grove.
The amount of nutrient the crop removes varies from a fraction of a lb/acre for some of the micronutrients to as much as 100 lb/acre of N or K from a high-producing grove. The following numbers were determined for healthy trees prior to HLB entering the state. Micronutrient requirements for HLB-affected citrus trees have been studied, and changes in recommendations for these nutrients are being considered. Research on other nutrients is ongoing and will be provided as studies are completed. For oranges, approximately 0.12 lb N/box is removed with the harvest. Therefore, crop removal ranges from 12 lb N/acre for a 100 box/acre yield to around 100 lb N/acre for a grove producing 800 boxes/acre. Using this same rate of N removal would suggest that groves with reduced yields would be in the range of 24 to 36 lb N/acre. This number is too low for HLB-affected trees, and the rate of removal for HLB-affected trees is being determined.
Nutrient uptake from applied fertilizers is not 100% efficient—that is, not all the fertilizer applied is taken up by the tree, so more nutrients must be applied than the minimum required by the tree. N use efficiency, expressed as lb N removed by the crop divided by lb N applied, ranges from 0.2 to 0.4 in groves with low to moderate yield. For healthy citrus trees, N efficiencies around 0.5 have been observed in groves with a good production record. Application of 200 lb N/acre supplies sufficient N for an 800 box/acre orange yield when N use efficiency is 0.5.
Effect of HLB on Nutrient Management
Nutrient uptake efficiency of HLB-affected trees may be at the low range of nitrogen (N) use efficiency because of root loss (up to 80% depending on HLB severity). Therefore, an HLB-affected orange grove picking 300 boxes per acre and requiring 12 lb of N per 100 boxes would need only 120 lb of N based on lower uptake efficiency of 30%. The UF/IFAS annual recommended range of N applications is 120 to 160 lb per acre for grapefruit and 125 to 240 lb per acre for mature sweet orange trees based on average fruit yield from healthy trees. Lower yields of HLB-affected trees would indicate that current rates should be 120 to 150 lb per acre for oranges. However, recent research has indicated that N requirement for HLB-affected trees should be in the range of 160 to 220 lb per acre because of reduced N use efficiency from root density reductions. Rates of N for grapefruit should be 120 to 160 lb of N per acre per year. Similar amounts of potassium (K) to those for nitrogen would also be required. These experimental results also indicate that frequent multiple applications of these nutrients are preferred instead of fewer applications as recommended for healthy citrus trees prior to HLB.
Leaf Nutrient Analysis
Leaf analysis is a useful tool to detect problems and adjust fertilizer programs for citrus trees because leaf nutrient concentrations are the most accurate indicators of sufficient nutrition of fruit crops. Leaves reflect nutrients taken up from the soil and redistributed throughout the plant, so the deficiency or excess of an element in the soil is often reflected in the leaf analysis. Nutrient deficiency or excess will cause citrus trees to grow poorly and produce lower yields and/or fruit quality. Determining potential nutritional problems should be a routine citrus-growing practice. Quantifying nutrients in trees or soils with leaf and soil analysis eliminates guesswork in adjusting a fertilizer program.
Leaf analysis should include N, phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulfur (S), manganese (Mn), zinc (Zn), copper (Cu), iron (Fe), and boron (B). Chlorine (Cl) concentration is sufficient under most field conditions, but Cl may become excessive where soil or irrigation water is saline. Similarly, molybdenum (Mo) deficiency or toxicity is rare. The goal in tissue analysis is to adjust fertilization programs such that nutritional problems and their costly consequences from higher costs and lower yields are prevented.
Leaf analysis integrates all the factors that might influence nutrient availability and uptake. It shows the relationship of nutrients to each other. For example, potassium deficiency may result from a lack of K in the soil or from excessive Ca, Mg, or sodium (Na). Similarly, adding N when K is low may result in K deficiency because the increased growth requires more K.
