Background
Dooryard (homegrown) avocado fruit are enjoyed by hundreds of thousands of Florida residents. In addition to providing fruit, they offer habitat for wildlife and canopy cover in urban areas. There are over 600,000 avocado trees in Florida home landscapes (Evans and Crane 2019). Most dooryard trees are in Miami-Dade, Broward, Palm Beach, and Lee Counties in southern Florida, but they are also planted in many central Florida counties.
Laurel wilt disease (LW) is caused by Raffaelea lauricola, a fungal mutualistic symbiont (two or more organisms mutually benefiting from living together) of the redbay ambrosia beetle (Xyleborus glabratus) (Figure 1). The redbay ambrosia beetle and R. lauricola are native to India, Japan, Myanmar, and Taiwan (Carrillo et al. 2014). A few years after the beetle and its symbiont were inadvertently introduced into the United States, other ambrosia beetle (AB) species already in Florida have been contaminated with Raffaelea lauricola, and some of them (X. bispinatus and X. volvulus) can transmit the disease (Carrillo et al. 2012; Carrillo et al. 2014). The beetles use the fungus directly as a food source, cultivating fungal gardens within their galleries. The fungus uses the tree's tissue as a source of nutrients.

Credit: D. Carrillo, UF/IFAS
Trees become infected with the LW pathogen when a contaminated ambrosia beetle bores into the tree's xylem (water-conducting tissue) to form galleries and inoculates these galleries with R. lauricola. The infection triggers the tree's natural defense mechanism designed to wall off the pathogen through the production of tyloses (outgrowths of xylem cells that detach from the cell wall and dam up the vascular tissue) and gums (Inch et al. 2012; Inch and Ploetz 2012). Unfortunately, before the tree can successfully wall off the fungus, the fungus repeatedly spreads to new sections of the tree, which keeps triggering the tree's defense system. After a relatively short period of time, the water-conducting tissue (xylem) becomes plugged with tyloses and gums, and water can no longer flow to the leaves and stems, causing the tree to wilt, die back, and die. Time from infection to tree death depends on several factors but can range from three weeks up to three months.
In the United States, native trees in the Lauraceae (Laurel plant family) and non-native trees in this family, such as avocado, are susceptible to LW (Table 1). In addition, at least 32 avocado cultivars have been documented to succumb to LW (Table 2).
Timeline on the Spread of Laurel Wilt in Florida
Laurel wilt was inadvertently introduced into the United States in 2002 at Port Wentworth, Georgia, when wooden packing material, infested with the redbay ambrosia beetle carrying the LW pathogen, was imported from Asia (Mayfield and Thomas 2006) (Figure 2). The redbay ambrosia beetle–fungal pathogen complex was detected in north Florida in 2005 and central Florida in 2006 due to movement of beetle-infested wood. In 2007, the first confirmed avocado tree death in a home landscape occurred in Jacksonville (Duval County), Florida (Mayfield et al. 2008). In February 2010, the redbay ambrosia beetle was detected in a natural area 21 miles (33.7 km) north of the south Florida avocado production area in Miami-Dade County (Okins 2010; Ploetz et al. 2011). By 2011 the first confirmed swampbay (Persea borbonia) tree to succumb to LW was documented in this natural area, and by 2012, LW was detected in a commercial avocado grove (J. Crane, personal communication). Since that time, LW has been detected in all of Florida's 67 counties (Figure 2).
