The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids, and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The polyphemus moth, Antheraea polyphemus (Cramer), is one of our largest and most beautiful silk moths. It is named after Polyphemus, the giant cyclops from Greek mythology who had a single large, round eye in the middle of his forehead (Himmelman 2002). The name is because of the large eyespots in the middle of the moth’s hind wings. The polyphemus moth also has been known by the genus name Telea, but it and the Old World species in the genus Antheraea are not considered to be sufficiently different to warrant different generic names. Because the name Antheraea has been used more often in the literature, Ferguson (1972) recommended using that name rather than Telea to avoid confusion. Both genus names were published in the same year. For a historical account of the polyphemus moth's taxonomy see Ferguson (1972) or Tuskes et al. (1996).
The family name Saturniidae is based on the eyespots of some members of the family that contain concentric rings reminiscent of the planet Saturn (Powell 2003). Because of their ease of rearing (Collins and Weast 1961, Villiard 1975) and large size, polyphemus moths are often reared by amateur enthusiasts and also have been used for numerous physiological studies—particularly for studies on molecular mechanisms of sex pheromone action.
Distribution
Polyphemus moths are our most widely distributed large silk moths. They are found from southern Canada down into Mexico and in all of the lower 48 states, except for Arizona and Nevada (Tuskes et al. 1996).
Description
Adults
The adult wingspan is 10 to 15 cm (approximately 4 to 6 inches) (Covell 2005). The upper surface of the wings is various shades of reddish brown, gray, light brown, or yellow-brown with transparent eyespots. There is considerable variation in color of the wings even in specimens from the same locality (Holland 1968). The large hind wing eyespots are ringed with prominent yellow, white (partial), and black rings. On the upper surface, there are pink-edged white ante-medial and post-medial lines on the forewing and a pinkish white-edged, black post-medial line on the hind wing. The undersides of the wings have areas with pinkish-white and others with various shades of brown.

Credit: Donald W. Hall, University of Florida

Credit: Donald W. Hall, University of Florida

Credit: Donald W. Hall, University of Florida

Credit: Donald W. Hall, University of Florida
Polyphemus antennae are quadripectinate (comb-like on four sides) with those of males being larger than those of females.

Credit: Donald W. Hall, University of Florida
Eggs
The slightly oval flattened eggs are white with two broad brown rings and are cemented to the substrate with a dark brown adhesive. Maximum reported dimensions in millimeters are 2.4 x 2 x 1.52 (length x width x height) (Peterson 1965).

Credit: Donald W. Hall, University of Florida
Larvae
Packard (1914) provided detailed descriptions of the eggs and each of the five larval instars and color drawings of instars 1 to 4. He gave the following lengths for the five instars: 1st instar: 5 to 6 mm (~1/4 in), 2nd instar: 14 to 15 mm (~2/3 in), 3rd instar: 20 to 25 mm (~1 in), 4th instar: 40 to 45 mm (~1 ½-1 ¾ in), 5th instar: 60 mm (2 1/3 in). First instar larvae are white with two black transverse bars on the tops and sides of each segment.

Credit: Donald W. Hall, University of Florida
Older instars are yellow-green, and the setae become relatively less prominent with each molt. The translucent yellowish-green to green, full-grown (fifth instar) caterpillars are 60 to 75 mm (2 1/3–3 in) in length (Godfrey et al. 1987). They have yellow mid-segmental lines that run from the sub-dorsal scoli (setae-bearing, wart-like bumps) touching the spiracles and to the lateral scoli on abdominal segments 2 to 7. The scoli are orange to red, and the lateral aspects of the dorsal and sub-dorsal scoli are silver. The dark area of the anal plate extends as a line part way across abdominal segment nine. Fifth instars have a yellow band that extends around the dorsal and lateral areas directly behind the head, yellow bands laterally at the bases of the thoracic legs, and a yellow mid-ventral line that extends the length of the abdomen. Fifth instar male larvae can be differentiated from female larvae by the presence of a black pit on the ventral aspect of the ninth abdominal segment of males that is lacking in females (Miller and Machotka 1980).