Tissue Analysis
- Determines if the soil is sufficiently supplying the essential nutrients.
- Confirms nutritional deficiencies, toxicities, or imbalances.
- Identifies “hidden” toxicities and deficiencies when visible symptoms do not appear.
- Evaluates the effectiveness of fertilizer programs.
- Provides a way to compare several fertilizer treatments.
- Determines the availability of elements not tested for by other methods.
- Helps in determining interactions between nutrients.
Steps in Leaf Analysis
Citrus trees affected by HLB are typically low to optimum for many nutrients, but sampling guidelines should be followed precisely to ensure that analytical results are meaningful.
Procedures for proper sampling, preparation, and analysis of leaves have been standardized to achieve meaningful comparisons and interpretations. If done correctly, the reliability of the chemical analysis, data interpretation, fertilization recommendations, and adjustment of fertilizer programs will be sound. Therefore, considerable care should be taken from the time leaves are selected for sampling to the time they are received at the laboratory for analysis.
Leaf Sample Timing
- Leaf samples must be taken at the correct time of year because nutrient concentrations within leaves continuously change. As leaves age from spring through fall, N, P, and K concentrations decrease while Ca and Mg increase. However, leaf mineral concentrations are relatively stable from four to six months after emergence in the spring.
- The best time to collect spring flush leaves of this age is July and August. If leaves are sampled later in the season, summer leaf growth can easily be confused with spring growth.
Leaf Sampling Technique
- A sampled citrus grove block or management unit should be no larger than 20 acres. The sampler should ensure that the leaves taken represent the sampled block.
- Each leaf sample should consist of about 100 leaves taken from nonfruiting twigs of 15 to 20 uniform trees of the same variety and rootstock and under the same fertilizer program.
- Use clean paper bags to store the sample. Label them with an identification number that can be referenced when the analytical results are received.
- Avoid immature leaves due to their rapidly changing composition.
- Do not sample abnormal-appearing trees, trees at the edge of the block, or trees at the end of rows, because they may be coated with soil particles and dust or have other problems.
- Do not include diseased, insect-damaged, or dead leaves in a sample.
- Select only one leaf from a shoot and remove it with its petiole (leaf stem).
- Leaves should be washed with soapy water and rinsed with distilled water within 24 hours of sampling.
Analysis and Interpretation
- The laboratory determines the total concentration of each nutrient in the leaf sample. Because total concentration is determined, there should be no difference in leaf analysis results between different laboratories.
- The laboratory usually interprets each result as deficient, low, optimum, high, or excessive, but the citrus grower can also interpret the results using UF/IFAS leaf analysis standards (Table 1). These standards are based on long-term field observations and experiments conducted in different countries with different citrus varieties, rootstocks, and management practices and are used to gauge citrus tree nutrition throughout the world.
- The goal in nutrition management is to maintain leaf nutrient concentrations within the optimum range every year. If the interpretation for a particular nutrient is not optimum, various strategies can be used to address the situation (Table 2).
Soil Nutrient Analysis
Soil analysis measures organic matter content, pH, and extractable nutrients, which are useful in formulating and improving a fertilization program. Soil analysis is particularly useful when conducted for several consecutive years so that trends can be observed.
Similar to leaf analysis, methods to determine the organic matter and soil pH are universal, so results should not differ between laboratories. However, soil nutrient extraction procedures vary from lab to lab. Several accepted chemical procedures exist that remove different amounts of nutrients from the soil because they vary in strength. To draw useful information from soil tests, consistency in the use of a single extraction procedure from year to year is important to avoid confusion when interpreting the amount of nutrients extracted.
A soil extraction procedure does not measure the total amount of nutrients present, nor does it measure the quantity actually available to citrus trees. A perfect extractant would remove nutrients from the soil in amounts that are exactly correlated with the amount available to the plant. Therefore, the utility of a soil testing procedure is how well the extractable values correlate with the amount of nutrients a plant can take up. The process of relating these two quantities is called calibration.
