This publication describes the biology, damage symptoms, and methods of control for the citrus red mite, Panonychus citri. This species is becoming a pest of concern on citrus grown under protective screen (also called “CUPS”) and is a commonly observed pest in commercial and dooryard citrus. The target audience includes growers, Extension agents, crop consultants, state and local agencies, and interested gardeners.
Introduction
The citrus red mite, Panonychus citri Kuwayama (McGregor) (Figure 1), is a cosmopolitan polyphagous species reported on 113 hosts but mainly found on Citrus spp. (Migeon and Dorkeld 2011; Vacante and Gerson 2012). In Florida, P. citri is a pest of economic importance in open citrus orchards (Qureshi et al. 2021). The mite is also a major pest in Citrus Under Protective Screen (CUPS), a new undercover production system tested in Florida (Demard 2022; Ferrarezi et al. 2019). Environmental conditions inside CUPS are different from those in open-air systems due to the screen used to exclude the Asian citrus psyllid, Diaphorina citri, vector of huanglongbing (HLB), or citrus greening disease. The mean temperatures inside CUPS systems are 3°C to 4.5°C higher in summer and 1.5°C higher in winter than mean temperatures in open-air systems (Schumann and Waldo 2016). Also, humidity ranges average from 70%–80% in CUPS; humidity ranges from 70-80% in CUPS while humidity ranges from 60-70% in outdoor systems (Demard 2022). Although the literature states that high temperature and humidity are detrimental to P. citri, it seems that CUPS provides suitable conditions for the pest to develop (Demard 2022).

Credit: Matt Bertone, North Carolina State University
Immatures and adults feed on citrus leaves causing silvery spots called stippling injuries. At high densities, mites feeding on leaves, fruits, and green twigs result in mesophyll collapse, shoot dieback, and leaf and fruit drop (Albrigo et al. 1981; Qureshi et al. 2021).
Distribution
Panonychus citri has a worldwide distribution (Zhang 2003) (Figure 2). The mite is present in citrus-growing regions, mostly along the coasts. It reaches levels of economic importance in areas such as the Mediterranean region (Israel, Turkey, and Spain), the USA (Florida, Alabama, and California), and China.
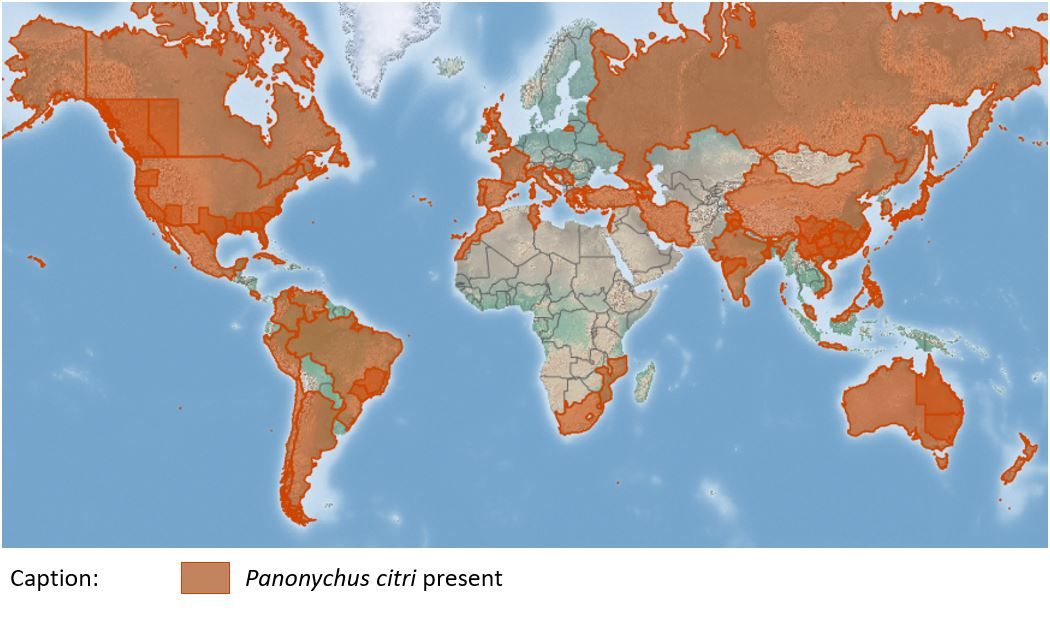
Credit: https://www.cabi.org/isc/datasheet/33682#toDistributionMaps
Host Range
Panonychus citri is reported from over 111 species of host plants, including almonds, pears, castor beans, grapes, palms, broadleaf ornamentals, and evergreen shrubs (Zhang 2003; Vacante 2010; Hoy 2011). Also, Panonychus citri is found on a variety of citrus. The mite reaches high populations on lemon and grapefruit followed by orange and tangerine (Childers and Fasulo 1995; Fleschner 1958). Troyer citrange is more susceptible than other varieties in California (Childers and Fasulo 1995).
