The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Sweetpotato weevil is the most serious pest of sweet potato, not only in the United States, but around the world. It causes damage in the field, in storage, and is of quarantine significance. It is inherently of interest to entomologists due to its strikingly colorful appearance and extremely long rostrum (beak).
Distribution
Sweetpotato weevil was first noted in the United States in Louisiana in 1875, and then in Florida in 1878 and Texas in 1890, probably entering by way of Cuba. It is now found throughout the coastal plain of the Southeast from North Carolina to Texas. It also is found in Hawaii and Puerto Rico, and widely around the world in tropical regions.
Life Cycle and Description
A complete life cycle requires one to two months, with 35 to 40 days being common during the summer months. The generations are indistinct, and the number of generations occurring annually is estimated to be five in Texas, and at least eight in Louisiana. Adults do not undergo a period of diapause in the winter but seek shelter and remain inactive until the weather is favorable. All stages can be found throughout the year if suitable host material is available.
Egg
Eggs are deposited in small cavities created by the female with her mouthparts in the sweet potato root or stem. The female deposits a single egg at a time and seals the egg within the oviposition cavity with a plug of fecal material, making it difficult to observe the egg. Most eggs tend to be deposited near the juncture of the stem and root (tuber). Sometimes the adult will crawl down cracks in the soil to access tubers for oviposition, in preference to depositing eggs in stem tissue. The egg is oval in shape and creamy white in color. Its size is reported to be about 0.7 mm (0.03 in) in length and 0.5 mm (0.02 in) in width. Duration of the egg stage varies from about five to six days during the summer to about 11 to 12 days during colder weather. Females apparently produce two to four eggs per day, or 75 to 90 eggs during their life span of about 30 days. Under laboratory conditions, however, mean fecundity of 122 and 50 to 250 eggs per female has been reported.
Larva
When the egg hatches the larva usually burrows directly into the tuber or stem of the plant. Those hatching in the stem usually burrow down into the tuber. The larva (Figure 1) is legless, white in color, and displays three instars. The mean head capsule widths of the instars are 0.29 to 0.32 mm (0.011 to 0.012 in), 0.43 to 0.49 mm (0.017 to 0.019 in), and 0.75 to 0.78 mm (0.029 to 0.031 in) for instars 1 to 3, respectively. Duration of each instar is 8 to 16, 12 to 21, and 35 to 56 days, respectively. Temperature is the principal factor affecting larval development rate, with larval development (not including the prepupal period) occurring in about 10 and 35 days at 30°C (86°F) and 24°C (75.2°F), respectively. The larva creates winding tunnels packed with fecal material as it feeds and grows.

Credit: James Castner, UF/IFAS
Pupa
The mature larva creates a small pupal chamber in the tuber or stem. The pupa is similar to the adult in appearance, although the head and elytra are bent ventrally. The pupa measures about 6.5 mm (0.26 in) in length. Initially the pupa is white, but with time this stage becomes grayish in color with darker eyes and legs. Duration of the pupal stage averages 7 to 10 days, but in cool weather it may be extended to up to 28 days.
Adult
Normally the adult emerges from the pupation site by chewing a hole through the exterior of the plant tissue, but sometimes it remains for a considerable period and feeds within the tuber. The adult (Figure 2) is striking in form and color. The body, legs, and head are long and thin, giving it an ant-like appearance. The head is black, the antennae, thorax and legs orange to reddish brown, and the abdomen and elytra are metallic blue. The snout is slightly curved and about as long as the thorax; the antennae are attached at about the mid point on the snout. The beetle appears smooth and shiny, but close examination shows a layer of short hairs. The adult measures 5.5 to 8.0 mm in length. Under laboratory conditions at 15°C (59°F), adults can live over 200 days if provided with food and about 30 days if starved. In contrast, their longevity decreases to about three months if held at 30°C (86°F) with food, and eight days without food. Adults are secretive, often feeding on the lower surface of leaves, and are not readily noticed. The adult is quick to feign death if disturbed. Adults can fly, but seem to do so rarely and in short, low flights. However, because they are active mostly at night, their dispersive abilities are probably underestimated. Females feed for a day or more before becoming sexually active but commence oviposition shortly after mating; the average preoviposition period is seven days. A sex pheromone produced by females has been identified and synthesized.

Credit: James Castner, UF/IFAS
Host Plants
This weevil feeds on plants in the plant family Convolvulaceae. Although it has been found associated with several genera, its primary hosts are in the genus Ipomoea. Among vegetable crops, only sweet potato, Ipomoea batatas, is a suitable host. Native plants can be important hosts of sweetpotato weevil. Railroad vine, Ipomoea pescaprae, and morning glory, Ipomoea panduratea, are among the suitable wild hosts.
Natural Enemies
Several natural enemies are known. Wasps such as Bracon mellitor Say, Bracon punctatus (Muesebeck), Metapelma spectabile Westwood (all Hymenoptera: Braconidae) and Euderus purpureas Yoshimoto (Hymenoptera: Eulophidae) have been reared from sweetpotato weevil larvae in the southeastern United States. There have been no studies of parasitoid effectiveness, but these species seem to be infrequent. Among predators, ants (Hymenoptera: Formicidae) seem to be most important. Diseases, especially the fungus Beauveria bassiana, have been observed to inflict high levels of mortality under conditions of high humidity and high insect density, but field conditions are rarely conducive for disease epizootics.