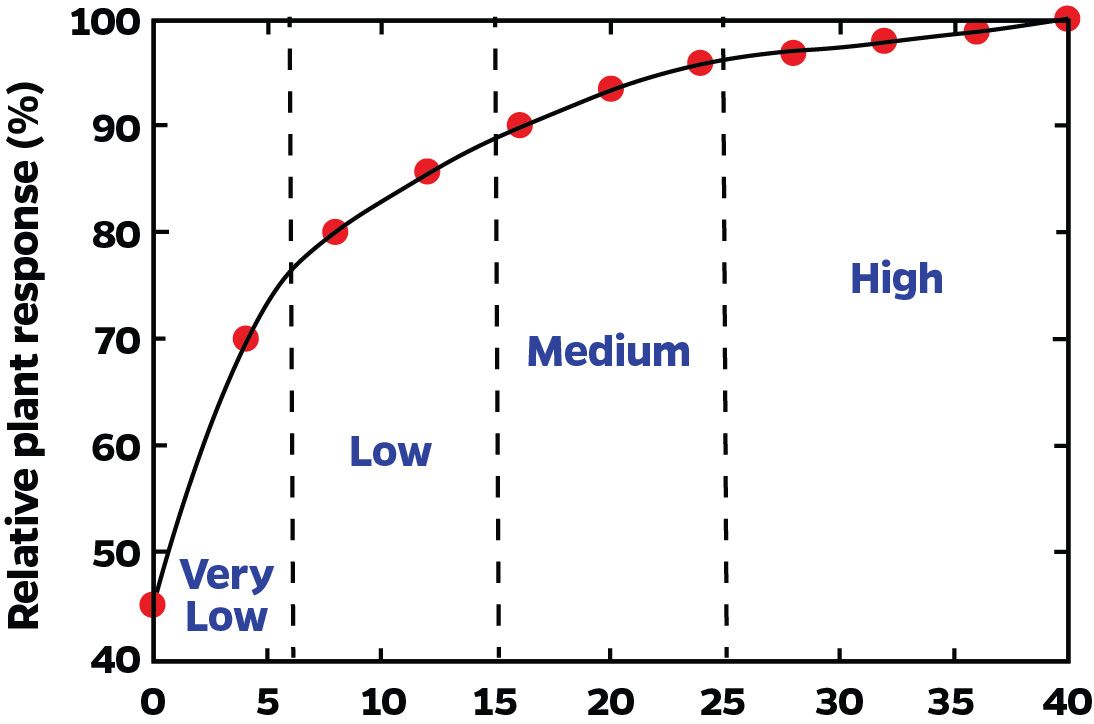
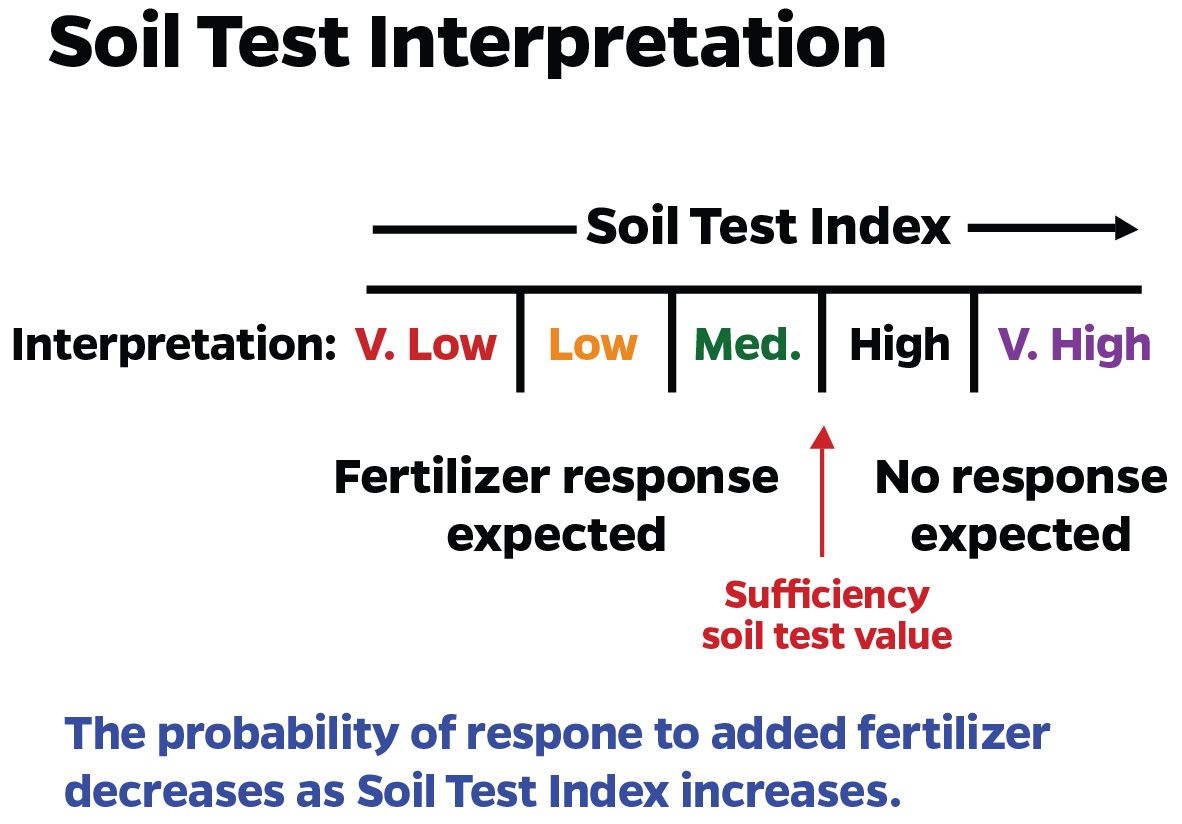
A soil test is only useful if it is calibrated with plant response. Calibration means that as a soil-test value increases, nutrient availability to plants increases in a predictable way (Figure 2). Low soil-test values imply that a crop will respond to fertilization with the particular nutrient. High soil-test values indicate the soil can supply all the plant needs, so no fertilization is required (Figure 3). Caution should be taken on some high soil-test values, such as Ca and P, where all extractable nutrients might not necessarily be available for plant uptake, and supplemental fertilizer might be required.
Credit: UF/IFAS
Credit: UF/IFAS
In Florida, soil testing for mobile, readily leached elements like N and K has no value. In addition to organic matter and pH, soil testing is important for P, Mg, Ca, and Cu. The UF/IFAS soil-test interpretations for P, K, and Mg using Mehlich-3 extraction were established from experiments conducted for many years (Table 3).
The single most useful soil test in a citrus grove is for pH. Soil pH greatly influences nutrient availability. Some nutrient deficiencies can be avoided by maintaining soil pH between 5.5 and 6.5. Deficiencies or excesses (toxicities) are more likely when the pH is outside this range.
In some cases, soil tests can determine the best way to correcting a deficiency identified by leaf analysis. For example, Mg deficiency may result from low soil pH or excessively high soil Ca. Dolomitic lime applications are advised if the pH is too low, but magnesium sulfate is preferred if soil Ca is very high and the soil pH is in the desirable range. If soil Ca is excessive and soil pH is relatively high, then foliar application of magnesium nitrate is recommended. Many citrus soils have become elevated in pH because irrigation water is high in Ca and carbonates from limestone aquifers. In this case, irrigation water acidification or application of soil-acidifying fertilizers is recommended. Soil-acidifying fertilizers are those formulations containing ammonium N sources or sulfur. The use of organic acid can also be considered as the organic acid can help in lowering the soil pH as well as conditions the soil and improves nutrient uptake. Recent studies have shown that organic acids such as humic and fulvic acid can improve the growth and yield of citrus trees.