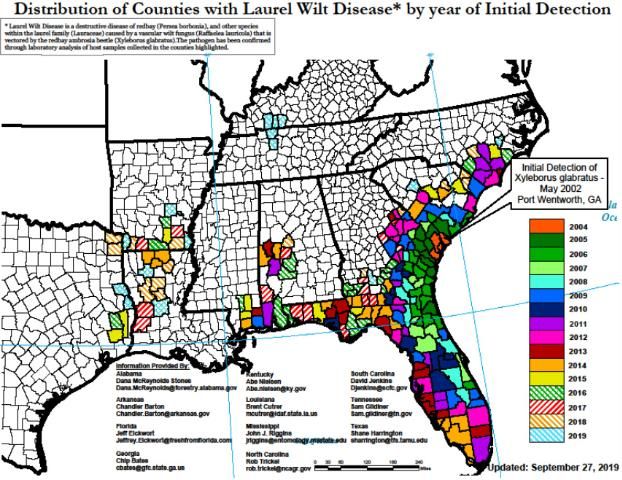
Credit: USDA Forest Service
Update
At present, LW has been detected in 11 southeastern US states, including Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Mississippi, North Carolina, South Carolina, Tennessee, and Texas (USDA Forest Service 2019) (Figure 2). The spread of LW by the redbay ambrosia beetle in natural areas has been estimated to be 15 to 34 miles per year, but the rate of movement through urban areas of Florida with native and avocado trees is unknown (Koch and Smith 2008). Over half a billion native trees in the Lauraceae family have been killed by the disease (Hughes et al. 2017; Snyder 2014; Spence et al. 2013; Rodgers et al. 2014). The potential for human-assisted spread of LW through the movement of ambrosia-beetle-infested wood or plant material is a major concern. Currently, LW has spread as far west as eastern Texas but has not yet reached California. These states have dooryard and commercial avocado trees and along with Mexico and Central and South America are at risk of LW (Crane 2015).
At least nine native and four non-native tree species in the Lauraceae family are susceptible to LW (Table 1). This includes redbay (Persea borbonia), a common understory tree found throughout the natural areas of the Florida peninsula and southeastern United States, and swampbay (P. palustris), which is common in some areas of southern Florida. The native California bay (Umbellularia californica) has been shown to be susceptible to attack by the redbay ambrosia beetle and the LW pathogen (Fraedrich 2008).
In addition to the redbay ambrosia beetle, two other native AB species (Xyleborus volvulus and X. bispinatus) have been documented to carry the LW pathogen and transmit the disease (Atkinson et al. 2013; Carrillo et al. 2012; Carrillo et al. 2014) (Figure 1). These beetles bore into host trees (e.g., avocado and redbay) and reproduce in the galleries (tunnels) they form, thus spending most of their lifecycle protected from predators. The development time from egg to adult is between one and two months, depending upon temperatures and tree host species. Logs, limbs, sections of limbs, and stumps may all be infested. Research has demonstrated that chipping wood significantly reduces the number of AB that emerge from the chipped wood (Spence et al. 2011). Furthermore, the laurel wilt pathogen is not able to survive in chipped wood. The time from initial beetle contact with a host tree to tree damage or death varies with the host species, tree health, and tree size, and ranges from about 21 days to about three months or more.
In addition to natural spread of LW by AB through natural areas, LW can be moved by infested wood products (e.g., firewood and logs moved by entrepreneurs, residents, landscape companies, pruning companies, and wood-turners); movement of wood products to landfills that don't burn or bury materials; and illegal dumping of wood products (logs, brush, limbs, etc.).
Symptoms of LW and AB Infestation
- Leaf and green stem wilt
- Leaf color change from green or light green to dark purplish green or bluish green to greenish brown to brown
- Dead, desiccated leaves hanging on the tree branches
- Stem and limb dieback
- Inspection of the trunk and major limbs may show dried sap (white, crystalline powder-like material) or sawdust tubes protruding from the bark.
- The sapwood below the bark will have dark blue-brown, brown, or black streaks. Normally this sapwood should be white to yellowish with no dark staining or streaking. The staining might not appear evenly along the tree.
- In addition, small, dark holes in the sapwood indicate wood-boring beetles are present.

Credit: J. H. Crane, UF/IFAS
Current Control Recommendations for Dooryard Avocado Trees
- Report any suspicious redbay, swampbay, camphor, sassafras, and avocado trees to the Division of Plant Industry (DPI) at 1-888-397-1517 or your local county UF/IFAS Extension office. These trees are all in the laurel family. Laurel wilt has not been documented in plants outside the laurel family. Trees in other families such as oaks, maples, mangoes, sapodilla, and citrus are not susceptible to the fungus that causes LW.
- A sample of sapwood from trees suspected of having LW may be collected and submitted to DPI or a UF/IFAS Plant Diagnostic Center (https://diagnostics.ifas.ufl.edu).
- Avocado, redbay, and other woody LW host trees should not be moved or sold as firewood, tree trimmings, BBQ smoke-wood, mulch, or wood-turning material. For more information, visit https://www.dontmovefirewood.org/.