Credit: Donald W. Hall, University of Florida

Credit: Donald W. Hall, University of Florida
Cocoon and Pupa
The single-layered cocoon is thicker and more elliptical than the cocoon of the luna moth (Wagner 2005).

Credit: Donald W. Hall, University of Florida
The pupa is obtect (wings and appendages are appressed to the body). Female pupae may be distinguished from males by the presence of a longitudinal notch on the ventral surface of the fourth totally exposed abdominal segment. This notch is lacking in males. Also, the much larger developing antennae of male pupae touch each other at the mid-ventral line, while those of female pupae do not.

Credit: Donald W. Hall, University of Florida
The pupa is anchored to a loosely spun pad of silk at the rear of the cocoon by a cremaster (spines at the tip of the abdomen) that facilitates emergence of the adult from the pupal exoskeleton.

Credit: Donald W. Hall, University of Florida
Life Cycle
The polyphemus moth is univoltine (one brood per year) from Pennsylvania northward (Holland 1968) and bivoltine (two broods per year) from the Ohio Valley southward (Tuskes et al. 1996). However, due to staggered emergence, adults may be found during every month of the year in Florida (Heppner 2003).
The adult moth escapes the pupal case by splitting it at the anterior end and pushing the top up. Unlike the other large silk moths, the polyphemus and luna moths lack an escape "valve" in the cocoon to aid their emergence. Therefore, in order to escape their cocoons, they secrete a trypsin-like cocoonase enzyme to digest the sericin component of the silk at the anterior end of the cocoon to soften it (Kafatos and Williams 1964, Hruska et al. 1973). The cocoonase is produced and released from the highly modified maxillary galeae (the structures that form the tongue or proboscis of moths and butterflies that feed as adults). The moths then tear their way from the cocoon by the use of chitinous spurs on the thorax near the bases of the front wings while moving about the cocoon in a circular pattern (Hilton 1965). Actually, there are two spurs on each side—a large spur and a smaller spur immediately anterior to it (see photo).
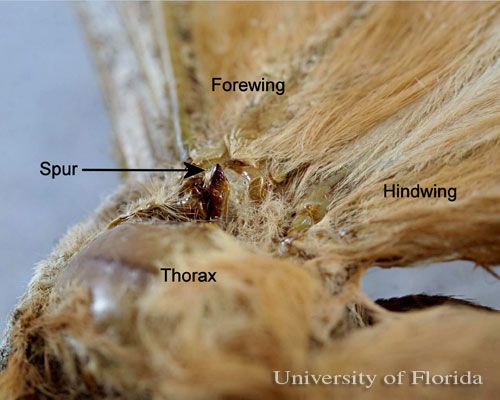
Credit: Donald W. Hall, University of Florida

Credit: Donald W. Hall, University of Florida
Adult eclosion (emergence from pupa) typically occurs in the afternoon with males usually beginning emergence several days before females from the same brood. Afternoon emergence allows time for expansion and drying of the wings prior to the evening flight period. Also, during the first day after emergence, the moth voids the liquid meconium, which is composed of the breakdown waste products of the old larval tissues.
The adults are strongly attracted to light—particularly UV wavelengths. There has been some concern that light pollution from man-made sources (particularly mercury vapor street lights) may deter silk moths from mating and have a negative impact on their populations in urban areas (Worth and Muller 1979). Fortunately, mercury vapor lamps are not used as commonly as they once were for street lights.
Females release a sex-attractant pheromone and may attract males from a distance beginning late evening of the day of emergence. The sex pheromone has been characterized as a 90:10 mixture, respectively, of trans-6, cis-11-hexadecadienyl acetate and trans-6, cis-11-hexadecadienal (Kochansky et al. 1975). While males are attracted all night long, maximal attractiveness is during the last two hours before sunrise (Kochansky et al. 1977). Mating pairs remain coupled throughout the day and separate at dusk. Either caged or tethered females can be used to attract wild males. Worth (1980) has designed a tether for this purpose that prevents the tethered moth from escaping or twisting the tethering line around its body.
Females begin laying eggs the evening after mating and continue for several nights. Eggs are laid singly or in groups of two or three on leaves of the host plant (Opler et al. 2012). In captivity, they will lay their eggs on any substrate. Adults have vestigial mouthparts and do not feed. Therefore, they are short-lived.
Caterpillars are solitary and develop through five instars. Immediately after hatching, caterpillars eat their egg shells (Opler et al. 2012). Older instars eat whole leaves and then sever the petioles to drop them to the ground (Tuskes et al. 1996)—possibly to obscure their presence from predators. The presence of large larvae on branches overhead may often be detected by the presence of frass (caterpillar droppings) on pavement of driveways or parking lots.