Damage
Sweetpotato weevil is often considered to be the most serious pest of sweet potato, with reports of losses ranging from 5% to 97% in areas where the weevil occurs. There is a positive relationship between vine damage or weevil density, and tuber damage. However, the plants exhibited some compensatory ability, with the relationship between vine damage and yield non-linear, and sometimes not significant.
A symptom of infestation by sweetpotato weevil is yellowing of the vines, but a heavy infestation is usually necessary before this is apparent. Thus, incipient problems are easily overlooked, and damage not apparent until tubers are harvested. The principal form of damage to sweet potato is mining of the tubers by larvae (Figure 3). The infested tuber is often riddled with cavities, spongy in appearance, and dark in color. In addition to damage caused directly by tunneling, larvae cause damage indirectly by facilitating entry of soil-borne pathogens. Even low levels of feeding induce a chemical reaction that imparts a bitter taste and terpene odor to the tubers. Larvae also mine the vine of the plant, causing it to darken, crack, or collapse. The adult may feed on the tubers, creating numerous small holes that measure about the length of its head. The adult generally has limited access to the tubers, however, so damage by this stage is less severe than by larvae. Adult feeding on the foliage seldom is of consequence.
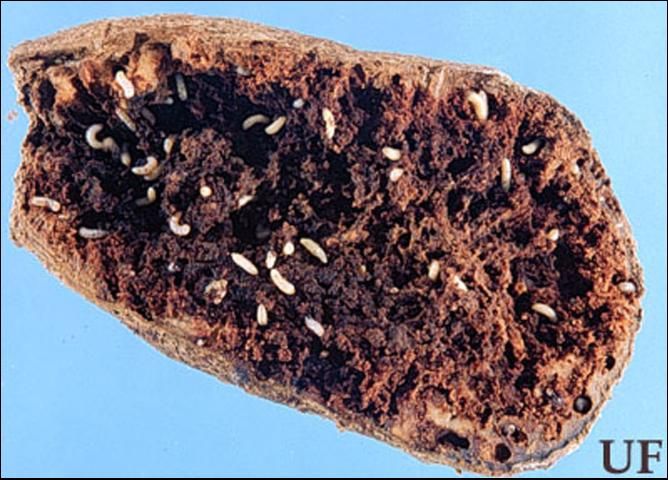
Credit: James Castner, UF/IFAS
Management
Sampling
Most of larvae are found in the upper 15 cm of the soil and the basal 15 cm of the of the vine, though they can be located elsewhere along the vine. Early in the season larvae are found about equally in the vine and tuber, but later in the season most occur in the tubers. Distribution of sweetpotato weevil in fields is aggregated.
Pheromone traps show great promise for monitoring of adult population density. Weevils respond to low concentrations of pheromone and apparently will move up to 280 m to a pheromone source. The sex pheromone also shows great potential for mating disruption and mass trapping.
Insecticides
The slips or cutting used to plant the crop should be free of weevils. This usually necessitates insecticidal treatment of the planting material. Especially if this is not done, but even if the slips are free of insects at planting, planting-time applications of insecticides are commonly made to the soil to prevent injury to the slips or cuttings by weevils present in the field. Either granular or liquid formulations are used, and systemic insecticides are preferred. Postplant applications are sometimes made to the foliage for adult control, especially if fields are likely to be invaded from adjacent areas, but if systemic insecticide is applied some suppression of larvae developing in the vine may also occur. Due to the long duration of the plant growth period, it is not uncommon for preplant or planting time applications to be followed by one or more insecticide applications to the plant or soil at mid season. Insecticides are also applied to tubers being placed into storage to prevent reinfestation and inoculation of nearby fields.
Cultural Practices
Cultural practices are sometimes recommended to alleviate weevil problem. Isolation is frequently recommended, and it is advisable to locate new fields away from previous crops and distant from sweet potato storage facilities, because both can be a source of new infestations. However, despite the infrequency of flight by adults, dispersal can occur over considerable distances. Dispersal rates of 150 m per day have been observed, with dispersal more rapid in the absence of suitable hosts. Even if the sweetpotato field cannot be moved a long distance between cropping cycles, field rotation is beneficial.
Sanitation is particularly important for weevil population management. Discarded tubers and unharvested tubers can support large population, and every effort should be made to remove such host material. Related to this, of course, is the destruction of alternate hosts; control of Ipomoea weeds is recommended.
Dry soil leads to cracking of the soil, which is a favorable environment for weevils. Thus, maintenance of moist soil with irrigation is helpful for weevil management. It also favors development of fungal diseases of weevils.
