The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The Gulf Coast tick (Figure 1), Amblyomma maculatum, was first described by Koch in 1844. Gulf Coast ticks are found in grass prairies and coastal uplands throughout much of the western hemisphere. The ticks are ectoparasites that feed on a variety of birds and mammals and will readily bite humans. Gulf Coast ticks are of increasing concern because of their ability to transmit several pathogens of veterinary and medical importance.

Credit: Jeffrey C. Hertz, edited by Jane Medley
Synonymy
Amblyomma maculatum Koch, 1844
= Amblyomma rubripes Koch, 1844 (synonym)
= Amblyomma ovatum Koch, 1844 (synonym)
= Amblyomma complanatum Berlese, 1888 (synonym)
= Amblyomma immaculatum Spicer & Dove, 1938 (misapplied name)
= Amblyomma (Anastosiella) maculatum Santos Dias, 1993 (synonym)
From the Catalogue of Life: 2010 Annual Checklist (ITIS 2014) and reviewed by Teel et al. (2010)
Distribution
Gulf Coast ticks have been reported throughout the Americas and on some of the islands in the Caribbean basin (Teel et al. 2010). The common name of this tick was given because of the prevalence of the tick along the Gulf Coast of the United States (Hooker et al. 1912) (Figure 2). Historically, Gulf Coast ticks were found only in the southeastern states but current collection records reveal a larger distribution. Incidental records have been collected as far north as Maine and Iowa to the east and west, respectively. It is thought that these records have not resulted in established populations. However, introductions of Gulf Coast tick-infested cattle from the southeast into Kansas and Oklahoma in the 1950s created additional populations throughout more than half of Oklahoma and a quarter of Kansas (Teel et al. 2010). New populations have been reported in Delaware and Maryland that appear to be established because of the number of adult specimens recovered pre- and post-winter (Florin et al. 2014). In Florida, Gulf Coast ticks are found throughout the state, but appear to have higher densities in the southern region (Allan et al. 2001).
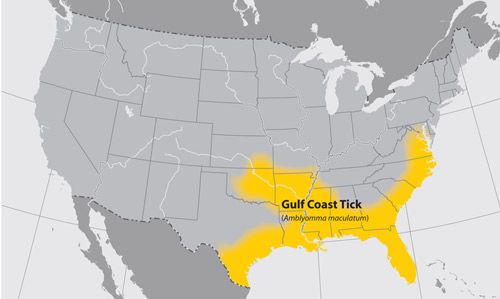
Credit: Centers for Disease Control and Prevention
Life Cycle and Description
The Gulf Coast tick life cycle consists of eggs and three active life stages (Figure 3): larvae, nymphs, and adults. The immature life stages resemble the adults; however, they are smaller, with larvae possessing six legs instead of the eight legs found on nymphs and adults. Characteristics of each life stage are detailed below (Figures 4–8).

Credit: Jeffrey C. Hertz
Gulf Coast ticks are considered three-host ticks because each active life stage feeds on a different host. In general, Gulf Coast tick larvae and nymphs feed on small animals, such as birds, rodents and rabbits, while adults feed on larger animals including dogs, coyotes, skunks, panthers, and bears (Teel et al. 2010). Gulf Coast ticks preferentially infest the ears of host animals and feed by inserting their barbed mouthparts, called a hypostome (Fig. 10), into the host's skin (Hooker et al. 1912). Feeding and mating occur in succession on the host. After eight days of blood feeding on a host and completion of spermatogenesis, male Gulf Coast ticks release a pheromone which alerts female ticks, also located on the host, that they are ready to mate (Rechav et al. 2000). Thereafter, female Gulf Coast ticks aggregate near the male and attach to the host to blood feed. Males move to the females and complete the mating process. After the blood meal and mating have been completed, the females drop off the host and find suitable microhabitats to lay their egg clutch, which typically takes up to 26 days (Drummond and Whetstone 1970); males remain on the host and mate with additional females.
