The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The oriental fruit fly, Bactrocera dorsalis (Hendel), is a very destructive pest of fruit in areas where it occurs. It is native to large parts of tropical Asia, has become established over much of sub-Saharan Africa, and is often intercepted in the United States, sometimes triggering eradication programs.

Credit: Scott Bauer, USDA
Synonymy
Bactrocera dorsalis was formerly known as Dacus dorsalis. Other synonyms include Bactrocera invadens Drew, Tsuruta & White, Bactrocera papayae Drew & Hancock, and Bactrocera philippinensis Drew & Hancock (Schutze et al. 2015).
Distribution
Countries with established infestations include (CABI 2020; Vargas et al. 2007):
- Asia: Bangladesh, Bhutan, Cambodia, China (southern), Hong Kong, India (numerous states), Indonesia, Japan (Ryukyu Islands), Laos, Malaysia, Myanmar, Nepal, Ogasawara Islands, Pakistan, Philippines, Sri Lanka, Taiwan, Thailand, Vietnam
- Africa: most countries of sub-Saharan Africa have become infested since the first appearance of oriental fruit fly (as Bactrocera invadens) in Kenya in 2003 (see Goergen et al. 2011).
- Pacific Islands: Mariana Islands, Tahiti, Hawaii
In the United States, oriental fruit fly is currently present on all major Hawaiian islands after being accidentally introduced there in 1944 or 1945 (Mau 2007).
Elsewhere in the US, there are chronic detections in California and Florida that often trigger eradication programs.
Four major oriental fruit fly infestations in California were eradicated between 1960 and 1997. Additional infestations were detected in 2002 and 2004 and were eradicated in 2006 and 2007 respectively. In July 2010, fruit flies were discovered in traps in Sacramento and Placer Counties. A quarantine was established, and an eradication program began (CDFA 2010).
While not established in Florida, oriental fruit fly and relatives, such as Bactrocera correcta, are regularly trapped in this state. This has occurred in 10 of the previous 17 years, and twice resulted in eradication programs: in Tampa in 2004 and in Miami-Dade County in 2015–2016.
Description
Adult
The adult, which is noticeably larger than a house fly, has a body length of about 8.0 mm; the wing is about 7.3 mm in length and is mostly hyaline. The color of the fly is very variable, but there are prominent yellow and dark brown to black markings on the thorax. Generally, the abdomen has two horizontal black stripes and a longitudinal median stripe extending from the base of the third segment to the apex of the abdomen. These markings may form a T-shaped pattern, but the pattern varies considerably. The ovipositor is very slender and sharply pointed.
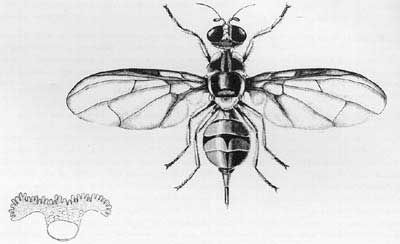

Credit: Okinawa Prefectural Fruit Fly Eradication Project Office

Credit: Scott Bauer, USDA
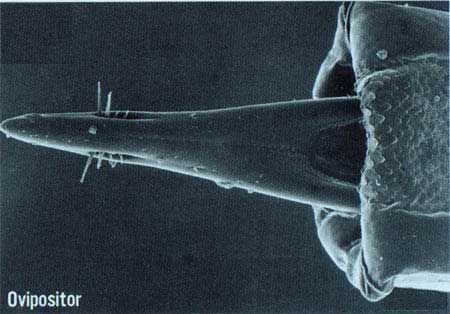
Credit: Okinawa Prefectural Fruit Fly Eradication Project Office
Egg
The white, elongate, and elliptical egg measures about 1.17 x 0.21 mm and has a chorion without sculpturing.