Biological Control
Entomopathogenic nematodes seem to have potential for practical biological suppression of sweetpotato weevil. Several strains of Steinernema carpocapsae (Nematoda: Steinernematidae) and Heterorhabditis bacteriophora (Nematoda: Heterorhabditidae) penetrate the soil and tubers, killing weevil larvae. At least in the soils of southern Florida, the infective nematodes are persistent, remaining active for up to four months. In some cases, nematodes are more effective than insecticides at reducing damage. Strains of the fungus Beauveria bassiana that are pathogenic to sweetpotato weevil have been identified and can be effective is there is adequate soil moisture. In Cuba, predatory ants have been manipulated to the benefit of weevil management by transporting the ant nests temporarily to sweetpotato fields.
Other Methods
Other methods of suppression are sometimes used, especially for postharvest treatment of tubers. Postharvest treatment not only prevents damage in storage but allows shipment of tubers to areas where sweetpotato weevil is not found but might survive. Traditionally, postharvest treatment has been accomplished with chemical fumigants, but they have fallen from favor. Irradiation is potentially effective, although older stages of insects are less susceptible to destruction. Storage in controlled atmospheres, principally low oxygen and high carbon dioxide, is very effective for destruction of weevils, but requires good storage conditions.
Some varieties of sweet potato have shown slight indications of resistance to sweetpotato weevil. However, effective resistance has not been identified despite numerous attempts to breed resistance into sweet potatoes.
The effectiveness of the pheromone trap has let to experimentation of traps that auto-infect with Beauveria bassiana fungus when the male weevils visit the trap.
Selected References
Cockerham KL, Deen OT, Christian MB, Newsom LD. 1954. "The biology of the sweet potato weevil." Louisiana Agricultural Experiment Station Techincal Bulletin 483. 30 pp.
Jansson RK. 1991. Biological control of Cylas spp. Pages 169-201. In Jansson RK, Raman KV (editors). Sweet Potato Pest Management: A Global Perspective. Boulder, CO: Westview Press.
Jansson RK, Lecrone SH, Gaugler R. 1993. "Field efficacy and persistence of entomopathogenic nematodes (Rhabditida: Steinernematidae, Heterorhabditidae) for control of sweetpotato weevil (Coleoptera: Apionidae) in southern Florida." Journal of Economic Entomolology 86: 1055–1063.
Jansson RK, Mason LJ, Heath RR. 1991a. Use of sex pheromone for monitoring and managing Cylas formicarius. Pages 97–138. In R.K. Jansson and K.V Raman (eds.). Sweet Potato Pest Management: A Global Perspective. Boulder CO: Westview Press.
Jansson RK, Hunsberger AGB, Lecrone SH, O'Hair SK. 1990a. "Seasonal abundance, population growth, and within-plant distribution of sweetpotato weevil (Coleoptera: Curculionidae) on sweet potato in southern Florida." Environomental Entomology 19: 313–321.
Jansson RK, Lecrone SH, Gaugler RR, Smart Jr GC. 1990b. "Potential of entomopathogenic nematodes as biological control agents of sweetpotato weevil Coleoptera: Curculionidae)." Journal of Economic Entomology 83: 1818–1826.
Jansson RK, Proshold FI, Mason LJ, Heath RR, Lecrone SH. 1990c. "Monitoring sweetpotato weevil (Coleoptera: Curculionidae) with sex pheromone: effects of dosage and age of septa." Tropical Pest Management 36: 263–269.
Korada RR, Naskar SK, Palaniswami, MS, Ray RC. 2010. "Management of sweet potato weevil [Cylas formicarius (Fab)]: an overview." Journal of Root Crops 36: 14–26.
Mullen MA, Jones A, Paterson DR, Boswell TE. 1985. "Resistance in sweet potatoes to the sweetpotato weevil, Cylas formicarius elegantulus (Summers)." Journal of Entomological Science 20: 345–350.
Mullen MA, Jones A, Arbogast RT, Schalk JM, Paterson DR, Boswell TE, Earhart DR. 1980. "Field selection of sweet potato lines and cultivars for resistance to the sweetpotato weevil." Journal of Economic Entomology 73: 288–290.
Reinhard HJ. 1923. "The sweet potato weevil." Texas Agricultural Experiment Station Bulletin 308. 90 pp.
Sharp JL. 1995. "Mortality of sweetpotato weevil (Coleoptera: Apionidae) stages exposed to gamma irradiation." Journal of Economic Entomology 88: 688–692.
Sherman M, Tamashiro M. 1954. The sweetpotato weevils in Hawaii, their biology and control. University of Hawaii Technical Bulletin 23. 36 pp.
Sutherland JA. 1986. "Damage by Cylas formicarius Fab. to sweet potato vines and tubers, and the effect of infestations on total yield in Papua New Guinea." Tropical Pest Management 32: 316–323.
Talekar NS. 1982. "Effects of a sweetpotato weevil (Coleoptera: Curculionidae) infestation on sweet potato root yields." Journal of Economic Entomology 75: 1042–1044.
Talekar NS. 1987. "Resistance in sweetpotato to sweetpotato weevil." Insect Science and its Application 8: 819–823.
Yasuda, K. 1999. "Auto-infection system for the sweet potato weevil, Cylas formicarius (Fabricius) (Coleoptera: Curclionidae) with entomopathogenic fungi Beauveria bassiana using a modified sex pheromone trap in the field." Applied Entomology and Zoology 34: 501–505.