Credit: Donald W. Hall, University of Florida
Caterpillars exposed to short photoperiods (8-12 hours) produce diapausing (overwintering) pupae while those exposed to long photoperiods (>17 hours) produce non-diapausing pupae (Mansingh and Smallman 1967). Both induction and termination of diapause also may be influenced by temperature (Mansingh and Smallman 1971). Polyphemus caterpillars are polyphagous and have been reported in nature from over 50 species of broad-leaved plants (Ferguson 1972, Heppner 2003, Tietz 1972, Tuskes et al. 1996) and will feed on a wide variety of species in captivity. Polyphemus caterpillars use the full range of cocoon placement behaviors found in the subfamily Saturniinae as described by Wagner and Mayfield (1980):
- Attached longitudinally and permanently to a twig so that the cocoon does not fall to the ground during the winter
- Attached apically by a silk peduncle (occasionally by a double peduncle) to a twig. Peduncle may be weak allowing cocoon to fall to ground during winter or strong so that cocoon remains on tree all winter. Attachment by a peduncle may be more common in the South (Wagner 2005)
- Attached among leaves on trees but with no peduncle so that most fall to ground during winter
- Spun on ground among fallen leaves, grasses or herbs
In the West, Native Americans sometimes fill the cocoons of polyphemus and other silk moths with sand or pebbles to make rattles by tying them to sticks or to their ankles for use as musical instruments (Himmelman 2002).
Defenses and Natural Enemies
Polyphemus caterpillars are never sufficiently common to cause significant damage to their host trees except occasionally in California where they may be pests of commercial plums (Tuskes et al. 1996). Their populations are regulated by natural enemies—including insect parasitoids and generalist predators.
Polyphemus caterpillars gain protection from predators by their cryptic green coloration. When threatened they often rear the front part of the body in a "Sphinx" pose—possibly to make them less caterpillar-like to a predator. If attacked, polyphemus caterpillars, as well as those of many other bombycoid moths, make a clicking noise with the mandibles—sometimes as a prelude to or accompanied by defensive regurgitation of distasteful fluids. Brown et al. (2007) found that ants and mice were deterred by the regurgitant of the polyphemus caterpillars and suggested that the clicking is a warning of the impending regurgitation.

Credit: Donald W. Hall, University of Florida
Cocoons hanging in trees are susceptible to attack by squirrels (Young 1982) and woodpeckers (Waldbauer 1970). Cocoons falling with the leaves in the fall or being spun among leaves or grasses on the ground may be more susceptible to attack by mice in the leaf litter, but Waldbauer (1996) believes that these cocoons gain protection from their random scattering in the litter and that the majority of cocoons that remain suspended in trees are attacked by woodpeckers.

Credit: Donald W. Hall, University of Florida
When threatened, adult polyphemus flip the front wings forward and also often flap the wings exposing the large hind wing eyespots—possibly to startle potential predators. Cryan (1973) reported an incident in which display of the eyespots appeared to protect a polyphemus in flight from blue jays.
Tuskes et al. (1996) listed the following insect parasitoids (with references) of polyphemus: ten species of tachinid flies (Arnaud 1978, O'Hara and Wood 2004), four species of chalcidoid wasps (Krombein et al. 1979, Peck 1963), one species of braconid wasp (Krombein et al. 1979), five species of ichneumonid wasps (Krombein et al. 1979, Townes 1944), and one species of proctotrupoid wasp (Collins and Weast 1961).
Old cocoons made by caterpillars that were parasitized by ichneumon wasps have an exit hole made by the wasp that is similar to that made by woodpeckers. However, they can be differentiated from cocoons attacked by woodpeckers by the presence inside of the cocoon of the ichneumon and the cadaver of the polyphemus caterpillar.

