The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The soft scale insect, Phalacrococcus howertoni Hodges & Hodgson, was first detected in Monroe County, Florida, in 2008 on the stem of a tropical shrub called croton, Codiaeum variegatum (L.) A. Juss. (Hodges and Hodgson 2010). The observation of the pest insect on the croton plant gave rise to its common name, the croton scale. This scale is a relatively new pest in Florida, and further studies on this insect are important as it may pose a significant threat to tropical and subtropical ornamental host plants such as croton, as well as ecologically important native plants such as buttonwood, Conocarpus erectus (L.).
Distribution
The croton scale has been detected in 21 counties in Florida including Monroe, Broward, Miami-Dade, Palm Beach, Duval, Indian River, Lee, Orange, Palm Beach, Putnam, and St. Lucie (Hodges 2008; Hodges and Hodgson 2010). The croton scale has also been reported in Guatemala, Barbados, the island of Guadeloupe in the French West Indies, and the Hawaiian island of O’ahu (Etienne et al. 2014; Roerk and Matsunaga 2019). The croton scale is considered by scientists to be morphologically distinct enough from other genera of soft scale to be placed in a new genus, which poses difficulties in determining its geographic point of origin, and its native country of origin is unknown.
Description
The croton scale was deemed taxonomically different from other soft scale insects in the family Coccidae, and a new species and genus classification was needed to describe this new insect (Hodges and Hodgson in 2010). Scale insects have discrete characteristics between males and females. Females are wingless, tiny, ovular sacs that are 4 mm (~1/8 in) long and 2 mm (~1/16 in) wide (Hodges and Hodgson 2010). Females range from yellow-green to green in color, and have dark linear marks on their dorsal side (Hodges and Hodgson 2010). Males are determined by having wings, orange bodies, and their body shape is not as ovular compared to females. Male croton scales are between 1.5 to 1.63 mm (~1/16 in) in length with two pairs of simple eyes, 10 segmented antennae, and the head appears diamond-shaped when viewed from the dorsal side of the insect (Hodges and Hodgson 2010). The male croton scale can be distinguished from other male soft scales by long bristles on antennal segments 7 to 10, and short fleshy, thick setae (hairs) that end in a blunt tip on the antennae and legs (Hodges and Hodgson 2010). Males are comparatively under studied as they are not observed as frequently, and male feeding habits are unknown. See Hodges and Hodgson (2010) for additional characteristics differentiating the male croton scale from other male scales.
Life Cycle and Biology
Eggs
Eggs are laid by females onto the foliage of host plants and immediately hatch into first instar nymphs upon oviposition (Amarasekare and Mannion 2011).
Nymphs
Nymphs of croton scales are referred to as "crawlers," because they are mobile and move around on their host plant. Female nymphs stay under the adult female’s body for approximately 24 hours before moving to feed on other parts of the host leaves (Amarasekare and Mannion 2011). Female nymphs go through three nymphal instars (first, second, and third instar), and increase in size in each life stage until reaching adulthood. Male croton scales go through two mobile nymphal life stages (Amarasekare and Mannion 2011) (Figure 1).
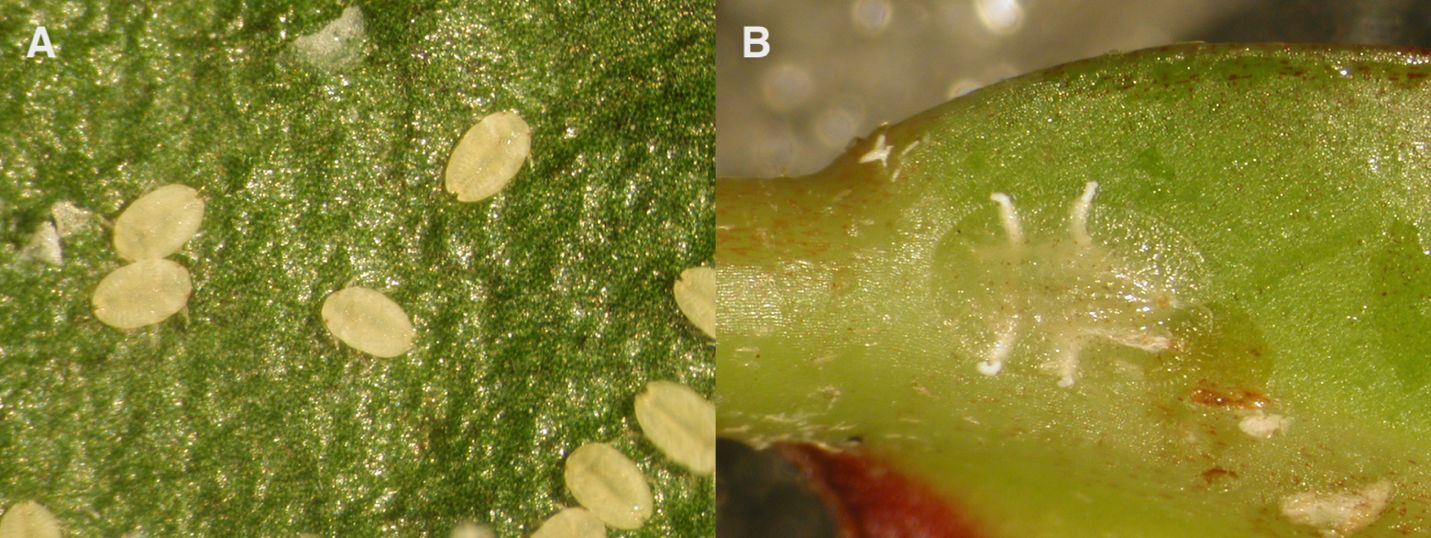
Credit: Catherine Mannion
Pseudo-Pupae
A pupal stage typically indicates complete metamorphosis (i.e. the pupa of a butterfly is its chrysalis); however croton scales do not undergo a true, complete metamorphosis. Instead, croton scales undergo a pseudo-pupal stage that is unique to male croton scale insects (Figure 2). At the end of the second nymphal instar, males can be differentiated from females as their bodies become more elongate in shape and eyes become pigmented (Amarasekare and Mannion). The elongation and eye pigmentation marks the beginning of the stationary final two male instars: the pre-pupal and pupal instars. In these final life stages, the males are sessile and begin to develop into mature adults as wing buds slowly mature through each pseudo-pupal life stage.