Steps in Soil Sampling
Standard procedures for sampling, preparing, and analyzing soil should be followed for meaningful interpretations of the test results and accurate recommendations.
Soil Sample Timing
- In Florida, if soil samples are collected once per year, the best time is at the end of the summer rainy season and prior to fall fertilization (September and October).
- Annual soil samples should be taken at the same time as leaf samples to save time and reduce cost.
Soil Sampling Technique
- The accuracy of soil-test interpretations depends on how well the soil sample represents the grove block or management unit sampled.
- Each soil sample should consist of one soil core taken about 8 inches deep at the dripline of 15 to 20 trees within the area wetted by the irrigation system in the zone of maximum root activity.
- Areas sampled should correspond to the grove blocks where leaf samples were taken. They should contain similar soil types with trees of roughly uniform size and vigor.
- Thoroughly mix the cores in a nonmetal bucket to form a composite sample. Take a subsample from this mixture and place it into a labeled paper bag.
Preparation for Analysis
- Soil samples should be dried in the oven before shipping to the laboratory for analysis.
Analysis and Interpretation
- The basic soil analysis package run by most agricultural laboratories includes soil pH and extractable P, K, Ca, and Mg. Organic matter is sometimes also part of the package, or it may be a separate analysis. Extractable Cu is normally determined upon request.
- The laboratory interprets each soil test result as very low, low, medium, high, or very high and may also provide fertilizer recommendations accordingly. A citrus grower should independently interpret the numerical results according to UF/IFAS guidelines based on the Mehlich-3 extractant used (Table 3).
- The interpretations should be used to make decisions regarding soil pH control or fertilizer application (Table 4).
Recommended Fertilizer Rates and Timing
Young Trees (1 to 3 Years after Planting)
Nitrogen
Recommended rates of N for the first three years a citrus tree is in a grove can be found in Table 5. A constant supply of N throughout the growing season is essential to achieve maximum tree growth and early fruit yield. It is recommended to apply controlled-release fertilizer or frequent fertigation (Table 5).
Phosphorus
If soil testing justifies P fertilizer application, test the soil again the following year and compare with Table 3 to determine if P fertilization can be decreased or omitted. A leaf tissue-testing program for P should begin at this time, comparing the results with the standards in Table 1.
Potassium
Apply K fertilizer at a K2O rate equal to the 1.25 times the N rate.
Calcium
If the soil pH is in the optimum range of 5.5 to 6.5, there is no need to apply Ca. If soil pH is below 5.5, the soil should be limed to pH 6.5, which will supply needed Ca. If soil pH is above 6.5, the soil will contain abundant Ca. At pH close to 8, Ca will form precipitates.
Nutrient Management
Applying fertilizer in several small doses increases fertilizer efficiency by maintaining more constant nutrient availability throughout the growing season and by reducing leaching if unexpected rain occurs (Table 4). A minimum of four to six applications of dry fertilizer is recommended. If fertigating, splitting fertigation into 10 to 30 applications per year is common and desirable. The cost of liquid injection during irrigation is relatively small, particularly if the injection can be automated. Two or three applications of controlled-release fertilizer are satisfactory for HLB-affected trees because nutrients are protected from leaching rains. Controlled-release formulations may be applied pre-plant, incorporated after planting, or broadcast to ensure uniform distribution of nutrients throughout the enlarging root zone of young trees.
Bearing Trees (4+ Years in Grove)
Nutrient management for bearing trees requires many of the same considerations important for nonbearing trees. Applications of dry soluble fertilizer (3–6) or fertigation (10–30) are suggested to supply a near-constant soil nutrient concentration for HLB-affected trees. Nitrogen continues to be the most important element for tree growth, fruit yield, and fruit quality, but others also have substantial effects on production and fruit quality. Removal of elements by harvesting the crop becomes significant but accounts for only part of the fertilizer requirement.
