The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Pseudomyrmex simplex is an arboreal ant species that is usually found in hollow twigs or vines, but rarely in grass culms or herbs like many other ants in this group. The species name simplex is derived from Latin, meaning “simple” or “plain,” which may be a reference to the singular black spot on either side of the gaster (the bulbous portion of the abdomen behind the two-humped petiole), or the lack of conspicuous setae on the first segment of the gaster, which most other members of the genus Pseudomyrmex have (Deyrup, 2017).

Credit: April Nobile, AntWeb.org
Synonymy
Pseudomyrma simplex F. Smith, 1877
Pseudomyrma delicatula Forel, 1899
Pseudomyrma delicatula capperi Forel, 1899
Pseudomyrma delicatula panamensis Forel, 1899
Pseudomyrma vittatus Forel, 1912
Distribution
Pseudomyrmex simplex is thought to be native to the Greater Antilles (and potentially Florida), because subfossil specimens were found encased in amber from the Dominican Republic (Ward, 1992). It is a common and widely distributed species, ranging from Florida south to Argentina.
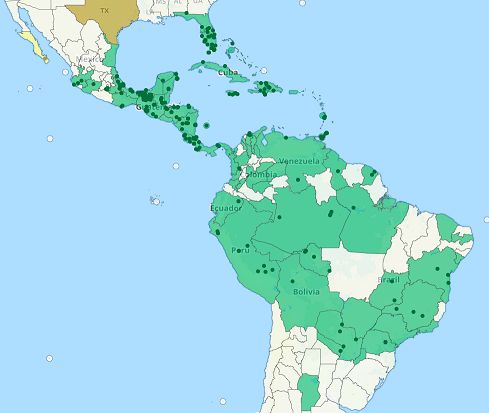
Credit: Map reproduced with permission from antmaps.org
Description
Pseudomyrmex simplex is the smallest of a group (referred to as the pallidus group) containing three other similar yellow-orange species, Pseudomyrmex leptosus, Pseudomyrmex pallidus, and Pseudomyrmex seminole. These species are difficult to tell apart and can all be found in Florida, making them more difficult to identify correctly. The differences between these species are small physical characteristics that typically cannot be seen by the naked eye.
Pseudomyrmex simplex in Florida always has a pair of brownish-black spots on the gaster. It also tends to be lighter in color, with a smooth, shiny head and shiny first gastral segment that lacks dense, fine pubescence that other species in the pallidus group have.

Credit: Gracen Brilmyer, AntWeb.org
Biology
Pseudomyrmex simplex has a strong preference for nesting in the dead twigs of woody shrubs or trees, rather than dead stalks of herbaceous plants that other species in the pallidus group prefer. Stem-dwelling ants in this group generally avoid nesting in stems that are beginning to rot or are easily soaked by rains, and they prefer to nest in twigs with very small inner diameters (roughly 2.2 millimeters ± 0.7 mm) (Carroll, 1979). Carroll (1979) also suggests that stem-dwelling ants in this group (such as Pseudomyrmex simplex) might prefer nests with narrow inner diameters because they offer protection against dehydration and larger-bodied, aggressive species that may try to displace them from their stems.
There is no specific information on the feeding habits of Pseudomyrmex simplex. However, it is noted that the feeding habits of adults in the subfamily are remarkably uniform, such that they are omnivorous and typically feed on honeydew, soft plant tissue, fungus, and the soft tissues of other insects (Creighton, 1950). Adults have a specialized process for feeding larvae which is unique to the ants in this subfamily, but not known elsewhere in the Formicidae family (Wheeler & Bailey, 1920). The adult ants will take food that they have previously collected and stored in their infrabuccal pocket, consume part of it, and deposit the remainder of the dried food “pellet” into the larva’s infrabuccal pocket in their trophothylax (an enlarged pocket behind the mouth). Creighton (1950) indicated that in all other ants, the contents of the infrabuccal pocket are typically discarded.
Two or more species in the pallidus group can co-occur in a broadly overlapping array of nesting sites. While they do not share individual nests, they can be found in very close proximity to each other, sometimes separated by only a few meters. Most nests of Pseudomyrmex simplex are queenless, which indicates that this species could be polydomous (where a single ant colony occupies two or more spatially separated nests) (Ward, 1985).
Hosts and Host Plants
Pseudomyrmex simplex is an associated prey species for larvae of the syrphid fly, Rhopalosyrphus ramulorum, which feed on ant brood. (Quevillon, 2020; Weems et al., 2003).