- At this time, there are no approved fungicide treatments to prevent or cure laurel-wilt-affected avocado trees in the home landscape.
- The issue of disposal of dead or dying trees is complicated by numerous state, county, and local regulations. Current recommendations for urban and rural residents with redbay, swampbay, or avocado trees that have LW vary, but these trees should be destroyed because of their potential as AB habitat and reproduction sites that will increase the beetle population and further spread the disease. Chipping and tarping infested wood material has been shown to be an effective sanitation procedure to reduce the potential for AB to survive and reproduce (Spence et al. 2011).
- Potential options for tree disposal vary by county and local regulations and may include cutting the tree down and placing the wood into the urban debris stream (i.e., the wood is taken to the local landfill and destroyed or buried); cutting the tree into sections, chipping or grinding the entire tree, then covering the debris with a tarp for at least seven days; or composting the tree by cutting it to the ground, placing all wood (or chips) on top of the stump, and covering it with a tarp all the way to the ground. However, composting is not allowed in some urban areas, so please contact your local county government for guidance. Burning is not recommended because of the necessity to obtain state, county, and/or municipal burn permits and the danger of uncontrolled burning by residents.
- Extreme caution should be used when moving live host trees (e.g., redbay and avocado) and wood products into counties, states, and areas outside the United States where LW is not yet found. Insect- and disease-free containerized host trees should only be purchased from registered nurseries, and trees showing any signs of wilt or dieback should be destroyed immediately.
Please contact your local UF/IFAS Extension office for more information (http://sfyl.ifas.ufl.edu/find-your-local-office/).
Frequently Asked Questions
- Are leaf wilt and stem dieback symptoms indicative of an ambrosia beetle–laurel wilt disease attack? Not necessarily. Wilting of leaves and young stems, dead leaves hanging onto the stems, and stem and limb dieback may be due to a lightning strike, flooding, severe drought events, or an infestation of one or more of the many AB already in Florida. However, these symptoms are similar to those caused by LW infection, and therefore the tree should be tested to determine if the LW pathogen is present.
- Can the laurel wilt pathogen live in soil? No, the LW pathogen needs a living host and does not survive in the soil.
- Can I replace my dead avocado tree with a new avocado tree? Yes. In general, AB are not attracted to young avocado trees with small stem diameter. This is thought to be because a small trunk and limbs are not a suitable site for AB to make galleries in which they can grow the LW pathogen and reproduce. Furthermore, the LW pathogen cannot survive in the soil, so newly planted avocado trees are not in danger of contracting the pathogen through the soil.
- Are there any LW pathogen-resistant avocado varieties? There do not appear to be any resistant avocado varieties, although testing continues.
- Should I apply insecticides to try to control ambrosia beetles? There are contact insecticides that can kill ambrosia beetles, but most of them are not available for homeowner use, and ambrosia beetles spend most (~90%) of their time inside the tree, where contact insecticides will not reach them.
More information about LW and AB may be found at:
- UF/IFAS (https://sfyl.ifas.ufl.edu)
- UF/IFAS Electronic Digital Information System (free publications) (https://edis.ifas.ufl.edu/)
- UF/IFAS Tropical Research and Education Center (https://trec.ifas.ufl.edu/people/jonathan-crane/lw-ab-website/)
- FDACS Division of Plant Industry (DPI) (https://www.fdacs.gov/Divisions-Offices/Plant-Industry)
- DPI—Laurel Wilt (https://www.fdacs.gov/Agriculture-Industry/Pests-and-Diseases/Plant-Pests-and-Diseases/Laurel-Wilt-Disease)
- DPI—Save the Guac (https://www.fdacs.gov/Consumer-Resources/Protect-Our-Environment/Save-the-Guac)
- Southern Forest Health (USDA Forest Service) (http://southernforesthealth.net/diseases)
Literature Cited
Atkinson, T. H., D. Carrillo, R. E. Duncan, and J. E. Peña. 2013. "Occurrence of Xyleborus bispinatus (Coleoptera: Curculionidae: Scolytinae) Eichhoff in South Florida." Zootaxa 3669 (1): 96–100. https://doi.org/10.11646/zootaxa.3669.1.10
Carrillo, D., R. E. Duncan, and J. E. Peña. 2012. "Ambrosia Beetles (Coleoptera: Curculionidae: Scolytinae) That Breed in Avocado Wood in Florida." The Fla. Entomologist 95 (3): 11–13. https://doi.org/10.1653/024.095.0306
Carrillo, D., R. E. Duncan, J. N. Ploetz, A. F. Campbell, R. C. Ploetz, and J. E. Peña. 2014. "Lateral Transfer of a Phytopathogenic Symbiont among Native and Exotic Ambrosia Beetles." Plant Pathology 63: 54–62. https://doi.org/10.1111/ppa.12073
Crane, J. H., E. A. Evans, D. Carrillo, R. C. Ploetz, and A. J. Palmateer. 2015. "The Potential for Laurel Wilt to Threaten Avocado Production Is Real." ACTAS, Proceedings of the VIII Congreso Mundial de la Palta, Lima, Peru. 13–18 Sept. 2015.