Nitrogen
Recommended N fertilizer rates (Table 6) provide enough N for canopy expansion towards containment size while producing maximum economic yields of high-quality fruit. The chosen N rate will depend on soil characteristics, yield potential, and tree needs as indicated by leaf analysis interpretation (Table 1).
- For grapefruit, the recommended annual N rate is 120 to 160 lb/acre.
- For oranges and other varieties, the recommended annual N rate is 120 to 200 lb/acre.
Mature Bearing Trees (8+ Years in Grove)
Once trees reach containment size, further canopy growth is not desired, so nutrition inputs can be stabilized. Nitrogen fertilizer management should focus on replacing N exported with the harvested crop plus that needed to maintain tree vegetative growth. The guidelines for annual N fertilizer rates accounts for the needs of both vegetative growth and crop removal (Table 6).
- For grapefruit, the recommended annual N rate is 120 to 160 lb/acre. The chosen N rate will depend on soil characteristics, desired fresh-fruit quality characteristics, yield potential, and tree needs as indicated by leaf analysis interpretation (Table 1).
- For oranges, the annual N rate should fall within the range of 125 to 240 lb/acre. The recommended rate for a specific grove depends on either expected yield potential (for 8-to-11-year-old trees) or 4-year running average production history (for trees 12 years and older) expressed as either fruit yield or soluble solids production. When basing N fertilization on expected yield potential, the rate should be chosen considering (1) how well the young, bearing trees have produced and (2) leaf tissue analysis. If leaf N is maintained in the optimum range, additional fertilizer likely will not produce additional fruit and may reduce quality.
Leaching Rain Rule. If more than 3 inches of rainfall accumulates within a 72-hour period after an N fertilizer application, “replacement” fertilizer may be applied within one week up to one-half of the N rate used in the preceding application (not to exceed 30 lb/acre).
Phosphorus
Determine the need for P fertilization using leaf tissue and soil test results.
- Sample leaves and soil.
- Compare the analytical results with the interpretations provided in Tables 1 and 2.
- Follow the P fertilization guidelines in Table 7.
Potassium
Apply K fertilizer at a K2O rate equal to 1.25 the N rate. If leaf K is consistently below optimum, increase the K2O rate by 25%, especially if the grove soil is calcareous.
Calcium
The optimum soil pH range is 5.5 to 6.5. If soil pH is below 5.5, the soil should be limed to pH 6.5, which will supply needed Ca. If soil pH is above 6.5, the soil will contain abundant Ca. Recent research on HLB-affected citrus trees indicate that leaf Ca is typically deficient, therefore, IFAS changed its recommendation for Ca to 40 lb per acre per year.
Timing of Fertilizer Applications
In Florida, citrus trees should always be fertilized with split doses because there are numerous season-related nutrient requirements to satisfy growth, yield, fruit quality, and BMPs, which would be difficult to achieve with a single fertilizer application.
- The active growing season of perennial citrus trees spans six to nine months, and sandy soils have insufficient nutrient storage capacity to sustain a whole season of growth from a single application of fertilizer.
- Citrus trees have peak times for nutrient demand in the late winter and spring, and moderate to low nutrient demand in the summer, fall, and winter.
- The summer rainy season is the period of greatest risk for leaching nutrients from the soil profile.
- Split fertilizer applications provide some insurance against disastrous results from any single application error or the effects of natural disasters such as hurricanes.