Credit: Donald W. Hall, University of Florida
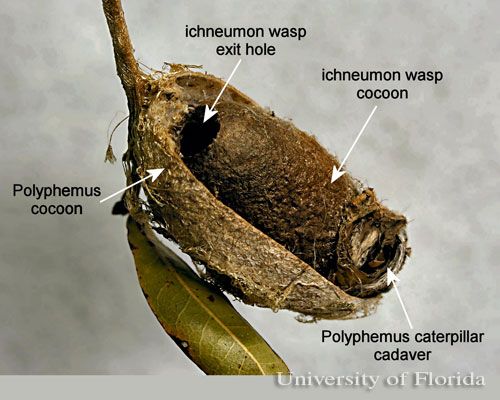
Credit: Donald W. Hall, University of Florida
Selected References
Arnaud PH. 1978. A Host-Parasite Catalog of North American Tachinidae (Diptera). United States Department of Agriculture Miscellaneous Publication 1319. Washington, D.C.
Brown SG, Boettner GH, Yack JE. 2007. Clicking caterpillars: acoustic aposematism in and other Bombycoidea. The Journal of Experimental Biology 210: 993-1005.
Collins MM, Weast RD. 1961.Wild Silk Moths of the United States: Saturniinae. Collins Radio Company. Cedar Rapids, Iowa. 138 pp.
Covell CV. 2005. A Field Guide to Moths of Eastern North America. Special Publication Number 12. Virginia Museum of Natural History. Martinsville, Virginia. 496 pp.
Cryan JP. 1973. Protective coloration used for defense by a polyphemus moth, Cramer, in flight. Engelhardtia 6(1): 14.
Ferguson DC. 1972. In Dominick RB, Edwards CR, Ferguson DC, Franclemont JG, Hodges RW, Munroe EG. The Moths of America North of Mexico, fasc. 20.2B, Bombycoidea (in part). EW Classey, LTD. Middlesex, England.
Godfrey GL, Jeffords M, Appleby JE. 1987. Saturniidae (Bombycoidea). In Stehr FW. Immature Insects. Kendall/Hunt Publishing Company. Dubuque, Iowa. pp. 513-521.
Heppner JB. 2003. Lepidoptera of Florida. Part 1. Introduction and Catalog. Volume 17 of Arthropods of Florida and Neighboring Land Areas. Division of Plant Industry. Florida Department of Agriculture and Consumer Services. Gainesville, Florida. 670 pp.
Hilton HO. 1965. Pupal escape mechanism of certain saturniid moths. Florida Entomologist 48: 239-24.
Himmelman J. 2002. Discovering Moths: Nighttime Jewels in Your Own Backyard. Down East Books. Camden, Maine. 232 pp.
Holland WJ. 1968.The Moth Book: A Guide to the Moths of North America. Dover Publications, Inc. New York. (First published in 1903 by Doubleday, Page and Company. New York) 479 pp.
Hruska JF, Felsted RL, Law JH. 1973. Cocoonases of silkworm moths: catalytic properties and biological function. Insect Biochemistry 3: 31-43.
Hruska JF, Felsted RL, Law JH, Berger E, Kafatos FC. 1973. Cocoonase. IV. Mechanism of activation of prococoonase from words Antheraea polyphemus. The Journal of Biological Chemistry 248(9): 3012-3020.
Kafatos FC, Williams CM. 1964. Enzymatic mechanism for the escape of certain moths from their cocoons. Science 146: 538-540.
Kochansky J, Tette J, Taschenberg EF, Carde RT, Kaissling K-E, Roelofs WL. 1975. Sex pheromone of the moth, Antheraea polyphemus. Journal of Insect Physiology 21: 1977-1983.
Kochansky J, Carde RT, Taschenberg EF, Roelofs WL. 1977. Rhythms of male attraction and female attractiveness, and an improved pheromone synthesis. Journal of Chemical Ecology 3(4): 419-427.
Krombein KV, Hurd PD, Jr, Smith DR, Burks BD. 1979.Catalog of Hymenoptera in America North of Mexico. Vol. 1, Symphyta and Apocrita (Parasitica). Smithsonian Institution Press, Washington, D.C. 2735 pp.
Mansingh A, Smallman BN. 1967. Effect of photoperiod on the incidence and physiology of diapause in two saturniids. Journal of Insect Physiology 13: 1147-1162.