Credit: Okinawa Prefectural Fruit Fly Eradication Project Office
Larva (General Description)
The third-instar, which has a typical maggot appearance, is about 10 mm in length and creamy white. The only band of spinules encircling the body is found on the first segment. The external part of the anterior respiratory organs, the spiracles, located one on each side of the pointed or head end of the larva, has an exaggerated and deflexed lobe at each side and bears many small tubercles. The caudal segment is very smooth. The posterior spiracles are located in the dorsal third of the segment as viewed from the rear of the larva. The mature larva emerges from the fruit, drops to the ground, and forms a tan to dark brown puparium about 4.9 mm in length.
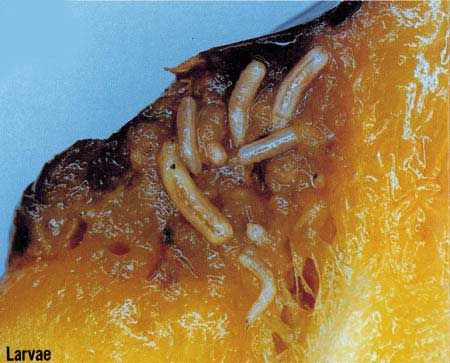
Credit: Okinawa Prefectural Fruit Fly Eradication Project Office
Larva (Scientific Description)
The larva of the oriental fruit fly is quite similar to that of the Mediterranean fruit fly (medfly) (Berg 1979; Hardy and Adachi 1956; Pruitt 1953). The following characteristics, in particular, distinguish larvae of the oriental fruit fly from the medfly (Heppner 1985): the anterior spiracles are aligned with a straighter distal margin than in the medfly, and the tubules (9–11) are noticeably bulbous; the cephalo-pharyngeal skeleton has a distinct sclerotized area between the post-hypostomial plates and the dorsal bridge; the caudal end has papillules I1-2 as distinct points, widely separated, on a raised margin, and D1-2 are less approximate; and the posterior spiracles are not as elongated (only about 3X width compared to 4–5X width in the medfly).
Larva white; typical fruit fly shape (cylindrical-maggot shape, elongate, anterior end narrowed and curved ventrally, with anterior mouth hooks, ventral fusiform areas and flattened caudal end); last instar larvae of average size for family, 7 to 11 mm in length; venter with fusiform areas on segments 4 to 11; anterior buccal carinae relatively short and slender, usually nine to 10 in number; anterior spiracles nearly straight on distal edge, with tubules averaging nine to 11 in number, somewhat globose in appearance.
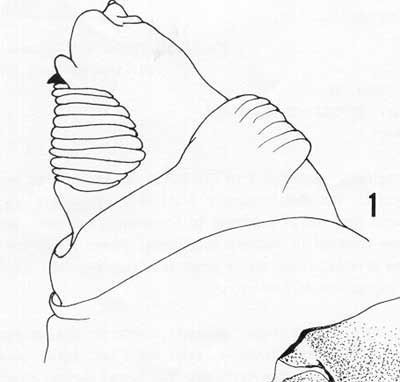
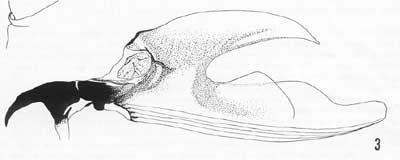
Cephalo-pharyngeal skeleton with large, convex, sharply pointed mouth hook each side, each hook about 2X hypostome length; hypostomium with prominent, semi-rounded subyhypostomium; post-hypostomial plates curved gradually to dorsal bridge, fused with sclerotized rays of central area of dorsal wing plate but with a semi-articulated area between; parastomium prominent; dorsal wing plate with posterior ray split; dorsal bridge anterior with a sclerotized point; pharyngeal plate about 25% longer than dorsal wing plate, with median area below dorsal bridge relatively unsclerotized, and a prominent hood.
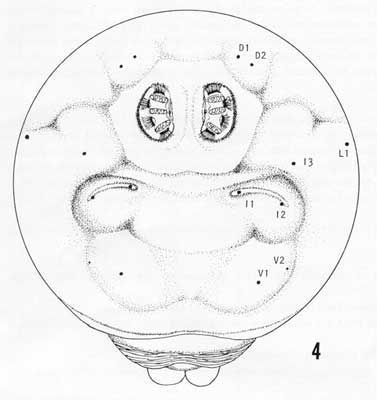
Caudal end with paired dorsal papillules (D1 and D2) diagonally dorsad to each spiracular plate; intermediate papillules (I1-2) as widely-separated pair on a large raised and curved elevation diagonally ventrad of each spiracular plate, with a remote I3 at about 45° from the I1-2 elevation; L1 on the median edge of the caudal end; a pair of ventral papillules (V1-2) approximately ventrad of I2 near the latero-ventral edge of the caudal end (V2 indistinct); posterior spiracles as three elongated (ca. 3X width) oval openings on each kidney-shaped spiracular plate, with dorsal and ventral spiracles angled to the caudal end center, and the median spiracle relatively straight; interspiracular processes (hairs) numerous, at four sites on each plate, latero-distal to spiracles, and the tips usually bifurcate; anal lobes entire and prominent.
The above descriptions were from larvae examined in verified samples from Hawaii (in immatures collection of the Florida State Collection of Arthropods).
Life History
Development from egg to adult under summer conditions requires about 16 days. The mature larva emerges from the fruit, drops to the ground, and forms a tan to dark brown puparium. Pupation occurs in the soil. About nine days are required for attainment of sexual maturity after the adult fly emerges. The developmental periods may be extended considerably by cool weather. Under optimum conditions, a female can lay more than 3,000 eggs during her lifetime, but under field conditions from 1,200 to 1,500 eggs per female is considered to be the usual production. Apparently, ripe fruit are preferred for oviposition, but immature ones may also be attacked.
Hosts and Damage
The oriental fruit fly has been recorded from 478 kinds of fruits and vegetables. However, avocado, mango, and papaya are the most commonly attacked.
In Hawaii, larvae were found in more than 125 kinds of hosts. Infestations of 50–80% have been recorded in pear, peach, apricot, fig, and other fruits in Western Pakistan. It is the principal pest of mangoes in the Philippines. It was a serious pest of citrus and other subtropical fruits in Japan, Okinawa, and the Japanese islands of Amami, Miyako, and Bonin before it was eradicated.
Damage, Quarantine, and Management
Injury to fruit, as with other members of this genus of fruit flies, occurs through oviposition punctures and subsequent larval development. It was introduced into the Hawaiian Islands about 1945, apparently by US military troops returning to the islands. Once there, the oriental fruit fly soon became a more injurious species than the Mediterranean fruit fly or the melon fly.

