Besnoitiosis is a skin condition affecting donkeys in Florida and throughout the United States. Previously thought to be extremely rare in the US, besnoitiosis was confirmed on multiple donkey farms between 2010 and 2012. It is currently considered an emerging infectious disease of American donkeys. Besnoitiosis is caused by infection with the protozoan parasite Besnoitia spp., which are cyst-forming coccidia that affect multiple host species worldwide (Olias, Schade, and Mehlhorn 2011). Besnoitia bennetti (B. bennetti) is the species known to infect equids and has been reported in donkeys in Africa, Asia, and more recently, the United States and Europe (Lienard et al. 2018; Dubey et al. 2005; Ness et al. 2012; Davis, Peters, and Dunstan 1997; Elsheikha et al. 2005; Terrell and Stookey 1973). Currently, the only reported cases of equine besnoitiosis in North America have been in donkeys.
Life Cycle
The Besnoitia life cycle involves both a definitive (predator) and an intermediate (prey) host. Infective Besnoitia oocysts are ingested by the intermediate host and develop into thick-walled connective tissue cysts containing millions of bradyzoite-stage parasites (Dubey et al. 2005; Dubey and Yabsley 2010). The cat has been identified as a definitive host for other species of Besnoitia. These include B. oryctofelisi, B. darlingi, and B. neotomofelis, which infect the rabbit, opossum, and Southern Plains woodrat, respectively (Dubey and Yabsley 2010; Dubey et al. 2004; Dubey and Lindsay 2003). Attempts to demonstrate the cat, or any other animal, as the definitive host in equine besnoitiosis have been unsuccessful, thus precluding researchers from elucidating the parasite’s life cycle and mode of disease transmission in equine infection.
Clinical Findings
Besnoitiosis primarily affects the skin, causing a miliary dermatitis that appears as tiny white bumps in the skin, mucous membranes, and conjunctiva (Figure 1). The skin over the muzzle, nostrils, ears, genitalia, and inner thigh appears to be preferentially affected. One of the most unique features of besnoitiosis is the development of “scleral pearls,” which are cysts along the border of the cornea and sclera of the eye (Figure 2). Cysts have also been identified in the testicles, nasal and oral cavities, trachea (airway), and esophagus of infected donkeys (Figure 3). Affected donkeys often have a history of chronic dermatitis (skin disease) that does not respond to treatment with antibiotics, ointments, medicated baths, and other typical skin remedies.
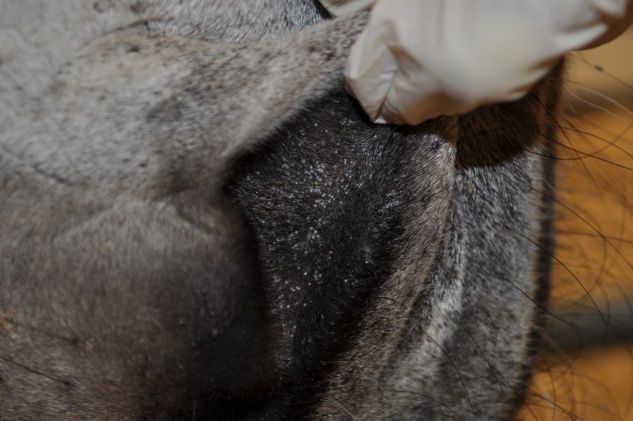
Credit: Sally DeNotta, UF
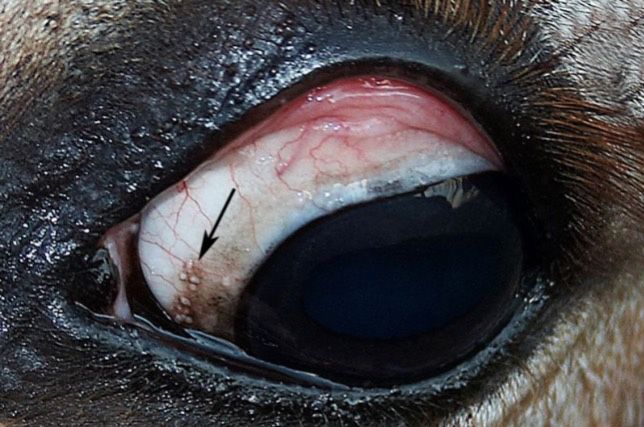
Credit: Sally DeNotta, UF
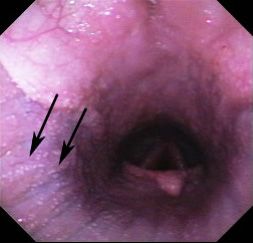
Credit: Sally DeNotta, UF
In an epidemiologic investigation of 416 donkeys from herds across the United States, young animals (median age 24 months) were at increased risk of developing besnoitiosis compared to older individuals. The most common lesions in infected donkeys were cysts in the nares (94%) and scleral pearls (81%) (Ness et al. 2014). Some infected animals remain otherwise healthy, while others appear to suffer weight loss and fail to thrive due to the disease. There are reports of Besnoitia infection in European cattle leading to severe illness and death, but this severe form of besnoitiosis has not been observed in donkeys (Olias, Schade, and Mehlhorn 2011; Jacquiet, Liénard, and Franc 2010; Schares et al. 2010).
Diagnosis
The current method of diagnosis for besnoitiosis in donkeys is direct identification of Besnoitia cysts within the skin by microscopic examination of a skin biopsy (Figure 4). Antibody tests used for cattle in Europe have been evaluated and found to be effective for identifying infected donkeys, but these assays are not currently available for use in the United States (Ness et al. 2014).