Description
Eggs
Eggs are pale to red, small in diameter (0.13–0.16 mm), and almost spherical but flattened on the bottom (Figure 3) (Zhang 2003).

Credit: Emilie Demard, UF/IFAS
Larvae and Nymphs
Larvae have only three pairs of legs while nymphs have four pairs of legs. There are two nymphal stages called the protonymph (second instar) and deutonymph (third instar). Nymphs resemble adults but are smaller (Figure 1). Between each molt, the mite becomes inactive to shed its old cuticle and move into the next stage. This stage is called the chrysalis stage and is associated with the thin white exuvia (molted skin of arthropods). Numerous exuviae can be seen on the underside of the leaf under high infestations as each mite will produce three exuviae during its lifespan (Figure 1) (Childers and Fasulo 1995).
Adults
Adults are dark red to purplish. Males are smaller and lighter in color compared to females (Figure 4) (Vacante 2010). Males have a slender and elongated abdomen bearing long legs while females have a rounded abdomen with the front legs shorter than the body. The aedeagus (the reproductive organ of male arthropods) is sigmoid, with a broad base. The distal end turns upward (Figure 5B) (Vacante 1985).
Females are about 0.4 mm long (Figure 4B) (Vacante 2010) and have 13 pairs of dorsal setae (stiff hairs or bristles found in arthropods) (Figure 5A) arising from strong tubercles (small projections at the base of the setae) (Zhang 2003; Vacante and Gerson 2012). Their tarsi (the “foot” or last segment of a mite leg) have claw-like empodia (lobes between the claws of the tarsi of some arthropods) with three pairs of hairs (Figure 5C) (Vacante and Gerson 2012).

Credit: Emilie Demard, UF/IFAS
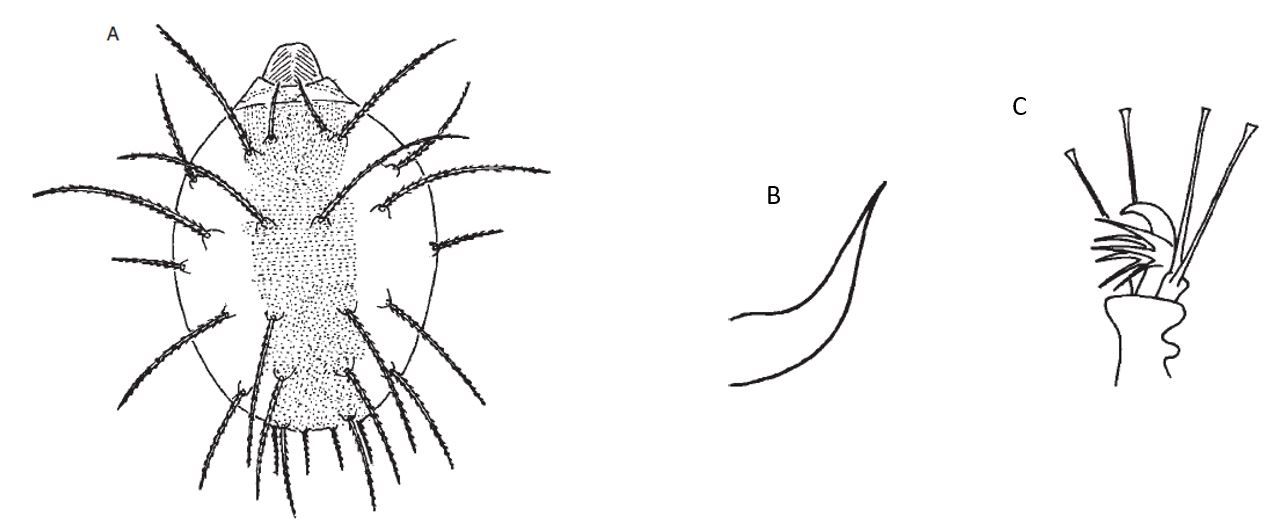
Credit: Vicenzo Vacante
Panonychus citri is morphologically similar to P. ulmi (Koch), the European red mite, except for the color of the tubercles, which are red in the former and white in the latter (Figure 6).

Credit: Matt Bertone, North Carolina State University and IRAC (available online at: https://www.irac-online.org/pests/panonychus-ulmi/)
Life Cycle
Females lay between 17 to 37 eggs over a period of 11 to 14 days (Childers and Fasulo 1995). At 24°C and 56% relative humidity (RH), eggs hatch in seven to nine days. The larval and the protonymphal stages live for two days each and the deutonymphal stage lasts between one to three days (Vacante 2010). Feeding starts in the protonymph stage. Females take an average of 12 days to develop from egg to adult, while males take 11 days when reared between 24°C–26°C and 50% to 70% RH. The lifespan of adults is about 23 days at 24°C–25°C (Childers and Fasulo 1995). Relative humidity of 65% is optimal for mite development and reproduction (Zhang 2003; Vacante 2010).