Pseudomyrmex simplex is an associated predator species of both free and berry-embedded adult coffee berry borer beetles, Hypothenemus hampei (Morris et al., 2018).
In Florida and Costa Rica, nests of Pseudomyrmex simplex have been collected from the dead twigs and stalks of Anacardium occidentale (cashew), Ardisia revoluta (ardisia, tecuico), Avicennia germinans (black mangrove), Baccharis halimifolia (groundseltree), Carya floridana (scrub hickory), Cladium jamaicense (saw-grass), Conocarpus erectus (button mangrove), Gliricidia sepium (gliricidia), Hibiscus tiliaceus (coast cottonwood), Laguncularia racemose (white mangrove), Metopium toxiferum (poisonwood), Nectandra coriacea (lancewood), Swietenia mahagoni (West Indian mahogany), and Terminalia catappa (West Indian-almond) (Ward, 1985).
Economic Importance
Studies by Larsen and Philpott (2010) on coffee farms in Mexico suggest that Pseudomyrmex simplex can be an effective predator of the coffee berry borer beetle, Hypothenemus hampei (Coleoptera: Curculionidae), which burrows into coffee fruits, damaging them. Pseudomyrmex simplex functions as one of several species of twig-nesting ants that are viewed as important, naturally occurring biological control agents of the coffee berry borer (Larsen & Philpott, 2010). Additionally, the small, slender body of Pseudomyrmex simplex is advantageous for biological control of these berry borers, because they can follow the beetles into the small holes, unlike larger-bodied ants.
Selected References
Carroll RC. 1979. A comparative study of two ant faunas: the stem-nesting ant communities of Liberia, West Africa and Costa Rica, Central America. The American Naturalist 113: 551-561. https://doi.org/10.1086/283412
Creighton WS. 1950. The ants of North America. Bulletin of the Museum of Comparative Zoology at Harvard College, 691 pp. Retrieved from https://archive.org/details/ants_06224/6224/page/4/mode/1up?view=theater&q=pseudomyrmex
Deyrup M. 2017. Ants of Florida: identification and natural history. Boca Raton: Taylor & Francis Group, LLC. https://doi.org/10.1201/9781315368023
Guénard B, Weiser M, Gomez K, Narula N, Economo E. 2017. The Global Ant Biodiversity Informatics (GABI) database: a synthesis of ant species geographic distributions. Myrmecological News 24: 83-89.
Janicki J, Narula N, Ziegler M, Guénard B, Economo E. 2016. Visualizing and interacting with large-volume biodiversity data using client-server web-mapping applications: The design and implementations of antmaps.org. Ecological Informatics 32: 185-193. https://doi.org/10.1016/j.ecoinf.2016.02.006
Larsen A, Philpott SM. 2010. Twig-nesting ants: the hidden predators of the coffee berry borer in Chiapas, Mexico. Biotropica 43: 342-347. doi:10.1111/j.1744-7429.2009.00603.x
Morris JR, Jiménez-Soto E, Philpott SM, Perfecto I. 2018. Ant-mediated (Hymenoptera: Formicidae) biological control of the coffee berry borer: diversity, ecological complexity, and conservation biocontrol. Myrmecological News 26: 1-17. Retrieved from https://escholarship.org/uc/item/4jq1d8hj
Quevillon LE. 2020. The ecology, epidemiology, and evolution of parasites infecting ants (Hymenoptera: Formicidae) (Order No. 28281200). ProQuest Dissertations & Theses Global, 399 pp. Retrieved from https://www.proquest.com/dissertations-theses/ecology-epidemiology-evolution-parasites/docview/2459435356/se-2?accountid=10920
Ward PS. 1985. The Nearctic species of the genus Pseudomyrmex (Hymenoptera: Formicidae). Quaestiones Entomologicae 21: 209-246.
Ward PS. 1992. Ants of the genus Pseudomyrmex (Hymenoptera: Formicidae) from Dominican Amber, with a synopsis of the extant Antillean species. Psyche 99: 55-85. https://doi.org/10.1155/1992/19146
Weems HV, Thompson FC, Rotheray G, Deyrup MA. 2003. The Genus Rhopalosyrphus (Diptera: Syrphidae). Florida Entomologist 86: 186-193. https://doi.org/10.1653/0015-4040(2003)086[0186:TGRDS]2.0.CO;2
Wheeler W, Bailey I. 1920. The feeding habits of Pseudomyrmines and other ants. Transactions of the American Philosophical Society 22: 235-279. doi:10.2307/1005485