Credit: Catherine Mannion
Adults
Adult males are short-lived, with longevity measured at less than one day in the laboratory at a temperature of 27°C (80.6°F) (Amarasekare and Mannion 2011). Females have been documented to have a lifespan of 60.3 to 61.5 days in the laboratory at a temperature of 27°C (80.6°F). Lifespan was determined to be the same on host plant croton and buttonwood (Amarasekare and Mannion). Adult females can produce multiple, overlapping generations over the course of their lifetime and can lay up to 400 eggs/female over the course of their lifetime. Overwintering, or the period where the scale insects are less active and slow down metabolic functions similar to a hibernating animal, is done in the adult stage (Hodges and Hodgson 2010). For examples of adult male and female croton scales, see Figure 3.

Credit: Catherine Mannion
Host Plants
Hodges and Hodgson (2010) documented 72 host plant species from 34 plant families commonly found in Florida in their report of croton scale as a new species. These plants were described as host plants, but not evaluated in terms of suitability for successful life cycle completion of the croton scale. Important economic agricultural and ornamental host plants include guava (Psidium guajava L.), mango (Mangifera indica L.), croton (Codiaeum variegatum (L.) A. Juss.), buttonwood (Conocarpus erectus L.), Mysore fig (Ficus mysorensis Roth), and strangler fig (Ficus aurea Nutt.).
Damage
Like other soft scale insect pests, female croton scales cause the most economic damage to host plants. The female croton scale produces large amounts of honeydew, or sugary frass (insect excrement). The honeydew coats the host plant around the croton scales, which can be found aggregating densely on the underside of host plant foliage. Honeydew is an ideal substrate for a fungus called sooty mold to grow. Sooty mold, like its name suggests, is dark and soot-like in color, and can coat host plant leaves, which blocks photosynthesis and leads to plant health decline.
Management
A good indicator of scale insects on your plants is the increased presence of ants. The ants’ behavior is often described as farming the scale insects because they protect the scales from natural enemies so they can feed on their sugary frass. If you follow the ants’ path on your plant, they will likely lead you to an infestation of scales. Kanga et al. (2016) reported predation of the croton scale by the ladybird species Thalassa montenzumae Mulsant. After exposure to different regimes of contact pyrethroids and systemic neonicotinoid insecticides, Kanga et al. (2016) recommend use of the ladybird as a biocontrol agent for croton scales four days following an insecticide application. Evaluation of the effectiveness of insecticides for croton scale management has not been thoroughly investigated. It is important to correctly identify the scale insect pest to ensure you are using an effective biological control organism and choosing an appropriate chemical treatment. More research is needed in this area to determine best integrated pest management for the croton scale.
Selected References
Amarasekare KG, Mannion CM. 2011. Life history of an exotic soft scale insect Phalacrococcus howertoni (Hemiptera: Coccidae) found in Florida. Florida Entomologist 94: 588-593.
Etienne J, Matile-Ferrero D, Kondo T. 2014. Phalacrococcus howertoni Hodges & Hodgson (Hemiptera: Coccoidea: Coccidae), a new soft scale record for the island of Guadeloupe. Ciencia y Tecnología Agropecuaria 15: 115-118.
Hodges G. 2008. A new exotic soft scale insect on croton in south Florida (Hemiptera: Coccoidea: Coccidae). Pest Alert P-01682. FDACS—Division of Plant Industry. Gainesville, Florida. https://ccmedia.fdacs.gov/content/download/68157/file/Phalacrococcushowertoni_1682.pdf
Hodges GS, Hodgson CJ. 2010. Phalacrococcus howertoni, a new genus and species of soft scale (Hemiptera: Coccidae) from Florida. Florida Entomologist 93: 8-23.
Kanga L, Francis N, Mannion C, Haseeb M. 2016. Biology and effects of selected pesticides on a predatory beetle, Thalassa montenzumae, a potential biocontrol of croton scale, Phalacrococcus howertoni, in south Florida. Institut National de la Recherche Agronomique. Poster, Session S1—Système de production agroécologiques, visions et pratiques des acteurs.
Williams ML. 2010. Annotated list of the scale insects of Guatemala. Entomologia Hellenica 19: 144-152.