Panonychus citri cannot develop under 10°C or above 40°C (Zhang 2003). It is susceptible to high temperatures (all stages die above 40°C) and very dry conditions (RH around 5%). High humidity will also reduce their population growth rate (Childers and Fasulo 1995). The susceptibility of the mite to extreme temperature and ranges of high or low humidity often causes the population to decline during summer (Zhang 2003; Vacante and Gerson 2012). In Florida, the citrus red mite occurs throughout the year but is most abundant during dry conditions in spring or early summer (March–June) and fall or early winter (September–December) (Jeppson 1978; Qureshi et al. 2021). Population of red mites can increase 8.5 times in 10 days under low humidity conditions (Childers and Fasulo 1995).
In the absence of males, females reproduce through arrhenotokous parthenogenesis, which means that virgin females lay unfertilized eggs that will develop into males only. When males are present, sexual reproduction occurs and mated females produce eggs that will develop into both sexes (Vacante 2010).
Dispersal
Dispersal occurs by wind, and mites float on air streams by holding on to silk threads. The phenomenon is called “ballooning” (Vacante and Gerson 2012). The mite can also be dispersed mechanically by humans during crop maintenance and harvesting activities (Vacante 2010). In a field study, Tashiro (1966) investigated the intra-tree movement of P. citri and noticed that adult females moved preferentially upward from the release point rather than horizontally or downward. Adult female maximum migration was 4 feet in a day, and dispersal increased two to three times around 70°F (21°C) compared with 50°F (10°C) (Tashiro 1966).
Economic importance
Panonychus citri is a serious pest of citrus in California, South Africa, Japan, and around the Mediterranean basin, but it is also commonly found in Florida, South America, and India (Hoy 2011). The species occurs on leaves, twigs, and fruit. Heavy infestations result in leaf- and fruit drops, twig dieback, and low-quality fruit (Vacante 2010; Vacante and Gerson 2012). Yield reduction of 17%–41% in “Tahiti” lime relative to control was observed in Florida among four pesticide treatment programs (Childers and Abou-Setta 1999).
