The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction and History
Metamasius callizona (Chevrolat) has no accepted common name, but it has been referred to as the "Evil Weevil" by bromeliad enthusiasts throughout Florida, as a result of the destruction it has caused to native populations of bromeliads in the southern portion of the state. A member of the weevil family Curculionidae, it is one of three species of the genus Metamasius present in Florida. Metamasius mosieri Barber, the smallest of the three and native to Florida, Cuba, and the Dominican Republic, has been collected in Florida infrequently. Unlike Metamasius callizona, its presence does not threaten populations of native bromeliads in Florida's natural areas. The third species, Metamasius hemipterus sericeus (Olivier), was first reported in Florida in Miami-Dade County in 1984 and has since become an important pest of sugarcane, bananas, and ornamental palms.

Credit: H. Nadel, University of Florida
Metamasius callizona, which was first encountered in Florida in 1989 at a Ft. Lauderdale (Broward County) bromeliad nursery, is thought to have entered the state in a shipment of bromeliads imported from Veracruz, Mexico. The nursery was treated, but within two months, the weevil was found to be established in northern Broward County and southern Palm Beach County. By 1991, the weevil was present in four counties in southern Florida, and by 1999, it was found in 12 additional counties. As a direct result of attack by Metamasius callizona, the Florida Endangered Plant Advisory Council has added two species of bromeliads [Tillandsia utriculata (L.) and Tillandsia fasciculata Swartz] to the list of endangered species under the 1998 Florida Administrative Code. Additionally, the weevil has entered the Everglades region, home of Florida's rarest populations of bromeliads.

Credit: J.H. Frank, University of Florida
Description
Adults, which are 11 to 16 mm (0.4 to 0.6 in.) long, are black with a single yellow or orange band crossing the wing covers above the midpoint (Figure 3). The first and second ventral abdominal segments of males are indented and may display brownish coloration, while female abdominal segments are flat and always black.

Credit: J. L. Castner, University of Florida
Metamasius callizona adults are easily distinguished from both Metamasius mosieri (Figure 4), which is 6 to 9 mm (0.24 to 0.35 in.) long and red and black with two black spots on the upper, red portion of the wing covers, and from Metamasius hemipterus sericeus (Figure 5), which is 9 to 14 mm (0.35 to 0.55 in.) long and may be either red or orange, and black, with variation in color pattern.

Credit: P.M. Choate, University of Florida

Credit: Robin M. Giblin-Davis, University of Florida

Credit: J. Salas, FONAIAP
Metamasius callizona eggs are elongate, averaging approximately 2 mm (0.08 in.) in length and 1 mm (0.04 in.) in width. They are white when first oviposited, later turning yellow and finally light brown.
Larvae (Figure 7) are cream-colored with a dark head. Mean head capsule widths for laboratory reared first- through fifth-instar larvae were determined to be 0.92 mm (0.04 in.), 1.21 mm (0.05 in.), 1.69 mm (0.07 in.), 2.10 mm (0.08 in.), and 2.73 mm (0.11 in.), respectively. While the first three instars have discrete size classes, field-collected fourth- and fifth-instar larvae cannot be distinguished. Pupae are generally found within a fibrous cocoon (Figure 8) made of plant material.