Credit: Scott Bauer, USDA
All Japanese territories were declared free of the oriental fruit fly in 1985, after an 18-year program of eradication combining insecticide-impregnated fiberblocks or cotton containing the powerful male attractant methyl-eugenol, and the sterile insect (sterile male) technique. Steiner traps baited with a lure and toxicant are also used to monitor the presence and control of the flies.

Credit: Okinawa Prefectural Fruit Fly Eradication Project Office

Credit: Okinawa Prefectural Fruit Fly Eradication Project Office
This pest has been intercepted on many occasions at ports of entry on the US mainland. The extensive damage caused by the oriental fruit fly in areas similar to Florida indicates that this species could rapidly become a very serious pest of citrus and other fruit and vegetables if it became established in Florida.
USDA-APHIS, in cooperation with threatened states, has established action plans that go into effect when fruit flies are trapped and reported (USDA 2008).
In Florida, the FDACS-Division of Plant Industry will cooperate with USDA-APHIS in regulating the actions of both commercial entities and homeowners.
Selected References
Armitage HM. 1951. Susceptibility of California fruits and vegetables to attack by the Oriental fruit fly (Dacus dorsalis). Bulletin of the Department of Agriculture California 40: 1–9.
Berg GH. 1979. Pictorial Key to Fruit Fly Larvae of the Family Tephritidae. San Salvador: Org. Internac. Reg. Sanidad. Agropec. 36 pp.
CABI. (December 2020). Bactrocera dorsalis. Distribution Maps of Plant Pests. https://www.cabidigitallibrary.org/doi/epdf/10.1079/DMPP/20203000830 (January 2023).
CDFA (California Department of Food and Agriculture). (August 2010). Oriental fruit fly. Pest Detection/Emergency Projects Branch. CDFA - Plant Health - Oriental Fruit Fly Project (ca.gov) (January 2023).
Drew RAI. 1989. The tropical fruit flies (Diptera: Tephritidae: Dacinae) of the Australasian and Oceanian region. Memoirs of the Queensland Museum 26. South Brisbane, Australia.
Drew RAI, Courtice AC, Teakle DS. 1983. Bacteria as a natural source of food for adult fruit flies (Diptera: Tephritidae). Oecologia (Berlin) 60: 279–284.
Ebeling W. 1959. Subtropical Fruit Pests. University of California, Division of Agricultural Science 436 pp.
FDACS. (August 2010). Exotic fruit flies found in Pinellas County. Florida Department of Agriculture and Consumer Services. (2 September 2010).
Foote RH, Blanc FL. 1963. The Fruit Flies or Tephritidae of California. Bulletin of the California Insect Survey 7: 1–117.
Fullaway DT. 1953. The Oriental fruit fly (Dacus dorsalis Hendel) in Hawaii. Proceedings of the Pacific Science Congress VII (1949), 4: 148–163.
Goergen G, Vayssières J-F, Gnanvossou D, Tindo M. 2011. Bactrocera invadens (Diptera: Tephritidae), a new invasive fruit fly pest for the Afrotropical Region: host plant range and distribution in West and Central Africa. Environmental Entomology. 40: 844–854.
Hardy DE. 1949. Studies in Hawaiian fruit flies (Diptera, Tephritidae). Proceedings of the Entomology Society of Washington 51: 181–205.