Damage
Leaves
Panonychus citri prefers to feed on the upper surface of young and middle-aged leaves, and it is generally located in the exterior canopy (Vacante 2010; Qureshi et al. 2021). All motile stages first orient along the mid-vein, then move to leaf margins when populations increase (Qureshi et al. 2021). Panonychus citri uses its piercing-sucking mouthpart to remove cytoplasm contents from plant cells except for some starch grains (Albrigo and McCoy 1974). This causes grey to yellow areas called stippling spots (Figure 7A), and under high infestations, these lesions merge to give the leaf a silvery appearance (Figure 7B). The loss of the photosynthetic tissues and mesophyll collapse may result in leaf fall.

Credit: Emilie Demard, UF/IFAS
Fruits
Damage to fruit may occur when mite population pressure is high, and mites start to colonize fruit. Immature fruit attacked by mites turn pale, but fruit will eventually color and ripen, while damaged mature fruit contains permanent straw yellow markings (Vacante 2010). The negative effects of mite feeding on the inner quality of the fruit are unknown; however, significant damage can cause fruit drop and yield loss.
Sampling Method
Numerous sampling methods have been studied around the world (Vacante 2010). In Florida, Qureshi et al. (2021) suggested a sampling method that provides 25% or less error margins when the motile mite densities (i.e., all stages except eggs) of P. citri and other spider mites are above two mites per leaf. Four or eight mature leaves per tree are examined every week or every two weeks. When examining one tree/acre, it is necessary to look at over 10 sample areas within a 10-acre block to achieve accuracy. With five or 10 trees/area, only four or five areas need to be examined. The number of samples declines as the mite densities increase. This method also is useful to determine a relationship between the average number of mites on the leaf and the percentage of leaves infested within a 10-acre block. For example, it helped in determining the treatment threshold for processed fruits, which is an average of five motile spider mites per leaf corresponding to an infestation level of 70%–80%. In California, the citrus red mite populations have been monitored by a presence–absence sampling protocol (Jones and Parrella 1984; Zalom et al. 1986).
Management
Chemical Control
The recommended chemical control of mites includes the use of abamectin, fenbutatin oxide, propargite, diflubenzuron, pyridaben, tolfenpyrad, spirodiclofen, spirotetramat, and some sulfur products (Qureshi et al. 2021). To avoid development of resistance, no miticide except petroleum oil should be used more than once a year. In China, P. citri was found to be resistant to abamectin, pyridaben, and dicofol (Meng et al. 2000; Marčić 2012). In California, resistance to demeton and parathion was demonstrated by Jeppson et al. (1958). Because resistance to spirotetramat and spirodiclofen occurred rapidly in China, bioassay methods were established to detect resistant populations of P. citri in San Joaquin Valley in California (Ouyang et al. 2012). However, no report of resistance has been documented yet.
More details can be found in the Florida Citrus Pest Management Guide: Ch. 10 Rust Mites, Spider Mites, and Other Phytophagous Mites: https://edis.ifas.ufl.edu/cg002.
Biological Control
Several predatory insects, mites, and pathogens (bacteria and fungi) have been shown to attack P. citri. Some common and abundant species are listed in Table 1. Multiple literature reviews also provide an exhaustive list of the natural enemies reported for P. citri (Van der Geest et al. 2000; Vacante 2010; Carrillo et al. 2015).
Table 1. Natural enemies of Panonychus citri (McGregor).