Following an incubation period of up to three weeks in optimum temperatures (27°C (80.6°F) at 70%–98% humidity), larvae hatch and aggregate on the lower surface of vegetation before ascending the foliage in search of their first host through a behavior called questing. Gulf Coast ticks are xerophilic, which means that they love drier environments, and can quest for an extended period of time in peak temperature environments to find suitable hosts (Yoder et al. 2008). Once on the host, immature Gulf Coast ticks remain attached until fully engorged, which can take up to 10 days (Teel et al. 2010). Replete ticks drop from their host and stay hidden in the environment as they undergo a molt to the next active life stage.
Seasonal peaks in Gulf Coast tick populations vary based on their geographic location (Teel et al. 2010). Coastal populations found in Texas are active from May through March, whereas inland populations in Oklahoma and Kansas are active from February through October (Teel et al. 2010). In Texas, adult activity peaks in August, followed by larvae in December and nymphs in January. In Oklahoma and Kansas, adult activity peaks in April, followed by larvae in June and nymphs in July. Gulf Coast ticks in Mississippi have been collected from March through November, with adults peaking during late July to early August (Goddard and Paddock 2005). Seasonal activity of Gulf Coast ticks in northern Florida occurs from February to September (Cilek and Olson 2000).
Eggs
Eggs (Figure 4) are smooth brown, shiny ellipsoids with an average size of 0.520 by 0.397 mm (.020 by .016 in) (l × w) (Hooker et al. 1912). Engorged females convert 52-69% of their body weight to producing eggs, resulting in approximately 10,000 eggs laid per clutch (Drummond and Whetstone 1970). The most eggs recorded from one engorged female Gulf Coast tick was 18,497 (Bishopp and Hixson 1936).

Credit: Lyle Buss
Larvae
Larvae (Figure 5) are minute, approximately 0.5 mm (0.02 in) in size, possess six legs, and appear pink, dull white, or gray-colored when engorged (Hooker et al. 1912). One often encounters thousands of larvae, often referred to as "seed ticks", due to a recent hatch of an egg clutch. Larvae are estimated to live up to six months depending on environmental conditions (Teel et al. 2010). As larvae age, they alter their behavior from host-seeking to water conservation (Fleetwood and Teel 1983). Newly hatched larvae are generally active in the mid- to late afternoon, when the saturation deficit is at its highest, whereas older larvae are active in the morning and evening hours (Fleetwood and Teel 1983).

Credit: Jeffrey C. Hertz
Nymphs
Nymphs (Figure 6) have eight legs and are approximately 1.33 by 0.75 mm (~1/16 by 1/32 in) (l × w), unengorged, and can appear three times larger when engorged (Hooker et al. 1912). The cuticle is shiny and can range in color from dark bluish gray to dull white (Hooker et al. 1912). Nymphs are estimated to live up to seven months depending on environmental conditions (Teel et al. 2010).

Credit: Jeffrey C. Hertz
Adults
Adults are approximately 6 mm (~1/4 in) in length, have eight legs, and exhibit sexual dimorphism, meaning they are recognizably different in appearance from one another (Hooker et al. 1912). Unengorged females have a reduced dark brown scutum, a plate on their back, with silvery white posterior ornamentation that includes one median and two lateral interrupted stripes. Whereas unengorged males' scutum covers the entire top of the tick, is dark brown with interconnected ornamentation of silvery white lines. Unfed adults have been shown to live as long as 16 months, and generally live twice as long as nymphs, and up to four times as long as larvae under optimal laboratory conditions (Teel et al. 2010). Engorged ticks look strikingly different than unengorged ticks (Figure 7).

Credit: Lyle Buss
In Florida, the ticks most likely confused with the male Gulf Coast tick are male lone star ticks and male American dog ticks (Figure 8). Female Gulf Coast ticks may be confused with female American dog ticks (Figure 9). The silvery ornamentation on male lone star ticks is restricted to six disassociated, symmetrical markings located on the lateral edges of the scutum, whereas Gulf Coast tick patterning is interconnected and covers most of the tick's dorsum. American dog ticks can be differentiated from Gulf Coast ticks by comparing the mouthparts. American dog tick mouthparts are short and pyramid-shaped in relation to the long, rectangular mouthparts of the Gulf Coast tick.