Credit: J.L. Castner, University of Florida

Credit: B. Larson, University of Florida

Credit: B. Larson, University of Florida
Distribution and Host Plants
Metamasius callizona was described from Mexico and originally thought to range from Mexico to western Panama, although the report from Panama has been considered questionable. In Mexico, the weevil has been reported on Aechmea mexicana Baker, Catopsis sp., and Tillandsia limbata Schltdl. from Veracruz, on Vriesia sp. from Puebla, on Tillandsia utriculata and Tillandsia streptophylla Scheidweiler near Coatepec, on Tillandsia roland-gosselinii Mez near Pochutla, and in a grower's shade houses near Cordoba on Tillandsia heterophylla Morren, Tillandsia ehlersiana Rauh, Tillandsia streptophylla, and Tillandsia deppeana Steudel. Habitats in Mexico where the weevil has been found are often shaded and rainy, over 700 m in altitude.
Between 1973 and 1987, USDA-APHIS plant inspectors intercepted 14 weevils determined to be Metamasius callizona, being imported primarily on Tillandsia spp. All specimens of Metamasius callizona found in imported shipments have originated in Mexico. An additional 122 weevils identified only as Metamasius were intercepted during the same time period on bromeliads known to be attacked by Metamasius callizona.
The weevil is also reported from Guatemala (Alta Verapaz: San Juan). A survey of the Alta Verapaz region in June–August 2000 failed to yield Metamasius callizona, although weevils of related species, principally Metamasius quadrilineatus Champion, were found in bromeliads. Despite the early record of Metamasius callizona from Panama (Chiriquí: Potrerillos), the species was not detected during a survey in 1994. Furthermore, while specimens of many other species of Metamasius are present in the Smithsonian Tropical Research Institute in Panama, there are no Metamasius callizona specimens in the collection, leading to the suggestion that the weevil's natural range does not extend throughout Central America. However, Metamasius callizona was found in Belize in the fall of 2010 and again in 2014, infesting Tillandsia utriculata.
The first specimen of Metamasius callizona in Florida was found on a nursery-grown Tillandsia ionantha Planchon, a plant native to Mexico and Central America on which adults will feed but not lay eggs. In natural areas, the weevil primarily attacks Tillandsia utriculata, Tillandsia fasciculata, and Tillandsia paucifolia Baker. The weevil is most damaging to Tillandsia utriculata plants, killing them directly. While Tillandsia fasciculata appears to be more resistant, damage to flowering plants can be highly destructive to their populations. The weevil has also been observed attacking Tillandsia balbisiana Schultes (a species that had been listed already in Florida as threatened) and Tillandsia flexuosa Swartz (already listed as threatened), Tillandsia variabilis Schlechtendal (threatened), Tillandsia simulata Small (a species precinctive [=endemic] to Florida), as well as Catopsis and Guzmania spp. The state's native bromeliads that are likely to be attacked by Metamasius callizona if it reaches their more isolated habitats include Tillandsia pruinosa Swartz (endangered), Guzmania monostachia (L.) (endangered), Catopsis berteroniana (Schultes) (endangered), Catopsis floribunda (Brongniart) (endangered), and Catopsis nutans (Swartz) (endangered). The weevil, which appears to be restricted to plants large enough to supply an adequate amount of stem tissue for larval development, has been found most often in large species of Tillandsia. It will most likely not attack Florida's other native bromeliads [Tillandsia bartramii Elliott, Tillandsia setacea Swartz, Tillandsia recurvata (L.), and Tillandsia usneoides (L.)], which probably do not provide enough plant material for completion of larval development due to their small size. See http://entnemdept.ufl.edu/frank/bromeliadbiota/flbrom.htm.
Since its first appearance in Broward County, the weevil has spread through much of southern Florida by both natural dispersal and by movement of infested plants. As of December 2009, Metamasius callizona had been reported in the following 23 counties in Florida (Figure 10), given with date of first report: Broward (November 1989), Palm Beach (November 1989), Lee (October 1990), Miami-Dade (May 1991), Charlotte (August 1994), St. Lucie (November 1995), Glades (March 1996), Collier (March 1996), Sarasota (August 1996), Manatee (September 1996), Martin (May 1997), Hendry (August 1997), Brevard (November 1998), Indian River (December 1998), Desoto (March 1999), Highlands (March 1999), Polk (December 2000), Okeechobee (April 2002), Hillsborough (June 2003), Osceola (October 2003), Pinellas (July 2006), Orange (November 2006), and Volusia (January 2009).
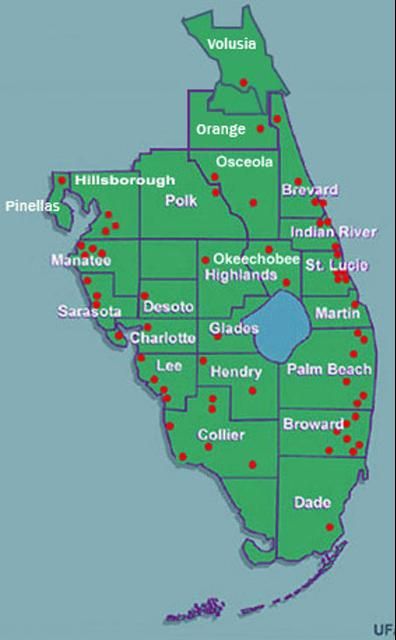
Credit: J. Salas, FONAIAP
In addition to attacking bromeliads in parks and other public lands, the weevil has infested private bromeliad collections and nurseries. Under greenhouse conditions, bromeliads mined and killed by Metamasius callizona have included Aechmea cvs., Ananas comosus (L.) (pineapple), Canistrum lindenii (Regel) Mez, Cryptanthus cvs., Dyckia sp., Fernseea bocainensis Pereira & Moutinho, Guzmania cvs., Hechtia sp., Hohenbergia sp., Neoregelia cvs., Neoregelia compacta (Mez), Nidularium cv., Orthophytum gurkenii Hutch., Pitcairnia angustifolia Solander, Quesnelia "testudo," Tillandsia fasciculata, Tillandsia paucifolia, Tillandsia utriculata, and Vriesia cvs.
Biology and Damage
When reared on a diet of pineapple stem, the weevil's development time from oviposition to adult emergence averaged approximately eight weeks under laboratory conditions (temperature of 26°C, relative humidity of 82%, photoperiod of 14:10 L:D). Mean duration of the egg stage was 8.3 days, while the larval stage averaged 36.4 days (5.0 days, 4.5 days, 4.4 days, 5.7 days, and 17.8 days for the first to fifth larval instars, respectively). The pupal stage lasted a mean of 11.8 days. Pre-ovipositional period of females averaged 28.9 days, and females lived an average 156.4 days in the laboratory. Minimal generational time in the field is estimated to be 10 weeks, with mean generational time projected to be 13 to 17 weeks. Larvae, pupae, and adults have been found throughout the year in southern Florida, suggesting continuous mating. Therefore, there are probably three to four continuous generations per year in Florida.
All life stages of the weevil may be present in the same plant. Adult females, which primarily feed on leaves but have been seen to feed on flowers, cut slits in host plant leaves close to their feeding sites, into which they lay eggs individually. Newly emerged larvae begin to consume leaf tissue as they move down to the base of the stem. They tunnel into the growing stem tissue, producing large holes in the base that may cause the plant to dislodge from its support structure on the tree. Larval damage is generally confined to the base of the plant (Figure 11) but can also reach up the flower stalk. Several weevils may successfully develop on the same host plant, provided there is sufficient plant tissue. However, larger larvae will attack smaller ones upon encountering them. The weevil usually pupates in the center of the plant's stem (Figure 12), within a cocoon it constructs from shredded plant material. When reared in planted pineapple tops, the weevil sometimes pupates in the soil near the base of the plant.