Numerous studies have evaluated the potential of predatory mites from the family Phytoseiidae as biological control agents of the citrus red mite. For instance, in Spain, the predatory mite Euseius stipulatus (Athias-Henriot) is a key natural enemy of P. citri maintaining the pest population under satisfactory control (Ferragut et al. 1992). This species has been released in California citrus orchards and is produced commercially in Peru for the control of P. citri (Aguirre-Gil et al. 2013). Euseius tularensis is a common predator of the citrus red mite in California. Fadamiro et al. (2013) showed that three commercial species of predatory mites, Galendromus occidentalis (Nesbitt), Phytoseiulus persimilis Athias-Henriot, and Neoseiulus californicus (McGregor) were effective in reducing P. citri populations in Alabama groves. In Florida, Typhlodromalus peregrinus (Muma) is an abundant phytoseiid found in Florida citrus grove and CUPS. While this species can develop and feed on P. citri, it did not provide good control under field conditions (Fouly et al. 1995, personal observation).
Cultural Control
Drought-stressed trees develop unhealthy root systems and become susceptible to P. citri damage (Jeppson et al.1975). Indeed, high transpiration results in low leaf moisture leading to leaf drop and twig dieback. Hence, adequate irrigation may reduce tree susceptibility to mite damage.
References
Aguirre-Gil, O. J., S. J. Jorge, and A. C. Busoli. 2013. “Control biologico classic na America Latina: o caso do Peru.” In: Busoli AC, de Alencar JRCC, Fraga DF, de Souza LA, de Souza BHS, Grigolli JFJ (eds) Tópicos em entomologia agrícola, vol IV. Gráfi ca Multipress, Jaboticabal-SP, p 66–75.
Albrigo, L. G., C. C. Childers, and J. P. Syvertsen. 1981. “Structural Damage to Citrus Leaves from Spider Mite Feeding.” Proc. Int. Soc. Citricult. Tokyo 2: 649Ð52.
Albrigo, L. G., and C. W. McCoy. 1974. “Characteristic Injury by Citrus Rust Mite to Orange Leaves and Fruit.” Proceedings of the Florida State Horticultural Society 87:48–55.
Bailey, S. F. 1939. “The Six-Spotted Thrips, Scolothrips sexmaculatus (Perg.).” Journal of Economic Entomology 32:43–47.
Carrillo, D., G. J. Moraes, and J. P. Peña. 2015. “Prospects for Biological Control of Plant Feeding Mites and Other Harmful Organisms.” Progress in Biological Control. Springer, Basel.
Childers, C. C., and T. R. Fasulo. 1995. Citrus Red Mite. Fact Sheet ENY-817. Available: https://ufdc.ufl.edu/IR00004619/00001.
Childers, C. C., and M. M. Abou-Setta. 1999. “Yield Reduction in ‘Tahiti’ Lime from Panonychus citri Feeding Injury Following Different Pesticide Treatment Regimes and Impact on the Associated Predacious Mites.” Experimental and Applied Acarology 23 (10): 771–783.
Demard, E. 2022. “Management of Phytophagous Mites in Citrus Under Protective Screen (CUPS).” Doctoral dissertation. Entomology and Nematology. University of Florida, Gainesville, FL, pp. 233.
Fadamiro, H. Y., Y. Xiao, M. Nesbitt, and C. C. Childers. 2009. “Diversity and Seasonal Abundance of Predacious Mites in Alabama Satsuma Citrus.” Annals of the Entomological Society of America 102:617–628.
Fadamiro, H. Y., C. Akotsen-Mensah, Y. F. Xiao, and J. Anikwe. 2013. “Field Evaluation of Predacious Mites (Acari: Phytoseiidae) for Biological Control of Citrus Red Mite, Panonychus citri (Trombidiformes: Tetranychidae).” Florida Entomologist 96:80–91.
Ferragut, F., R. Laborda, J. Costa-Comelles, and F. García-Mari. 1992. « Feeding Behavior of Euseius stipulates and Typhlodromus phialatus on the Citrus Red Mite Panonychus citri (Acari: Phytoseiidae, Tetranychidae).” Entomophaga 37: 537–543.