Mansingh A, Smallman BN. 1971. The influence of temperature on the photoperiodic regulation of diapause in saturniids. Journal of Insect Physiology 17: 1735-1739.
Miller TA, Machotka SV. 1980. Sex-related morphological characters in larvae of Hyalophora gloveri gloveri and Antheraea polyphemus (Saturniidae). Journal of the Lepidopterists' Society 34(1): 69-73.
O'Hara JE, Wood DM. 2004. Catalogue of the Tachinidae (Diptera) of North America north of Mexico. Associated Publishers. Gainesville, Florida 410 pp.
Opler, PA, Lotts K, Naberhaus T, coordinators. 2012. Butterflies and Moths of North America.
Packard AS. 1914. Monograph of the Bombycine Moths of North America. Part 3. Memoirs of the National Academy of Science 12: 1-516.
Peck O. 1963. A catalogue of the Nearctic Chalcidoidea (Insecta: Hymenoptera). Canadian Entomologist. Supplement 30. Canada Department of Agriculture. Ottawa, Canada. 1092 pp.
Peterson A. 1965. Some eggs of moths among the Sphingidae, Saturniidae, and Citheroniidae (Lepidoptera). Florida Entomologist 48: 213-219.
Powell JA. 2003. Lepidoptera (Moths, Butterflies). Resh VH, Carde RT, editors. Encyclopedia of Insects. Elsevier Science (USA) San Diego, California. pp. 631-663.
Tietz HM. 1972. An Index to the Described Life Histories, Early Stages and Hosts of the Macrolepidoptera of the Continental United States and Canada. Part 1. The Allyn Museum of Entomology. Sarasota, Florida. (Distributed by Entomological Reprint Specialists. Los Angeles, California). 536 pp.
Townes HK. 1944. A Catalog and Reclassification of the Nearctic Ichneumonidae (Hymenoptera). Part 1. Memoirs of the American Entomological Society. American Entomological Society. Philadelphia, Pennsylvania. Volume 11: 1-477.
Tuskes PM, Tuttle JP, Collins MM. 1996. The Wild Silk Moths of North America: The Natural History of the Saturniidae of the United States and Canada. Cornell University Press. Ithaca, New York. 250 pp.
Villiard P. 1975. Moths and How to Rear Them. Dover Publications, Inc. New York, New York. 242 pp.
Wagner DL. 2005. Caterpillars of Eastern North America. Princeton University Press. Princeton, New Jersey. 512 pp.
Wagner WH, Mayfield 1980. Foodplants and cocoon construction in Antheraea polyphemus (Lepidoptera: Saturniidae) in southern Michigan. The Great Lakes Entomologist 13(3): 131-138.
Waldbauer GP. 1996. Insects Through the Seasons. Harvard University Press. Cambridge, Massachusetts. 289 pp.
Waldbauer GP, Sternburg JG, George WG, Scarbrough AG. 1970. Hairy and downy woodpecker attacks on cocoons of urban Hyalophora cecropia and other saturniids (Lepidoptera). Annals of the Entomological Society of America 63(5): 1366-1369.
Worth CB. 1980. An elegant harness for tethering large moths. Journal of the Lepidopterists' Society 34(1): 61-63.
Worth CB, Muller J. 1979. Captures of large moths by an UV light trap. Journal of the Lepidopterists' Society 33: 261-264.
Young AM. 1982. Predation on the pupae of Saturniidae (Lepidoptera) by gray squirrels in Wisconsin. The Great Lakes Entomologist 15(2): 145.