Introduction
The invasion of the Indo-Pacific lionfish (Pterois volitans/miles complex) into the western Atlantic Ocean is considered the most successful marine fish invasion (Morris and Whitfield 2009). Lionfish were first detected offshore of southeast Florida in 1985 and are now established throughout the western Atlantic Ocean, the Caribbean Sea, and the Gulf of Mexico (GOM) (Schofield 2010, Schofield 2018). Lionfish consume native species and compete with them for habitat and food (Morris and Akins 2009), and negatively impact native reef fish communities (Green et al. 2012; Albins 2015; Dahl et al. 2016). Lionfish occupy many marine habitats in the invaded range, including artificial and natural reefs (Morris and Whitfield 2009; Dahl and Patterson 2014), patch reefs in estuaries (Jud and Layman 2012), seagrass beds (Claydon et al. 2012), and mangroves (Barbour et al. 2010). Lionfish have broad tolerances to temperature (Kimball et al. 2004) and salinity (Jud et al. 2015), and they are found as deep as 400 m or 1,300 ft (Schofield 2010). Given the abundance and distribution of invasive lionfish, natural mechanisms may be necessary to achieve region-wide population control.
Densities of lionfish in their native Indo-Pacific range are substantially lower than densities reported in their invaded western Atlantic range (Kulbicki et al. 2012). However, the primary mechanism that controls their native range densities (e.g., predators, parasites, disease, or competition for resources) is unknown. Reports indicate that native predators may be learning to consume lionfish in parts of their invaded range (Maljkovic et al. 2008; Diller et al. 2014; Muñoz 2017), but there is no evidence that invasive lionfish populations are controlled by either predators (Hackerott et al. 2013; Valdivia et al. 2014) or parasites (Sikkel et al. 2014; Sellers et al. 2015; Fogg et al. 2016; Tuttle et al. 2017). Thus, natural biocontrol of lionfish to this point is likely limited to competition for resources (Chagaris et al. 2017) and cannibalism (Dahl et al. 2017).
High lionfish population densities (Dahl and Patterson 2014) and low genetic diversity (Pérez-Portela et al. 2018) in the invaded range could make their populations more susceptible to disease. Lionfish support different communities of environmentally derived microbes than native Caribbean reef fishes (Stevens and Olson 2013), and these microbial communities are differentially colonized by microbes from their local environments (Stevens and Olson 2015). The lack of an adequate immune response to pathogens (i.e., disease-causing microbes) present in the invaded systems may cause invasive lionfish populations to be susceptible to disease (Lee and Klasing 2004; White and Perkins 2012).
Here, we report the first observed incidences of an emerging ulcerative skin disease in lionfish in the Western Atlantic Ocean, the GOM and the Caribbean Sea. This disease has recently appeared in the population, and its geographic range appears to be expanding. The purpose of this extension publication is to (1) describe the gross pathology (i.e., tissue appearance and condition) of the disease, (2) present initial reports of the chronology and geography for reported observations of this ulcerative skin disease, (3) outline our current knowledge gaps and research efforts, and, (4) encourage divers, lionfish harvesters, conservation organizations, and scientists working with lionfish to report and submit photograph observations of lionfish with skin ulcers to the Fish Kill Hotline at the Florida Fish and Wildlife Conservation Commission (FWC), 1-800-636-0511.
Gross Pathology of Ulcerations
Lionfish with skin ulcers were collected in the northern Gulf of Mexico (nGOM) and processed for diagnostic evaluation by researchers at the University of Florida (UF) and FWC. Representative fish were subjected to necropsy (i.e., postmortem dissection of carcasses). Necropsy results provided evidence that skin ulcers were variable in size, location, and number among ulcerated lionfish. Deep skin ulcers often exposed skeletal muscle, with a sloughing of dead tissue (Figure 1). Skin tissue near the ulcers showed evidence of healing, suggesting that the initial wound occurred potentially days or weeks before those lionfish were collected.