Peak demand for nutrients by bearing citrus trees is during the period from bloom to the end of fruit growth phase 1 (cell division). In Florida, the physiological fruit drop during May/June marks the end of the cell division stage. The timing of the main bloom may vary by variety and year but typically occurs in March. During the weeks leading up to bloom, the trees must be primed with sufficient nutrients to be ready for the explosion of new growth. The nutrient priming can be achieved either through a fall fertilizer application during late September/October of the previous year, or with a prebloom fertilizer application in late winter (February). The disadvantage of a fall fertilizer application (after October) is that during the fruit maturation phase in the fall and winter, persistent high levels of nutrients (especially N) can inhibit fruit color break and suppress Brix levels at the intended harvest time. Incomplete color break and low Brix can reduce pack-out for fresh fruit, while delays in harvesting intended to improve fruit quality can increase the risk of preharvest fruit drop in HLB-endemic groves. Fall or winter fertilizer applications can also make psyllid control more difficult and increase the susceptibility of trees to freeze injury.
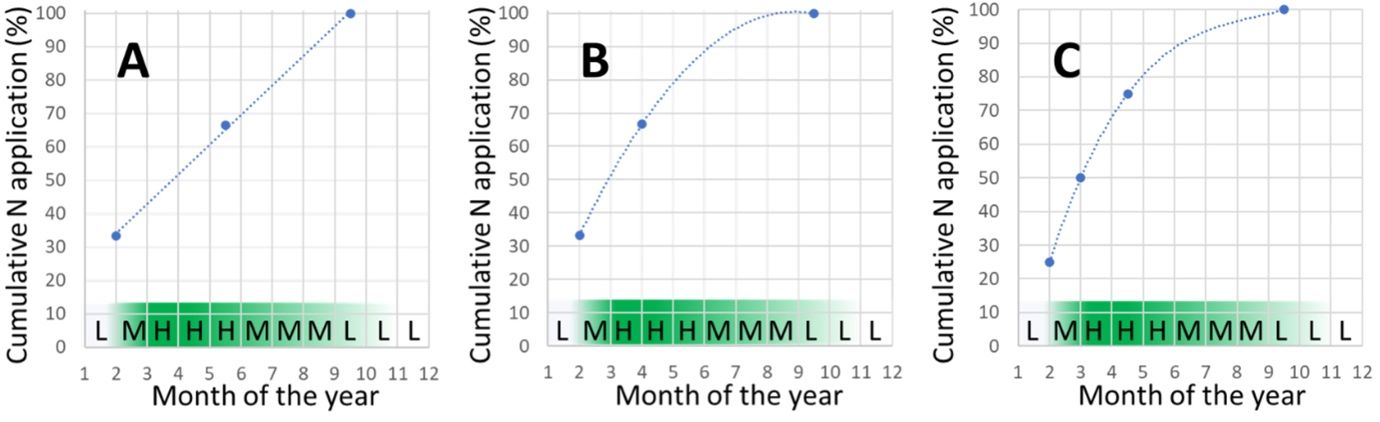
Split applications of dry soluble fertilizer are often made in equal increments for convenience (e.g., three splits of 33.3%, or four splits of 25%). Some fertilizer application equipment may not be able to calibrate correctly for very small fertilizer doses, and excessively numerous splits would be costly to apply, which is why typically three to four equal splits of dry fertilizers are recommended for Florida citrus. When the fertilizer is applied by fertigation, growers have much more flexibility for customizing the number and size of split applications. Figure 4A shows the incorrect method for applying three splits of fertilizer in a linear fashion using equal time increments, which mismatches with the peak nonlinear demand for nutrients by citrus in the spring. In situation (A), the trees would likely encounter nutrient deficits during April. A better method for splitting three doses of fertilizer is shown in Figure 4B, where two of the three fertilizer increments are strategically timed during the period of peak nutrient demand (bloom to the end of fruit growth phase 1). Similarly, the suggested strategy for applying four equal splits of fertilizer is shown in Figure 4C.