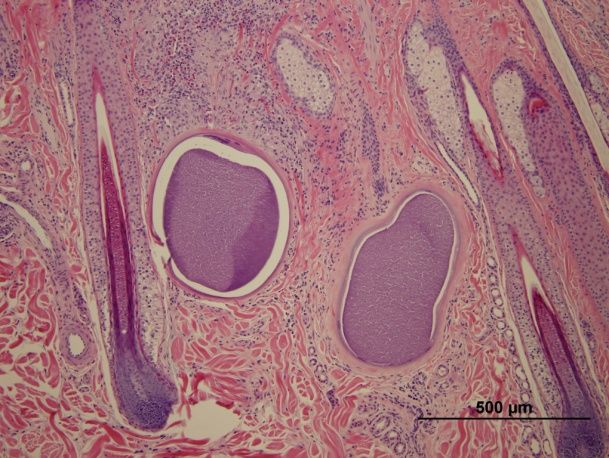
Credit: Sally DeNotta, UF
Prevention
Effective prevention of besnoitiosis is hindered by lack of simple methods for screening donkeys for infection. All donkeys should be examined for visible lesions. Animals with suspected lesions should be tested via skin biopsy. Avoiding purchasing animals from herds with known infections is also advisable, although there are currently no data to support this recommendation in donkeys, specifically.
Therapy
There are currently no known effective treatments for equine besnoitiosis. Treatment with high doses of antiprotozoal ponazuril for 45 days was not effective in two naturally infected donkeys (Ness et al. 2012). Reported attempts to treat infected animals with the oral antiprotozoal medications trimethoprim-sulfamethozazole or nitazoxanide have had variable success, but limited case numbers and lack of knowledge regarding clinical disease make drawing useful conclusions from the existing literature difficult (Dubey et al. 2005; Elsheikha et al. 2005). The potential for natural recovery from besnoitiosis and the long-term prognosis for infected animals remain unknown. The author has followed several infected donkeys for five years or longer. These cases have not spontaneously resolved, nor have they responded to the aforementioned therapies.
Summary
Besnoitiosis is an infectious skin condition affecting donkeys in Florida. Donkey owners should monitor their herds for clinical signs of besnoitiosis and alert their veterinarians to any suspected infections.
References
Bennett, S. C. 1927. “A Peculiar Equine Sarcosporidium in the Anglo-Egyptian Sudan.” Vet. J. 83:297–304.
Bennett, S. C. 1933. “Globidium Infections in the Sudan.” J. Comp. Path. Thera. 46:1–15.
Davis, W. P., D. F. Peters, and R. W. Dunstan. 1997. “Case Report: Besnoitiosis in a Miniature Donkey.” Vet. Dermatol. 8:139–143.
Dubey, J. P., and D. S. Lindsay. 2003. “Development and Ultrastructure of Besnoitia oryctofelisi Tachyzoites, Tissue Cysts, Bradyzoites, Schizonts and Merozoites.” Int. J. Parasitol. 33:807–819.
Dubey, J. P., C. Sreekumar, T. Donovan, et al. 2005. “Redescription of Besnoitia bennetti (Protozoa: Apicomplexa) from the Donkey (Equus asinus).” Int. J. Parasitol. 35:659–672.
Dubey, J. P., C. Sreekumar, B. M. Rosenthal, et al. 2004. “Redescription of Besnoitia tarandi (Protozoa: Apicomplexa) from the Reindeer (Rangifer tarandus).” Int. J. Parasitol. 34:1273–1287.
Dubey, J. P., and M. J. Yabsley. 2010. “Besnoitia neotomofelis n. sp. (Protozoa: Apicomplexa) from the Southern Plains Woodrat (Neotoma micropus).” Parasitology 137:1731–1747.
Elsheikha, H. M., C. D. Mackenzie, B. M. Rosenthal, et al. 2005. “An Outbreak of Besnoitiosis in Miniature Donkeys.” J. Parasitol. 4:877–881.
Jacquiet, P., E. Liénard, and M. Franc. 2010. “Bovine Besnoitiosis: Epidemiological and Clinical Aspects.” Vet. Parasitol. 174:30–36.
Lienard, E., A. Nabuco, S. Vandenabeele, et al. 2018. “First Evidence of Besnoitia bennetti Infection (Protozoa: Apicomplexa) in Donkeys (Equus asinus) in Belgium.” Parasit. Vectors 11(1): 427.
Ness, S. L., J. Peters-Kennedy, G. Schares, et al. 2012. “Investigation of an Outbreak of Besnoitiosis in Donkeys in the Northeastern United States.” J. Am. Vet. Med. Assoc. 240(11): 1329–1337.
Ness, S. L., G. Schares, J. Peters-Kennedy, et al. 2014. “Serological Diagnosis of Besnoitia bennetti Infection in Donkeys (Equus asinus).” J. Vet. Diagn. Invest. 26(6): 778–782.
Olias, P., B. Schade, and H. Mehlhorn. 2011. “Molecular Pathology, Taxonomy and Epidemiology of Besnoitia Species (Protozoa: Sarcocystidae).” Infection, Genetics and Evolution 11:1464–1576.
Schares, G., W. Basso, and M. Majzoub, et al. 2010. “Comparative Evaluation of Immunofluorescent Antibody and New Immunoblot Tests for the Specific Detection of Antibodies against Besnoitia besnoiti Tachyzoites and Bradyzoites in Bovine Sera.” Vet. Parasitol. 171:32–40.
Terrell, T. G., and J. L. Stookey. 1973. “Besnoitia bennetti in two Mexican Burros.” Vet. Path. 10:177–184.