Credit: B. Larson, University of Florida

Credit: J.L. Castner, University of Florida
Damage is often accompanied by the production of a light brown, gelatinous material, which may be the plant's defensive reaction. This gel can be seen covering entrance holes to the weevil mines. Other symptoms of weevil damage include adult feeding marks on leaves, browning of leaves (Figures 13 and 14), and decomposition of the base of the central leaves, which can easily be pulled out when larval mining is substantial.

Credit: J.H. Frank, University of Florida

Credit: P.M. Choate, University of Florida
In ornamental bromeliad nurseries, the mining of Metamasius callizona larvae directly kills some species. Extent of damage to natural bromeliad populations in Florida varies. For example, heavy infestations of large, flowering plants have caused severe damage to populations of Tillandsia utriculata, which reproduce primarily by seed. Tillandsia fasciculata plants are able to produce offsets, which aids their populations to some extent in escaping decimation by the weevil. However, the absence of any natural enemies of the weevil in Florida has resulted in much greater destruction to bromeliad populations than that occurring in any part of the weevil's natural range.
Management
Where infestations of Metamasius callizona occur on ornamental bromeliads grown in nurseries, a reduced concentration of an insecticide labeled for beetle adults and grubs is recommended, applied as a spray or dip every two to three months to prevent infestations. This recommendation is based on general principles and not on specific efficacy data, because no chemical trials of any pesticide have been carried out against the weevil. See https://edis.ifas.ufl.edu/features/handbooks/MGHSouth.html.
In natural areas, where the pest is threatening native populations of bromeliads, chemical control is not feasible, in part because the epiphytic growth habit of the host plants makes them inaccessible from the ground. More importantly, many of the populations affected by the pest are found in state and county parks where use of chemical pesticides is not acceptable because of possible effects on non-target organisms.

Credit: J.H. Frank, University of Florida
Biological control offers the most likely success in management of the weevil. One potential biological control agent was discovered and studied at the Escuela Agrícola Panamericana in Zamorano, Honduras. This parasitoid tachinid fly, Lixadmontia franki (Figures 16 and 17), was found attacking the closely related weevil species Metamasius quadrilineatus in Honduran cloud forests, primarily in species of Tillandsia. The female fly larviposits at the base of leaves, near the entrance to the tunnel made by weevil larvae, and the fly larvae enter the tunnel to seek out the host. Fly larvae attack the larval stage of the weevil, and while parasitized weevil larvae may construct a pupation chamber, they die before pupating. Percent parasitism of weevils in field-collected bromeliads in Honduras has ranged from 5 to 67% and seems to increase during the summer rainy season to reach a maximum in October–November.