Ferrarezi, R. S., J. A. Qureshi, A. L. Wright, M. A. Ritenour, and N. P. F. Macan. 2019. “Citrus Production under Screen as a Strategy to Protect Grapefruit Trees from Huanglongbing Disease.” Frontiers in Plant Science 10:1598–1598.
Fleschner, C. A. 1958. “Field Approach to Population Studies of Tetranychid Mites on Citrus and Avocado in California.” In: Proc. 10th International Congress Entomology, Montreal (Edited by E. C. Becker), Vol. 2, pp. 669–74. Mortimer, Ottawa.
Fouly, A. H., M. M. Abou-Setia, and C. C. Childers. 1995. “Effects of Diet on the Biology and Life Tables of Typhlodromalus peregrinus (Acari: Phytoseiidae).” Environmental Entomology 24:870–874.
Goldarazena, A., H. Aguilar, H. Kutuk, and C. C. Childers. 2004. “Biology of Three Species of Agistemus (Acari: Stigmaeidae): Life Table Parameters Using Eggs of Panonychus citri or Pollen of Malephora crocea as Food.” Experimental and Applied Acarology 32:281–291
Hall, I. M., D. K. Hunter, and K. Y. Arakawa. 1971. “The Effect of the b-exotoxin Fraction of Bacillus thuringiensis on the Citrus Red Mite.” Journal of Invertebrate Pathology 18:359–362.
Haney, P. B., R. F. Luck, and D. S. Moreno. 1987. “Increases in Densities of the Citrus Red Mite, Panonychus citri (Acarina: Tetranychidae), in Association with the Argentine Ant, Iridomyrmex humilis (Hymenoptera: Formicidae), in Southern California Citrus.” Entomophaga 32(1). https://doi.org/10.1007/BF02390931
Hoy, M. A. 2011. Agricultural Acarology: Introduction to Integrated Mite Management CRC Press, Boca Raton, Florida.
Huang, P. K., X. N. Luo, and S. L. Song. 1988. “Biological Control of Panonychus citri by Releasing Stethorus siphonulus Kapur (Coleoptera: Coccinellidae) in Southeastern China.” Acta Phytophylactica Sinica 15:1–6 (in Chinese).
Jeppson, L. R. 1978. “Bionomics and Control of Mites Attacking Citrus.” Proceedings of the International Society of Citriculture. Lake Alfred, FL. 2:441–451.
Jeppson, L. R., H. H. Keifer, and E. W. Baker. 1975. Mites Injuries to Economic Plants. University of California Press. Los Angeles.
Jeppson, L. R., M. J. Jesser, and J. O. Complin. 1958. « Resistance of the Citrus Red Mite to Organic Phosphates in California.” Journal of Economic Entomology 51:232–233.
Ji, J, T. Lin, Y. Zhang, J. Lin, L. Sun, and X. Chen. 2013. “A Comparison between Amblyseius (Typhlodromips) swirskii and Amblyseius eharai with Panonychus citri (Acari: Tetranychidae) as Prey: Developmental Duration, Life Table and Predation.” Systematic and Applied Acarology 18:123–129.
Jones, V. P., and M. P. Parrella. 1984. “Presence–Absence Sampling of Citrus Red Mite on Lemons.” California Agriculture 40 (3): 31–32.
Marčić, D. 2012. “Acaricides in Modern Management of Plant-Feeding Mites.” Journal of Pest Science 85:395–408.
McCoy, C. W. 1981. Pest control by the fungus Hirsutella thompsonii, in Microbial Control of Pests and Plant Diseases, 1970–1980 (Edited by H. D. Burges), Academic Press, London, UK, pp. 499–512.
McMurtry, J. A., C. G. Huffaker, and M. van den Vrie. 1970. Ecology of Tetranychid Mites and Their Natural Enemies: A Review. I. Tetranychid Enemies: Their Biological Characters and the Impact of Spray Practices vol. 40, Hilgardia.