Credit: Gulf Coast tick, Jeffrey C. Hertz; lone star tick, Chris Holderman; and American dog tick, Lyle Buss

Credit: Gulf Coast tick, Jeffrey C. Hertz; and American dog tick, Lyle Buss
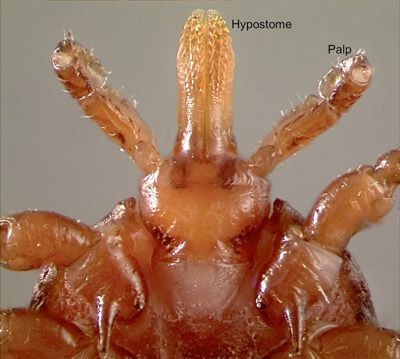
Credit: Jeffrey C. Hertz
Hosts
Gulf Coast ticks have been collected from 71 species of birds and mammals in the United States (Teel et al. 2010). Immature life stages are reported to feed on the head and neck region of their hosts, while adults prefer to concentrate on the external regions of the animal's ears (Hooker et al. 1912; Bishopp and Trembley 1945; Teel et al. 2010). Larval and nymphal stages typically parasitize small animals, but may prefer birds like the bobwhite quail, lark buntings, and meadowlark as primary hosts (Hixson 1940; Bishopp and Trembley 1945; Robbins et al. 2010). Adult Gulf Coast ticks are more likely to feed on large, wild and domestic mammals including white-tailed deer, cattle, horses, swine, and companion animals (Teel et al. 2010). Only the coyote (Canis latrans Say) and domestic sheep (Ovis aries L.) have had all active life stages of the Gulf Coast tick collected from them (Teel et al. 2010).
Medical and Veterinary Importance
The historical importance of Gulf Coast ticks has centered on the impact due to livestock infestations. Gulf Coast tick bites can cause severe inflammation, abscesses, and predispose livestock hosts to the primary screwworm, Cochliomyia hominivorax Coquerel, where this pest occurs and can cause other tissue damage (Hooker et al. 1912; Bishopp and Trembley 1945; Teel et al. 2010). Additionally, severe infestations on livestock have been shown to cause lethargy, debilitation, and altered body composition (Teel et al. 2010). Livestock infested with Gulf Coast ticks were often recognized by drooping "gotched" ears (Edwards 2011), which reduced market value at sale (Hooker et al. 1912). Today, the importance of the Gulf Coast tick is more profound because of the recognition of the impacts of pathogenic organisms transmitted by this tick that result in serious medical conditions and diseases affecting humans and their domesticated and companion animals.
Rickettsiosis
Rickettsia rickettsii, the causative agent of Rocky Mountain spotted fever, was the only known tick-borne spotted fever to occur in the United States. This changed in 2002 when a rickettsial species, identified 60 years earlier, was implicated as the causative agent of rickettsiosis in a middle-aged man from Virginia (Paddock et al. 2004). This newly recognized pathogen, Rickettsia parkeri, is primarily transmitted by Gulf Coast ticks and is now recognized as a human pathogen of increasing public health concern in the southeastern United States (Walker et al. 2008; Cragun et al. 2010; Parola et al. 2013). Gulf Coast tick Rickettsia parkeri infection rates in some areas have been recorded as high as 55% (Nadolny et al. 2014).
Canine Hepatozoonosis
Gulf Coast ticks are recognized as the principal vector of Hepatozoon americanum, the causative agent of American canine hepatozoonosis in the southern United States (Ewing and Panciera 2003). Interestingly, the protozoan pathogen is not transmitted through the bite of an infected tick; rather, transmission occurs when dogs consume infected ticks while grooming. In wild canines, transmission often occurs through predation of infected prey (Baneth 2011).
Leptospirosis
Laboratory experiments have shown Gulf Coast ticks are capable of transmitting the causative agent of leptospirosis in livestock, Leptospira pomona (Burgdorfer 1957). Burgdorfer used capillary tubes filled with concentrated infected blood to artificially feed ticks. The ticks were then allowed to feed on naïve guinea pigs approximately two weeks later and some of the guinea pigs became ill with infection caused by the spirochete pathogen. However, the role of Gulf Coast ticks in natural transmission of Leptospira in the United States is unclear.