Histopathology (i.e., microscopic examination of tissue) and bacteriology (i.e., examination of the bacterial community) were performed on lionfish to identify the causative agent (i.e., disease-causing organism). These evaluations were performed on multiple tissues, including the leading edges of skin ulcers, as well as kidney and brain tissue. Microscopic wet mount evaluations (i.e., examination of fresh tissue squashes) of the fins, gills, and skin ulcers were negative for parasites, water molds, or other potential infectious causes. Ultimately, a causative agent was not identified during these initial evaluations.

Observations of Ulcerated Lionfish
The first documented report of lionfish presenting with skin ulcers occurred on August 5, 2017, from lionfish harvested off artificial reefs near Destin, FL (Table 1). On this day, 201 (40%) of 503 harvested lionfish had skin ulcers. Additionally, a single dead lionfish was observed on the seabed adjacent to an artificial reef, and multiple dead lionfish were observed floating at the sea surface (personal communication, J. Livingston, Dreadknot Charters).
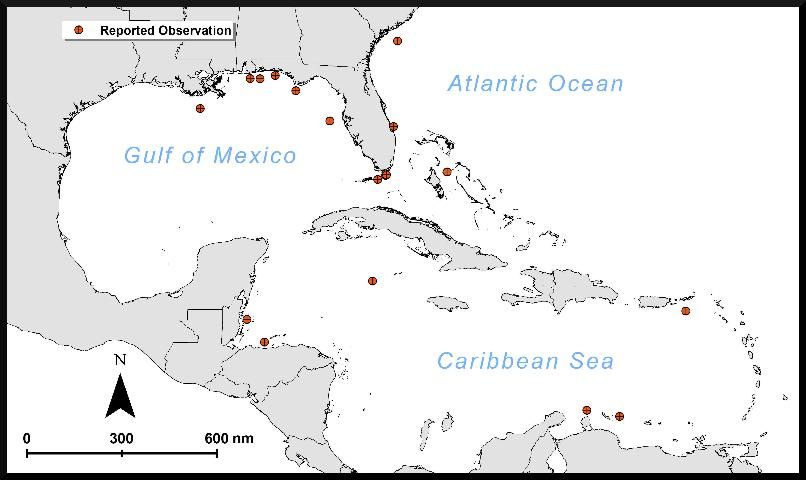
Lionfish with the ulcerative skin disease have since been reported from waters of the West Florida Shelf, the East Florida Shelf, and the Florida Keys, as well as throughout the Caribbean Sea (Table 1; Figure 2). To date, reports from the nGOM have also had the highest reported numbers and percentages of ulcerated lionfish. In the wider Caribbean region, ulcerated lionfish have been reported from The Bahamas, Grand Cayman, Aruba, St. Croix, Bonaire, Útila, and Belize (Table 1; Figure 2). Images of lionfish published via various internet media and included in personal communication with the authors suggest ulcerated lionfish in the Caribbean Sea and East Florida Shelf regions (Figure 1, E and F) have a gross pathology similar to that found in ulcerated lionfish observed in the nGOM (Figure 1, A–D). However, it is unknown whether the causative agent is shared among regions where ulcerated lionfish have been observed.
Preliminary data collected from lionfish harvested in the nGOM indicate a general decline in the proportion of lionfish presenting with skin ulcers through winter and early spring 2018. Additionally, many of the ulcerated lionfish harvested during spring and early summer of 2018 had scar tissue suggestive of healing associated with wounds (Figure 1, C).
Knowledge Gaps and Upcoming Research
Further research to determine a causative agent will help clarify how the ulcers affect lionfish flesh, impacts on lionfish health, and the potential for the disease to transmit to other species. To our knowledge, there have been no reported cases of human illness due to consuming ulcerated lionfish. However, if a lionfish has ulcers or appears unhealthy, we suggest you avoid direct contact and not eat the ulcerated lionfish. If you have touched an ulcerated fish, wash with soap and water. Fresh lionfish is a tasty, healthful source of protein when handled and prepared properly. Eating invasive lionfish is encouraged as one of the best options to mitigate their impacts on native reef communities.