Evans, Edward A., and J. H. Crane. 2009. “Estimates of the Replacement Costs of Commercial and Backyard Avocado Trees in South Florida: FE825/FE825, 12/2009”. EDIS 2009 (10). Gainesville, FL. https://doi.org/10.32473/edis-fe825-2009.
Fraedrich, S. W. 2008. "California Laurel Is Susceptible to Laurel Wilt Caused by Raffaelea lauricola." Plant Dis. 92: 1469. https://doi.org/10.1094/PDIS-92-10-1469A
Fraedrich, S. W., T. C. Harrington, R. J. Rabaglia, M. D. Ulyshen, A. E. Mayfield III, J. L. Hanula, J. M. Eickwort, and D. R. Miller. 2008. "A Fungal Symbiont of the Redbay Ambrosia Beetle Causes a Lethal Wilt in Redbay and Other Lauraceae in the Southeastern United States." Plant Dis. 92: 215–224. https://doi.org/10.1094/PDIS-92-2-0215
Fraedrich, S. W., T. C. Harrington, C. A. Bates, J. Johnson, L. S. Reid, G. S. Best, T. D. Leininger, and T. S. Hawkins. 2011. "Susceptibility to Laurel Wilt and Disease Incidence in Two Rare Plant Species, Pondberry and Pondspice." Plant. Dis. 95: 1056–1062. https://doi.org/10.1094/PDIS-11-10-0841
Fraedrich, S. W., T. C. Harrington, and G. S. Best. 2015. "Xyleborus glabratus Attacks and Systemic Colonization by Raffaelea lauricola Associated with Dieback of Cinnamomum camphora in the Southeastern United States." For. Path. 45: 60–70. https://doi.org/10.1111/efp.12124
Hughes, M. A., K. Shin, J. Eickwort, and J. A. Smith. 2012. "First Report of Laurel Wilt Disease Caused by Raffaelea lauricola on Silk Bay in Florida." Disease Notes 96: 910. https://doi.org/10.1094/PDIS-02-12-0149-PDN
Hughes, M. A., G. Brar, R. C. Ploetz, and J. A. Smith. 2013. "Field and Growth Chamber Inoculations Demonstrate Persea indica a Newly Recognized Host of the Laurel Wilt Pathogen, Raffaelea lauricola." Plant Health Progress 14 (1). doi:10.1094/PHP-2013-0814-02-BR. https://doi.org/10.1094/PHP-2013-0814-02-BR
Hughes, M. A., A. Black, and J. A. Smith. 2014. "First Report of Laurel Wilt Caused by Raffaelea lauricola on Bay Laurel (Laurus noblis) in the United States." Plant Dis. 98: 1159. https://doi.org/10.1094/PDIS-02-14-0194-PDN
Hughes, M. A., J. J. Riggins, F. H. Koch, A. I. Cognato, C. Adersno, J. P. Formby, T. J. Dreaden, R. C. Ploetz, and J. A. Smith. 2017. "No Rest for the Laurels: Symbiotic Invaders Cause Unprecedented Damage to South USA Forests." Biological Invasions 19: 2143–2157. https://doi.org/10.1007/s10530-017-1427-z
Inch, S. A., and R. C. Ploetz. 2012. "Impact of Laurel Wilt, Caused by Raffaelea lauricola, on Xylem Function in Avocado, Persea americana." For. Plath. 42: 239–245. https://doi.org/10.1111/j.1439-0329.2011.00749.x
Inch, S., R. Ploetz, B. Held, and R. Blanchette. 2012. "Histological and Anatomical Responses in Avocado, Persea americana, Induced by the Vascular Wilt Pathogen, Raffaelea lauricola." Botany 90: 627–635. https://doi.org/10.1139/b2012-015
Koch, F. H., and W. D. Smith. 2008. "Spatio-temporal Analysis of Xyleborus glabratus (Coleoptera: Curculionidae: Scolytinae) Invasion in Eastern U.S. Forests." Environ. Entomol. 37: 442–452. https://doi.org/10.1603/0046-225X(2008)37[442:SAOXGC]2.0.CO;2
Mayfield, A. E. III, and M. C. Thomas. 2006. "The Redbay Ambrosia Beetle, Xyleborus glabratus Eichhoff (Scolytinae: Curculionidae)." FDACS Pest Alert June 2006.