Heartwater
In the laboratory, Gulf Coast ticks have been shown to be competent vectors of Ehrlichia (formerly Cowdria) ruminantium, the Heartwater pathogen (Mahan et al. 2000). Heartwater is a fatal disease of wild and domestic ruminants (i.e. cattle, deer, sheep) and is transmitted by ticks in the genus Amblyomma (Mahan et al. 2000). Heartwater occurs in sub-Saharan Africa and on some islands in the Caribbean (Deem 1998) but has not been detected in the United States. However, two of the principal vectors, Amblyomma variegatum (Fabricius) and Amblyomma hebraeum Koch, have been reported on imported wildlife in Florida and could introduce Ehrlichia ruminantium to naïve host species (Wilson and Bram 1998; Burridge et al. 2000). If introduced, Ehrlichia ruminantium potentially could be maintained in animal disease cycles by the Gulf Coast tick.
Tick Paralysis
Tick paralysis is a potentially fatal medical condition caused by tick toxins released during feeding that affect the host's motor neurons, leading to paralysis and respiratory failure (Espinoza-Gomez et al. 2011). Once the tick is removed or voluntarily detaches, full recovery is usually observed within 24 hours (Edlow 2010). Gulf Coast ticks have been responsible for paralysis in dogs (Gothe et al. 1979; Sonenshine 1993) and humans (Paffenbarger 1951; Espinoza-Gomez et al. 2011), although these incidents are uncommon.
Management
Understanding the Gulf Coast tick's biology, seasonality, and habitat distribution are paramount to a successful management program. Integrated tick management involves personal protection, acaricide application, habitat modification, and host animal exclusion. An excellent resource for southern tick management programs, including the identification and biology of other ticks, is the TickApp maintained by Texas A&M University (Teel et al. 2011) and the TickEncounter Resource Center maintained by the University of Rhode Island.
The most important way for humans to avoid Gulf Coast tick bites is to take personal protective measures. It is best to cover as much exposed skin as possible with clothing if entering tick habitat, which includes knee-high grasses. Tucking the clothing into other clothing, such as pulling socks over trouser bottoms, decreases skin exposure and forces ticks to seek an open area. This makes ticks more likely to be detected, especially on light-colored clothing. Additionally, a repellent should be used on any exposed skin and on clothing (CDC). The most widely used and effective repellants are DEET (skin-use) and permethrin (clothing-use only), but other repellants are available for use. Always check the EPA's repellent website for the most up-to-date information. Finally, after leaving a tick habitat, inspect all clothing and equipment, conduct a full body check, shower within two hours, and place clothes in a dryer on high heat for at least one hour (CDC). Please consult with your veterinarian for the best protection methods to protect cats and dogs from tick bites. Anytime that a tick is found attached to you or your animals, it should be carefully removed and submitted for identification to a local Cooperative Extension office. Ticks can be safely removed by grasping the tick as close to the skin as possible using fine-tipped tweezers then pulling upward steadily, without twisting or jerking, until the mouthparts release. The bite area should be cleaned thoroughly with alcohol or soap and water.
Acaricides can be applied to the host animal or to off-host habitats. Infestations on cattle may be controlled with acaricide-impregnated ear tags, sprays, or pour-on products. To ensure proper application usage and safety for off-host acaricide application, always carefully review the product label and follow application rates and instructions. Regardless of acaricide selection, use the product only in accordance with label and safety instructions. As always, consult your local Cooperative Extension office or veterinarian with any questions about protecting livestock from ticks.
Habitat modification to reduce tick numbers in a given area may be accomplished in a variety of ways. Mowers and select herbicides can be used to reduce brush and weedy tick harborage. Prescribed burning generates temperatures sufficient to kill ticks and remove leaf litter, undergrowth, and other vegetation, which critically impacts off-host tick survival (Stafford et al. 1998; Allan 2009); however, prescribed burn studies have shown variable results as a long-term tick reduction strategy (Ginsberg 2014). As previously mentioned, Gulf Coast ticks retain moisture extremely well and have been shown to survive in areas following prescribed burns (Gleim et al. 2013). If prescribed burns are considered as an option for controlling ticks, check local restrictions and consult with a professional organization specializing in prescribed burn techniques prior to beginning. It is important to continue to monitor tick populations before and after the burns to determine if populations are reduced; moreover, reducing tick populations with fire does not guarantee a reduction in tick-borne pathogens.