Collaborative research between UF and FWC scientists is underway to describe the cause of this ulcerative disease. This research uses molecular tools to evaluate lionfish sampled across a wide geographic range and determine whether the disease originates from a virus. Using a multidisciplinary approach, tissue samples from ulcerated lionfish and seemingly healthy lionfish will be processed using polymerase chain reaction (PCR) assays that target known fish viruses. Viral and bacterial genetic material sourced directly from the environment will be processed using next-generation sequencing techniques. Determining the causative agent will provide insight into the lethal and sublethal effects of the disease on lionfish and the likelihood that the disease will transmit to other species.
Concurrently, research is being conducted to evaluate the effects of the ulcerative disease on individual lionfish and on lionfish populations. We are continuing long-term monitoring efforts at nGOM artificial and natural reef sites via remote operated vehicle video (ROV) surveys. Data from ROV surveys of lionfish densities conducted before disease emergence will be compared to data on lionfish densities collected after disease emergence. We are also examining the relative condition (i.e., whether fish are heavier or lighter than predicted for a given length), to compare the relative condition of lionfish with skin ulcers to the relative condition of apparently healthy individuals. Finally, commercial lionfish catches and tournament landings are being analyzed for potential changes in population size and age structure. Together, this information will be used to assess potential changes in lionfish abundances and evaluate implications for the lionfish fishery and reef fish communities.
Where to Report Observations of Ulcerated Lionfish
If you observe lionfish with skin ulcers, please report your observations to the FWC Fish Kill Hotline by telephone (1-800-636-0511), online (https://app.myfwc.com/FWRI/FishKillReport/Submit.aspx), or by using the FWC Reporter mobile application from iTunes or Google Play. If possible, please include the following: 1) the date of the occurrence, 2) the location, 3) the bottom water temperature, and 4) photographs of the lionfish. The Fish Kill Hotline is maintained by the FWC group based in St. Petersburg at the FWC Research Institute, the research arm of the FWC. Every report is documented in the searchable database and available online at https://app.myfwc.com/FWRI/FishKillReport/SearchResults.aspx.
Conclusion
The first recorded observation of an ulcerative skin disease in lionfish occurred on August 5, 2017, in northwest Florida. Subsequent observations have been reported offshore of states bordering the GOM, along the US east coast, and in locations throughout the Caribbean. The highest numbers of ulcerated lionfish have been reported in northwest Florida. Pathologic examinations found skin ulcers that varied in size and location, often with skin tissue decay that exposed deeper muscle tissues. While a pathogen was not identified during initial diagnostic evaluations, we anticipate molecular tools will enable us to better understand the origin of this ulcerative disease and the potential for this disease to be transmitted to other species. At the same time, we are evaluating the lethal and sublethal effects of this ulcerative skin disease and how the disease may ultimately impact lionfish populations, the lionfish fishery, and reef fish communities. We ask for your help: please report any observations of ulcerated lionfish to the FWC Fish Kill Hotline (1-800-636-0511).
Acknowledgments
We thank Jeff Hill for his initial advice and review and the many individuals and organizations who provided information regarding the chronology and geography of observed ulcerations: (alphabetical order) Alfonso Aguilar-Perera, Brian Asher, Micah Bakenhaster, Buck Beasley, Rachel Bowman, Brian Bounds, Bibi Burnette, Paul Cordone, Caren Eckrich, Corey Eddy, Allie ElHage, Meaghan Faletti, Tom Frazer, Michael Funk, Steve Garner, Scott Harrell, Andre Johnson, Bradley Johnson, Michelle Johnston, Lisette Keus, Joe Livingston, Josh Livingston, Paul Maneval, Carl Molitor, Eliezer Muñoz, Sean Patterson, Michael Pement, Tommy Phelps, Alan Pierce, Adam Richardson, Serena Rivero, Andy Ross, Ben Schapiro, Grayson Shepard, Tim Shivers, Jerome Spruit, Kali Spurgin, Ron Surrency, Joe Tarnecki, Hanna Tillotson, Bas Tol, Cory Trier, Justin Wallheiser, Jason Washington, Adam Wise, Virgil Zetterlind, Bonaire Lionfish Hunters, Caribbean Oceanic Restoration and Education Foundation, Cayman United Lionfish League, Coast Watch Alliance, Deepwater Mafia Dive Club, and Lionfish Caribbean. Financial support for this research is provided by the Florida Restore Act Centers of Excellence Program, Gulf of Mexico Research Initiative, and Florida Fish and Wildlife Conservation Commission. Financial support for H.E. Harris is provided by the National Science Foundation Graduate Research Fellowship Program [grant no. DGE-1315138]. Additional support provided by Florida Sea Grant. Opinions, findings, conclusions, or recommendations expressed in this document do not necessarily reflect the views of our supporting organizations.