Credit: R. Cave, University of Florida

Credit: P.M. Choate, University of Florida
Lixadmontia franki flies brought into Florida and maintained in a quarantine facility were found to parasitize Metamasius callizona in bromeliad plants. The fly's natural host in Central America is Metamasius quadrilineatus, and it also will parasitize Metamasius mosieri, both of these being bromeliad-killing weevils. It would not attack Metamasius hemipterus sericeus. A strong fly colony was established in the quarantine facility, and Federal and State release permits were issued in 2007. Releases began at the end of June 2007 and are planned to be continued through 2008 in some Federal Preserves and State and county parks with concurrent evaluation.
The Florida Council of Bromeliad Societies (FCBS) has initiated a project to collect seeds of native bromeliads threatened by Metamasius callizona. Plants grown from those seeds will eventually be replaced in their natural setting if weevil populations are successfully reduced. For more information on the seed collection project and biological control efforts, see the http://entomology.ifas.ufl.edu/frank/savebromeliads/.
At least 20 other species of weevils are known to attack bromeliads in Central and South America. Several related species of weevils have been found in shipments of bromeliads from Neotropical countries, including Metamasius flavopictus (Champion) (Figure 18), Metamasius sellatus Champion (Figure 19), and Metamasius quadrilineatus (Figure 20). Therefore, in addition to managing populations of Metamasius callizona already present in the state, it is necessary to prevent the accidental importation of additional weevil pests. There are two important preventive tactics. Dipping imported plants in insecticides labeled for beetles both before leaving the country of export and after arriving in Florida reduces the risk, although the effect of chemicals on eggs is unknown. However, the most effective preventive tactic is to import only seeds of Neotropical bromeliads.

Credit: J. Lotz, Division of Plant Industry

Credit: J. Lotz, Division of Plant Industry

Credit: J.H. Frank, University of Florida
Selected References
Creel OR. 2000. The evil weevil: what will Florida lose? The Palmetto 19: 10-11, 14-16. (26 June 2013)
Frank JH. 1997. Protection of Florida's native bromeliads by control of Metamasius callizona. Journal of the Bromeliad Society 47: 60-64.
Frank JH. 1999. Bromeliad-eating weevils. Selbyana 20: 40-48.
Frank JH. 1999. Further spread of the weevil Metamasius callizona in Florida. Journal of the Bromeliad Society 49: 206-209.
Frank JH. 2000. Florida's native bromeliads imperiled by exotic evil weevil. The Palmetto 19: 6-9, 12.
Frank JH, Thomas MC. 1994. Metamasius callizona (Chevrolat) (Coleoptera: Curculionidae), an immigrant pest, destroys bromeliads in Florida. Canadian Entomologist 126: 673-682.
Frank JH, Cooper TM, Larson BC. 2006. Metamasius callizona (Coleoptera: Dryophthoridae): longevity and fecundity in the laboratory. Florida Entomologist 89: 208-211.
Frank JH, Fish D. 2008. Potential biodiversity loss in Florida bromeliad phytotelmata due to Metamasius callizona (Coleoptera: Dryophthoridae), an invasive species. Florida Entomologist 91: 1-8.
O'Brien CW, Thomas MC. 1990. The species of Metamasius in Florida (Coleoptera: Curculionidae). Florida Department of Agriculture and Consumer Services, Division of Plant Industry, Entomology Circular 330: 1-4.
O'Brien CW, Thomas MC, Frank JH. 1990. A new weevil pest of Tillandsia in south Florida. Journal of the Bromeliad Society 40: 203-205, 222.
Salas J, Frank JH. 2001. Development of Metamasius callizona (Coleoptera: Curculionidae) on pineapple stems. Florida Entomologist 84: 123-126.
Suazo A, Cave RD, Frank JH. 2008. Reproductive biology and development of Lixadmontia franki (Diptera: Tachinidae), a parasite of bromeliad-eating weevils. Florida Entomologist 91: 453-459.
Vaurie P. 1966. A revision of the neotropical genus Metamasius (Coleoptera, Curculionidae, Rhynchophorinae). Species groups I and II. American Museum of Natural History Bulletin 131: 213-337.
Wood DM, Cave RD. 2006. Description of a new genus and species of weevil parasitoid from Honduras (Diptera: Tachinidae). Florida Entomologist 89: 239-244.