Ticks generally require hosts to move them from one area to another. Thus, host exclusion not only involves restricting unwanted wildlife species from an area, it also includes preventing your animals from entering tick habitat. Fencing may limit access to larger tick-infested mammals but does not prevent dispersion of immature Gulf Coast ticks by birds or many small hosts. Lastly, routine inspection of livestock and companion animals should be done to detect Gulf Coast ticks, and other ectoparasites, to prevent incidental introductions.
For more information on tick management see the Tick App for Texas and the Southern Region (Teel et al. 2011)
Acknowledgements
Dr. Susan Little and Lisa Coburn, Oklahoma State University, provided tick specimens for photographs. Dr. Becky Trout Fryxell and Sarah Mays, the University of Tennessee, and Chris Holderman, University of Florida, provided editorial comments. Lyle Buss and Jane Medley assisted with editing photographs.
Selected References
Allan BF. 2009. Influence of prescribed burns on A. americanum (Acari: Ixodidae) in the Missouri Ozarks. Journal of Medical Entomology 46: 1030–1036.
Allan SA, Simmons L, Burridge MJ. 2001. Ixodid ticks on white-tailed deer and feral swine in Florida. Journal of Vector Ecology 26: 93–102.
Baneth G. 2011. Perspectives on canine and feline hepatozoonosis. Veterinary Parasitology 181: 3–11.
Bishopp FC, Hixson H. 1936. Biology and economic importance of the Gulf Coast tick. Journal of Economic Entomology 29: 1068–1076.
Bishopp FC, Trembley HL. 1945. Distribution and hosts of certain North American ticks. The Journal of Parasitology 31: 1–54.
Burgdorfer W. 1957. Artificial feeding of ixodid ticks for studies on the transmission of disease agents. Journal of Infectious Diseases 100: 212–214.
Burridge MJ, Simmons L, Allan SA. 2000. Introduction of potential Heartwater vectors and other exotic ticks into Florida on imported reptiles. The Journal of Parasitology 86: 700–704.
CDC. (2014). Centers for Disease Control and Prevention. Preventing Tick Bites. (7 October 2014).
Cilek JE, Olson MA. 2000. Seasonal distribution and abundance of ticks (Acari: Ixodidae) in northwestern Florida. Journal of Medical Entomology 37: 439–444.
Cragun WC, Bartlett BL, Ellis MW. 2010. The expanding spectrum of eschar-associated rickettsioses in the United States. Archives of Dermatology 146: 641–648.
Deem SL. 1998. A review of Heartwater and the threat of introduction of Cowdria ruminantium and Amblyomma spp. ticks to the American mainland. Journal of Zoo and Wildlife Medicine 29: 109–113.
Drummond RO, Whetstone TM. 1970. Oviposition of the Gulf Coast tick. Journal of Economic Entomology 63: 1547–1551.
Edlow JA. 2010. Tick paralysis. Current Treatment Options in Neurology 12: 167–177.
Edwards KT. 2011. Gotch ear: a poorly described, local, pathologic condition of livestock associated primarily with the Gulf Coast tick, Amblyomma maculatum. Veterinary Parasitology 183: 1–7.
Espinoza-Gomez F, Newton-Sanchez O, Flores-Cazares G, la Cruz-Ruiz De M, Melnikov V, Austria-Tejeda J, Rojas-Larios F. 2011. Tick paralysis caused by Amblyomma maculatum on the Mexican pacific coast. Vector-Borne & Zoonotic Diseases 11: 945–946.
Ewing SA, Panciera RJ. 2003. American canine hepatozoonosis. Clinical Microbiology Reviews 16: 688–697.
Fleetwood SC, Teel PD. 1983. Variation in activity of ageing Amblyomma maculatum Koch (Acarina: Ixodidae) larvae in relation to vapor pressure deficits in pasture vegetation complexes. Protection Ecology 5: 343–352.