Meng, H. S., K. Y. Wang, X. Y. Jiang, and M. Q. Yi. 2000. “Studies on the Resistance of Panonychus citri to Several Acaricides.” Pesticides 39:26–28 (in Chinese).
Migeon, A., and F. Dorkeld. 2011. Spider Mites Web: a comprehensive database for the Tetranychidae. Available from: https://www1.montpellier.inra.fr/CBGP/spmweb/ (Accessed 5/13/2020).
Muma, M. M. 1967. “Biological Notes on Coniopteryx vicina (Neuroptera: Coniopterygidae).” Florida Entomologist 50:285–293.
Ouyang, Y., G. H. Montez, L. Liu, and E. E. Grafton-Cardwell. 2012. “Spirodiclofen and Spirotetramat Bioassays for Monitoring Resistance in Citrus Red Mite, Panonychus citri (Acari: Tetranychidae).” Pest Management Science 68:781–787. https://doi.org/10.1002/ps.2326
Qureshi, J. A., L. L. Stelinski, X. Martini, and L. M. Diepenbrock. 2021. “2021–2022 Florida Citrus Production Guide: Rust Mites, Spider Mites, and Other Phytophagous Mites” CG002/ENY-603, Rev. 3/2021. EDIS 2021 (CPG). https://doi.org/10.32473/edis-cg002-2021
Tashiro, H. 1966. “Intratree Dispersal of the Citrus Red Mites, Panonychus citri (Acarina: Tetranychidae).” Annals of the Entomological Society of America 59:1206–1210.
Tian, M. Y. 1995. “Efficacy of Amblyseius nicholsi and Stethorus punctillum as Natural Enemies of Citrus Red Mite.” Journal of Biological Control 11:153.
Shi, W. B., and M. G. Feng. 2006. “Field Efficacy of Application of Beauveria bassiana Formulation and Low Rate Pyridaben for Sustainable Control of Citrus Red Mite Panonychus citri (Acari: Tetranychidae) in Orchards.” Biological Control 39:210–217.
Schumann, A. W., and L. Waldo. 2016. “CUPS Quick Start Guide.” In: Interim Sustainable Solutions for Fresh Citrus Fruit Production in Florida, edited by A. W. Schumann. UF/IFAS, Citrus Research and Education Center, Lake Alfred, FL, pp. 13.
Vacante, V. 1985. “Prima raccolta di acari Tetranichidi in Sicilia.” Phytophaga 1:1–114.
Vacante, V. 2010. Citrus Mites: Identification, Bionomy and Control CABI International, Wallingford, UK.
Vacante, V., and U. Gerson. 2012. “Acari” pp. 88–108. In: Integrated Control of Citrus Pests in the Mediterranean Region, edited by V. Vacante and U. Gerson. Bentham Science Publishers, Sharjah, UAE.
Van der Geest, L. P., S. L. Elliot, J. A. Breeuwer, and E. A. Beerling. 2000. “Diseases of Mites.” Experimental and Applied Acarology 24:497–560.
Yue, B. S., and J. H. Tsai. 1995. “Agistemus exsertus Gonzalez (Acari: Stigmaeidae) as a Predator of Citrus Red Mite (Panonychus citri [McGregor]).” Journal of the New York Entomological Society 103 (1): 107–113.
Zalom, F. G., L. T. Wilson, C. E. Kennett, N. V. O’Connell, D. L. Flaherty, and J. G. Morse. 1986. “Presence-Absence Sampling of Citrus Red Mite.” California Agriculture. 40 (3): 15–16.
Zhang, Z. Q. 2003. Mites of Greenhouses: Identification, Biology and Control CAB International, Wallingford, UK.
Zhou, C. A., J. J. Zou, J. C. Peng, Z. Y. Ouyang, L. C. Hu, Z. L. Yang, and X. B. Wang. 1991. “Predation of Major Natural Enemies on Panonychus citri and Its Comprehensive Evaluation in Citrus Orchards in Hunan, China.” Acta Phytophylacica Sinica 18 (3): 225–229.