Mayfield, A. E. III, J. A. Smith, M. Hughes, and T. J. Dreaden. 2008. "First Report of Laurel Wilt Disease Caused by a Raffaelea sp. on Avocado in Florida." Plant Dis. 96: 976. https://doi.org/10.1094/PDIS-92-6-0976A
Okins, K. 2010. Entomology Specimen Report E2010-1101-1. Gainesville: FDACS-DPI.
Ploetz, R. C., J. E. Peña, J. A. Smith, T. J. Dreaden, J. H. Crane, T. Schubert, and W. Dixon. 2011. "Laurel Wilt, Caused by Raffaelea lauricola, Is Confirmed in Miami-Dade County, Center of Florida's Commercial Avocado Production." Disease Notes 95 (12): 1589. https://doi.org/10.1094/PDIS-08-11-0633
Ploetz, R. C., and J. Konkol. 2013. "First Report of Gulf Licaria, Licaria trianda, as a Suspect of Laurel Wilt." Plant Dis. 97: 1248. https://doi.org/10.1094/PDIS-01-13-0027-PDN
Rodgers, L., A. Derksen, and T. Pernas. 2014. "Expansion and Impact of Laurel Wilt in the Florida Everglades." Fla. Entomologist 97 (3): 1247–1250. https://doi.org/10.1653/024.097.0335
Snyder, J. R. 2014. "Ecological Implications of Laurel Wilt Infestation on Everglades Tree Islands, South Florida." U.S. Geological Survey Open-File Report 2014-1225. https://doi.org/10.3133/ofr20141225
Spence, D., M. Hughes, and J. Smith. 2013. "Laurel Wilt: an Exceptionally Damaging Exotic Disease that Threatens Florida's Forests." Journal of Florida Studies 1 (2). https://www.journaloffloridastudies.org/0102laurelwilt.html.
Spence, D., J. Smith, A. Mayfield III, J. Hulcr, R. Ploetz, and L. Stelinski. 2011. Assessing the Survival of the Redbay Ambrosia Beetle and Laurel Wilt Pathogen in Wood Chips. FOR289 (archived). Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://doi.org/10.32473/edis-fr351-2011
Spence, D. J., J. A. Smith, R. Ploetz, J. Hulcr, and L. L. Stelinski. 2013. "Effect of Chipping on Emergence of the Redbay Ambrosia Beetle (Coleoptera: Curculionidae: Scolytinae) and Recovery of the Laurel Wilt Pathogen from Infested Wood Chips." Entomological Soc. of America 106: 2093–2100. https://doi.org/10.1603/EC13072
USDA Forest Service. 2019. "Distribution of Counties with Laurel Wilt Disease by Year of Initial Detection." https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/fseprd571973.pdf.
Acknowledgments
The authors thank the following supporting entities: USDA-NIFA grant 2015-51181-24257: Laurel Wilt of Avocado: Management of an Unusual and Lethal Disease; UF/IFAS; the Florida Avocado Administrative Committee; UF/IFAS Extension Miami-Dade County; and the Florida Department of Agriculture and Consumer Services.