References
Albins, M. A. 2015. "Invasive Pacific lionfish Pterois volitans reduce abundance and species richness of native Bahamian coral-reef fishes." Marine Ecology Progress Series 522:231–243.
Barbour, A. B., M. L. Montgomery, A. Adamson, E. Díaz-Ferguson, and B. R. Silliman. 2010. "Mangrove use by the invasive lionfish Pterois volitans." Marine Ecology Progress Series 401:291–294.
Chagaris, D., S. Binion-Rock, A. Bogdanoff, K. Dahl, J. Granneman, H. Harris, J. Mohan, M. B. Rudd, M. K. Swenarton, R. Ahrens, W. F. Patterson, J. A. Morris, and M. Allen. 2017. "An ecosystem-based approach to evaluating impacts and management of invasive lionfish." Fisheries 42(8):421–431. Taylor & Francis.
Chai, J. Y., K. D. Murrell, and A. J. Lymbery. 2005. "Fish-borne parasitic zoonoses: Status and issues." International Journal for Parasitology 35(11–12):1233–1254.
Claydon, J. B., M. C. Calosso, and S. B. Traiger. 2012. "Progression of invasive lionfish in seagrass, mangrove and reef habitats." Marine Ecology Progress Series 448:119–129.
Dahl, K. A., and W. F. Patterson. 2014. "Habitat-specific density and diet of rapidly expanding invasive red lionfish, Pterois volitans, populations in the northern Gulf of Mexico." PLoS ONE 9(8):e105852.
Dahl, K. A., W. F. Patterson, A. Robertson, and A. C. Ortmann. 2017. "DNA barcoding significantly improves resolution of invasive lionfish diet in the northern Gulf of Mexico." Biological Invasions 19(6):1917–1933.
Dahl, K. A., W. F. Patterson, and R. A. Snyder. 2016. "Experimental assessment of lionfish removals to mitigate reef fish community shifts on northern Gulf of Mexico artificial reefs." Marine Ecology Progress Series 558:207–221.
Diller, J. L., T. K. Frazer, and C. A. Jacoby. 2014. "Coping with the lionfish invasion: Evidence that naïve, native predators can learn to help." Journal of Experimental Marine Biology and Ecology 455(2014):45–49. Elsevier B.V.
Fogg, A. Q., C. F. Ruiz, S. S. Curran, and S. A. Bullard. 2016. "Parasites from the Red Lionfish Pterois volitans from the Gulf of Mexico." Gulf and Caribbean Research 27(1):SC 1–5.
Green, S. J., J. L. Akins, A. Maljkovic, and I. M. Côté. 2012. "Invasive lionfish drive Atlantic coral reef fish declines." PLoS ONE 7(3):e32596. Public Library of Science.
Hackerott, S., A. Valdivia, S. J. Green, I. M. Côté, C. E. Cox, L. Akins, C. A. Layman, W. F. Precht, and J. F. Bruno. 2013. "Native predators do not influence invasion success of Pacific lionfish on Caribbean reefs." PLoS ONE 8(7):e68259. Public Library of Science.
Jud, Z. R., and C. A. Layman. 2012. "Site fidelity and movement patterns of invasive lionfish, Pterois spp., in a Florida estuary." Journal of Experimental Marine Biology and Ecology 414–415:69–74.
Jud, Z. R., P. K. Nichols, and C. A. Layman. 2015. "Broad salinity tolerance in the invasive lionfish Pterois spp. may facilitate estuarine colonization." Environmental Biology of Fishes 98(1):135–143.
Kimball, M. E., J. M. Miller, P. E. Whitfield, and J. A. Hare. 2004. "Thermal tolerance and potential distribution of invasive lionfish (Pterois volitans/miles complex) on the east coast of the United States." Marine Ecology Progress Series 283:269–278.
