The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The emerald ash borer, Agrilus planipennis Fairmaire (Figure 1), is a highly destructive wood-boring beetle that feeds on the phloem of ash trees (Fraxinus spp.). Though it has not been found in Florida, there is potential for it to establish via movement of infested wood into the state and the presence of ash trees in Florida. Since first being recorded in Michigan in 2002, the emerald ash borer has broadened its range in the United States and has killed millions of ash trees. (To learn how to inspect your trees for emerald ash borer, click here.)

Credit: Pennsylvania Department of Conservation and Natural Resources—Forestry, Bugwood.org
Synonymy
There are several synonyms of Agrilus planipennis, which are listed below (Jendek 1994):
- Agrilus planipennis Fairmaire (1988)
- Agrilus marcopoli Obenberger (1930)
- Agrilus feretrius Obenberger (1936)
- Agrilus marcopoli ulmi Kurosawa (1956)
Distribution
The first discovery of emerald ash borer in the United States occurred in the summer of 2002, when it was collected from ash trees in Michigan (Haack et al. 2002). Within five years, it had spread to Illinois, Indiana, Maryland, Ohio, Pennsylvania, Virginia, and West Virginia. As of June 2016, emerald ash borer had been confirmed in all of the green states shown in Figure 2, with Texas as the most recent state, confirmed in 2016. It has also been sighted in Ontario and Quebec (EAB Timeline). The native range of emerald ash borer is Russia, Mongolia, Japan, China, and Taiwan (Jendek 1994).
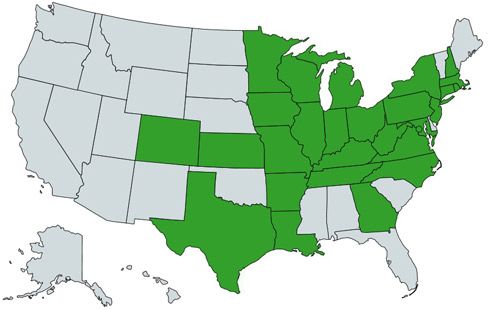
Credit: Haleigh Ray, created with https://mapchart.net/usa.html
Description
The elytra (hardened forewings) of the emerald ash borer are a bright, emerald green color, giving the species its common name (Wang et al. 2010). The dorsal side of the abdomen of the beetle ranges from a golden green to a copper color, with a metallic sheen (Figure 3).

Credit: Lyle J. Buss, UF/IFAS
Life Cycle
Eggs
Emerald ash borer eggs are deposited in tree bark crevices for protection, which makes it difficult to determine the total fecundity of adult female beetles in the field. In one study, dissections of females showed an average of 32 fully formed eggs and 71 immature eggs per female (Wang et al. 2010). Another study found females to lay 40 to 53 eggs (Rutledge 2012). Eggs begin as an ivory color but darken to brown three to four days after being deposited. They are approximately 1.23 mm in length and just under 1 mm in width. The surrounding environment plays a role in egg development. Eggs held at 18°C–23°C hatched in 17 to 19 days, while eggs at 24°C–26°C hatched in 12 to 13 days (Wang et al. 2010).
Larvae
The larval stage of the emerald ash borer is the longest stage of the beetle's life cycle, lasting approximately 300 days and four instars (Wang et al. 2010). The larvae are translucent and ivory colored, with a brown head covered mostly by the prothorax (Figure 4). They reach a length of approximately 25–32 mm (Emerald Ash Borer Information Network 2016). The mouthparts are the only visible external structure on the head (Wang et al. 2010). Larvae bore through bark and begin to feed in the outer phloem of trees, creating curved galleries (Figure 5). The larvae complete their four instars during summer and fall while feeding and overwinter in the fourth instar in a prepupal stage (Wang et al. 2010, Herms and McCullough 2014).
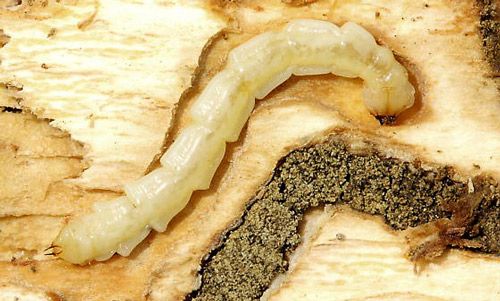
Credit: Stephen Luk, Toronto

Credit: Stephen Luk, Toronto
Pupae
The emerald ash borer pupal stage is short, only lasting an average of 20 days at 18°C–20°C (Wang et al. 2010, Herms and McCullough 2014). Pupae are 11–16 mm in length, and 3–5 mm in width.
Adults
The adults of the emerald ash borer (Figure 6) chew through the wood and emerge from trees from a small, D-shaped exit hole about 3–4 mm in width (Figure 7) (Wang et al. 2010). In a laboratory study, the average longevity of the adult females was 20.5 days (range of 3 to 52 days) after emergence from the exit hole, and males lived an average of 22.8 days (range of 3 to 53 days) after emergence (Wang et al. 2010).