Credit:
Some trial and error with the timing of fertilizer splits may be required to obtain the best results, especially when targeting optimum color development for fresh fruit. In fresh-fruit production, knowledge of the expected harvest time (early-, mid-, or late-season varieties) is important to help determine the timing of the last fertilizer split for the growing season. For early varieties, the last split application of fertilizer N should be in early to midsummer, rather than late summer to early fall for the mid- to late-season varieties. Be aware that fertilizer applications made during the summer carry a higher risk of leaching events due to the higher probability of rain during the summer season in Florida. Leaf sampling will also provide guidance for fresh-fruit growers to target leaf tissue N in the high end of the optimal range (2.7%) during the bloom, fruit set, cell division, and cell enlargement stages, and in the low end of the optimal range (2.5%) or the low range (2.2% to 2.4%) during the fruit maturation stage in the fall and winter (see Table 1).
The optimum timing of K fertilizer splits is slightly different from that for N. In general, less K than N is required in the bloom and fruit-set period, while more K than N is required in the cell division, cell enlargement, and maturation stages. This is particularly important for fresh-fruit production because adequate K at the right time is important for proper fruit sizing. Examples of fertilizers that could be used to provide a suitable N:K2O ratio in the fertilizer blend at the correct times are (1) 15:3:11 applied prebloom, (2) 9:3:18 at the cell division stage, and (3) 12.5:3:18 at the cell enlargement stage.
Effect of Huanglongbing on Micronutrient Requirements
Leaf chlorosis develops as a result of infection with Candidatus Liberibacter asiaticus (CLas), including interveinal chlorosis of young leaves, similar in symptomology to Mn and Zn deficiencies that develop early in the growing season. Leaf chlorosis is followed by blotchy mottling of older leaves, which develops later in the growing season. Symptoms similar to those of nutrient deficiency develop in HLB-affected trees, including K, P, Mg, Ca, Mn, Zn, and Fe. HLB causes feeder roots to decline within a few months after infection and before foliar symptoms develop. Feeder roots are responsible for the bulk of nutrient uptake, and their decline likely explains the deficiency symptoms that develop in the canopy. Research has demonstrated that HLB symptoms can be reduced by foliar or ground applications of secondary macronutrients and micronutrients, especially Ca, Mg, Mn, and Zn. These responses have promoted the development and use of enhanced foliar nutritional programs in Florida. The efficacy of these programs has been a topic of considerable discussion and debate. Fertilization programs have varied considerably among growers and have consisted of various rates and application schedules of essential macro- and micronutrients.
Foliar nutrition applications are not likely to lead to past production levels in the short term. Research* has found that maintaining leaf concentration of essential nutrients increased canopy volume and occasionally yield. Application of the current UF/IFAS foliar recommendations (Table 8) three times per year following flushes in March, May, and September was found to maintain leaf concentrations in the optimum range with improved canopy density and yield. For example, the previous UF/IFAS recommendation for Mn and Zn was 5 lb metallic per acre per year. In 2022, UF/IFAS recommendations for Mn and Zn for HLB-affected citrus trees were changed to three applications for a total of 15 lb metallic per acre per year.
*Morgan, K. T., R. E. Rouse, and R. C. Ebel. 2016. "Foliar Applications of Essential Nutrients on Growth and Yield of ‘Valencia’ Sweet Orange Infected with Huanglongbing." HortScience 51 (12): 1482–1493. https://doi.org/10.21273/HORTSCI11026-16
Table 1. Guidelines for interpretation of leaf analysis based on 4-to-6-month-old spring-flush leaves from nonfruiting twigs of healthy trees.
Table 2. Adjusting a citrus fertilization program based on leaf tissue analysis.
Table 3. Interpretation of soil analysis data for citrus using the Mehlich-3 extractant.
Table 4. Adjusting a citrus fertilization program based on soil analysis.
Table 5. Recommended N rates and minimum number of applications for nonbearing citrus trees (1–3 years old).
Table 6. Recommended N rates and minimum number of applications for bearing citrus trees.
Table 7. Recommendations for P fertilization of bearing citrus trees based on leaf tissue and soil tests.
Table 8. Recommended methods, timing, and rates for micronutrient application to citrus groves.