Kulbicki, M., J. Beets, P. Chabanet, K. Cure, E. Darling, S. R. Floeter, R. Galzin, A. Green, M. Harmelin-Vivien, M. Hixon, Y. Letourneur, T. L. D. Loma, T. McClanahan, J. McIlwain, G. Moutham, R. Myers, J. K. O'Leary, S. Planes, L. Vigliola, and L. Wantiez. 2012. "Distributions of Indo-Pacific lionfishes Pterois spp. in their native ranges: implications for the Atlantic invasion." Marine Ecology Progress Series 446, 189–205.
Lee, K. A., and K. C. Klasing. 2004. "A role for immunology in invasion biology." Trends in Ecology and Evolution 19(10):523–529.
Maljkovic, A., T. E. Van Leeuwen, and S. N. Cove. 2008. "Predation on the invasive red lionfish, Pterois volitans (Pisces: Scorpaenidae), by native groupers in the Bahamas." Coral Reefs 27(3):501.
Morris, J. A., and P. E. Whitfield. 2009. "Biology, ecology, control and management of the invasive Indo-Pacific lionfish: An updated integrated assessment." NOAA Technical Memorandum NOS NCCOS 99. 57 pp.
Morris, J. A., and J. L. Akins. 2009. "Feeding ecology of invasive lionfish (Pterois volitans) in the Bahamian archipelago." Environmental Biology of Fishes 86(3):389–398.
Muñoz, R. C. 2017. "Evidence of natural predation on invasive lionfish, Pterois spp., by the spotted moray eel, Gymnothorax moringa." Bulletin of Marine Science 93(3):789–790.
Pérez-Portela, R., A. Bumford, B. Coffman, S. Wedelich, M. Davenport, A. Fogg, M. K. Swenarton, F. Coleman, M. A. Johnston, D. L. Crawford, and M. F. Oleksiak. 2018. "Genetic homogeneity of the invasive lionfish across the northwestern Atlantic and the Gulf of Mexico based on Single Nucleotide Polymorphisms." Scientific Reports 8(1):5062. Nature Publishing Group.
Schofield, P. J. 2010. "Update on geographic spread of invasive lionfishes (Pterois volitans [Linnaeus, 1758] and P. miles [Bennett, 1828]) in the Western North Atlantic Ocean, Caribbean Sea and Gulf of Mexico." Aquatic Invasions 5(SUPPL. 1):117–122.
Sellers, A. J., G. M. Ruiz, B. Leung, and M. E. Torchin. 2015. "Regional variation in parasite species richness and abundance in the introduced range of the invasive lionfish, Pterois volitans." PLoS ONE 10(6):e0131075. Public Library of Science.
Sikkel, P. C., L. J. Tuttle, K. Cure, A. M. Coile, and M. A. Hixon. 2014. "Low susceptibility of invasive red lionfish (Pterois volitans) to a generalist ectoparasite in both its introduced and native ranges." PLoS ONE 9(5):e95854. Public Library of Science.
Stevens, J. L., and J. B. Olson. 2013. "Invasive lionfish harbor a different external bacterial community than native Bahamian fishes." Coral Reefs 32(4):1113–1121. Springer Berlin Heidelberg.
Stevens, J. L., and J. B. Olson. 2015. "Bacterial communities associated with lionfish in their native and invaded ranges." Marine Ecology Progress Series 531:253–262.
Tuttle, L. J., P. C. Sikkel, K. Cure, and M. A. Hixon. 2017. "Parasite-mediated enemy release and low biotic resistance may facilitate invasion of Atlantic coral reefs by Pacific red lionfish (Pterois volitans)." Biological Invasions 19(2):563–575. Springer International Publishing.
Valdivia, A., J. F. Bruno, C. E. Cox, S. Hackerott, and S. J. Green. 2014. "Re-examining the relationship between invasive lionfish and native grouper in the Caribbean." PeerJ 2:e348.
White, T. A., and S. E. Perkins. 2012. "The ecoimmunology of invasive species." Functional Ecology 26(6):1313–1323. Wiley/Blackwell.