Hardy DE. 1969. Taxonomy and distribution of the oriental fruit fly and related species (Tephritidae, Diptera). Proceedings of the Hawaiian Entomological Society. 20: 395–428.
Hardy DE, Adachi M. 1956. Diptera: Tephritidae. Insects Micronesia (Honolulu) 14: 1–28.
Heppner JB. 1985. Larvae of fruit flies. II. Ceratitis capitata (Mediterranean fruit fly) (Diptera: Tephritidae). Florida Department of Agriculture and Consumer Services, Division of Plant Industry Entomology Circular 273.
Liquido NJ. 1991. Effect of ripeness and location of papaya fruits on the parasitization rates of Oriental fruit fly and melon fly (Diptera: Tephritidae) by braconid (Hymenoptera) parasitoids. Environmental Entomology 20: 1732–1736.
Mau MFA. (April 2007). Bactrocera dorsalis (Hendel). Crop Knowledge Master. http://www.extento.hawaii.edu/Kbase/Crop/Type/bactro_d.htm (5 August 2015).
Phillips VT. 1946. The biology and identification of trypetid larvae (Diptera: Trypetidae). Memoirs of the American Entomological Society 12: 1–161.
Pruitt JH. 1953. Identification of Fruit Fly Larvae Frequently Intercepted at Ports of Entry of the United States. University of Florida (Gainesville), MS thesis. 69 pp.
Schutze MK, Aketarawong N, Amornsak W, Armstrong KF, Augustinos AA, Barr N, Bo W, Bourtzis K, Boykin LM, Cáceres C, Cameron SL, Chapman TA, Chinvinijkul S, Chomic A, De Meyer M, Drosopoulou E, Englezou A, Ekesi S, Gariou-Papalexiou A, Geib SM, Hailstones D, Hasanuzzaman M, Haymer D, Hee AKW, Hendrichs J, Jessup A, Ji Q, Khamis FM, Krosch MN, Leblanc L, Mahmood K, Malacrida AR, Mavragani-Tsipidou P, Mwatawala M, Nishida R, Ono H, Reyes J, Rubinoff D, San Jose M, Shelly TE, Srikachar S, Tan KH, Thanaphum S, Haq I, Vijaysegaran S, Wee SL, Yesmin F, Zacharopoulou A, Clarke R. 2015. Synonymization of key pest species within the Bactrocera dorsalis species complex (Diptera: Tephritidae): taxonomic changes based on a review of 20 years of integrative morphological, molecular, cytogenetic, behavioural and chemoecological data. Systematic Entomology 40: 456–471.
Steck GJ. (July 2007). Oriental Fruit Fly Information. FDACS-DPI. (24 June 2008).
USDA. (2008). Fruit Fly Information, Quarantine Areas and Action Plans. Plant Health. http://www.aphis.usda.gov/plant_health/plant_pest_info/fruit_flies/index.shtml (5 August 2015).
USDA. 2016. A Review of Recorded Host Plants of Oriental Fruit Fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae). Version 2.1 A Product of the USDA Compendium of Fruit Fly Host Information (CoFFHI). A Farm Bill Project. July 22, 2016.
Vargas RI, Leblanc L, Putoa R, Eitam A. 2007. Impact of introduction of Bactrocera dorsalis (Diptera: Tephritidae) and classical biological control releases of Fopius arisanus (Hymenoptera: Braconidae) on economically important fruit flies in French Polynesia. Journal of Economic Entomology 100: 670–9.
Wong TTY, McInnis DO, Nishimoto JI. 1989. Relationship of sexual maturation rate to response of Oriental fruit fly strains (Diptera: Tephritidae) to methyl eugenol. Journal of Chemical Ecology 15: 1399–1405.