Florin DA, Brinkerhoff RJ, Gaff H, Jiang J, Robbins RG, Eickmeyer W, Butler J, Nielsen D, Wright C, White A, Gimpel ME, Richards AL. 2014. Additional U.S. collections of the Gulf Coast tick, Amblyomma maculatum (Acari: Ixodidae), from the State of Delaware, the first reported field collections of adult specimens from the State of Maryland, and data regarding this tick from surveillance of migratory songbirds in Maryland. Systematic & Applied Acarology 19(3): 257–262.
Ginsberg HS. 2014. Tick control. In Sonenshine DE, Roe RM (eds.). Biology of Ticks. pp. 409–444.
Gleim ER, Conner LM, Yabsley MJ. 2013. The effects of Solenopsis invicta (Hymenoptera: Formicidae) and burned habitat on the survival of Amblyomma americanum (Acari: Ixodidae) and Amblyomma maculatum (Acari: Ixodidae). Journal of Medical Entomology 50: 270–276.
Goddard J, Paddock CD. 2005. Observations on distribution and seasonal activity of the Gulf Coast tick in Mississippi. Journal of Medical Entomology 42: 176–179.
Hooker WA, Bishopp FC, Wood HP, Hunter WD. 1912. The life history and bionomics of some North American ticks. USDA, Bureau of Entomology.
Mahan SM, Peter TF, Simbi BH, Kocan K, Camus E, Barbet AF, Burridge MJ. 2000. Comparison of efficacy of American and African Amblyomma ticks as vectors of Heartwater (Cowdria ruminantium) infection by molecular analyses and transmission trials. The Journal of Parasitology 86: 44–49.
Nadolny RM, Wright CL, Sonenshine DE, Hynes WL, Gaff HD. 2014. Ticks and spotted fever group rickettsiae of southeastern Virginia. Ticks and Tick-Borne diseases 5: 53–57.
Paddock CD, Sumner JW, Comer JA, Zaki SR, Goldsmith CS, Goddard J, McLellan SL, Tamminga CL, Ohl CA. 2004. Rickettsia parkeri: a newly recognized cause of spotted fever rickettsiosis in the United States. Clinical Infectious Diseases 38: 805–811.
Paffenbarger RS Jr. 1951. Tick paralysis: implicating Amblyomma maculatum. The New Orleans Medical and Surgical Journal 103: 329–332.
Parola P, Paddock CD, Socolovschi C. 2013. Update on tick-borne rickettsioses around the world: a geographic approach. Clinical Microbiology Reviews 26: 657–702.
Rechav Y, Drey C, Fielden LJ, Goldberg M. 2000. Production of pheromones by artificially fed males of the tick Amblyomma maculatum (Acari: Ixodidae). Journal of Medical Entomology 37: 761–765.
Stafford KC III, Ward JS, Magnarelli LA. 1998. Impact of controlled burns on the abundance of Ixodes scapularis (Acari: Ixodidae). Journal of Medical Entomology 35: 510–513.
Teel P, Strey OF, Hurley JA. (2011). The TickApp for Texas and the southern region: a mobile smartphone application. (7 October 2014).
Teel PD, Ketchum HR, Mock DE, Wright RE, Strey OF. 2010. The Gulf Coast tick: a review of the life history, ecology, distribution, and emergence as an arthropod of medical and veterinary importance. Journal of Medical Entomology 47: 707–722.
Walker DH, Paddock CD, Dumler JS. 2008. Emerging and re-emerging tick-transmitted rickettsial and ehrlichial infections. Medical Clinics of North America 92: 1345–1361.
Wilson DD, Bram RA. 1998. Foreign pests and vectors of arthropod-borne diseases. In Buisch WW, Hyde JL, Mebus CA (eds.). Foreign Animal Diseases. pp. 225–239.
Yoder JA, Christensen BS, Croxall TJ, Schumaker LK, Tank JL. 2008. Moisture requirements for activity/survival of the Gulf Coast tick, Amblyomma maculatum Koch (Acari: Ixodidae), based on a water balance study of all life stages. International Journal of Acarology 34: 285–292.