Credit: Stephen Luk, Toronto

Credit: Stephen Luk, Toronto
Host Plants
In North America, Agrilus planipennis has killed both healthy and stressed green ash (Fraxinus pennsylvanica), white ash (Fraxinus americana), black ash (Fraxinus nigra), and blue ash (Fraxinus quadrangulata). So far, there have been no observed emerald ash borer attacks on non-ash trees in North America (Anulewicza et al. 2008). Outside of North America, emerald ash borers (Agrilus planipennis and synonymous species) have been reported from Juglans spp., Pterocarya spp., and Ulmus spp., in addition to Fraxinus spp. (Anulewicza et al. 2008). Several of these genera have common species in North America, such as the American elm (Ulmus americana) and black walnut (Juglans nigra), that are found in both forest and landscape settings.
In a study by Anulewicza et al. (2008), choice tests were conducted using black walnut and American elm to determine whether emerald ash borer would land on the alternative (non-ash) hosts, whether the females would oviposit in these hosts, and if so, whether the larvae would develop successfully. It was found that even when the Agrilus planipennis females landed on non-ash trees and deposited eggs, the larvae did not develop on species other than Fraxinus spp. (Anulewicza et al. 2008). The beetles non-discriminately landed on all three tree species, but almost 80% of the eggs found were from Fraxinus logs, suggesting the females distinguish between the tree species and choose ash trees for oviposition (Anulewicza et al. 2008).
Economic Importance
Since the emerald ash borer's discovery in the United States, it is estimated that over 53 million ash trees (Fraxinus spp.) have been killed throughout Michigan, Indiana, and Ohio (Kovacs et al. 2010). In the forests of southeastern Michigan, the emerald ash borer has killed over 99% of three species of ash trees (Fraxinus nigra, Fraxinus americana, and Fraxinus pennsylvanica, these are the black, white, and green ash, respectively) (Herms and McCullough 2014). Because ash trees are popular in urban landscapes, much of the damage has been noted from developed areas. This gives the emerald ash borer the potential to cause damage to ash trees throughout the United States, as the trees are widespread (MacFarlane and Meyer 2005). Symptoms of emerald ash borer damage include the presence of D-shaped emergence holes (Figure 7), bark splitting, and tree canopy dieback (Figure 8).

Credit: Damaged tree: Stephen Luk, Toronto; healthy tree: NetPS Plant Finder
One study by Kovacs et al. (2010) used computer simulations to estimate a range of emerald ash borer infestation and the associated costs, by the year 2019. The simulation encompassed 25 states, and predicted the mean cost of treatment, removal, and replacement of infested ash trees on developed land would be approximately $10.7 billion. Another study by Sydnor et al. (2007) estimated the cost to be approximately $25 billion if all ash trees were to be removed and replaced with a different type of tree.
The infestation has already affected many plant-related industries, including nurseries, sawmills, logging, and producers that use ash wood for tools, pallets, and railroad ties (Herms and McCullough 2014). As the emerald ash borer continues to spread to new states, these industries in those states could be affected as well.
Management
Due to the considerable impact the emerald ash borer causes, the national Emerald Ash Borer Science Advisory Panel has recommended a program to slow the spread of the insect and reduce the densities of the existing populations (Poland and McCullough 2006). One major component of this program is the restricted movement of potentially infested material that can harbor the emerald ash borer. United States federal quarantines regulate movement of any ash material that may be infested, including trees, limbs, cut firewood, logs, and bark chips, and limit nursery sales of ash trees in Michigan (Poland and McCullough 2006). Once products are cleared from quarantine they can be moved. In order to detect emerald ash borer presence and infestation levels, traps baited with chemical lures mimicking host volatiles (chemicals released from the plant) are suspended in ash trees (Figure 9). Currently in Florida, USDA and FDACS are monitoring in north, central, and southwest Florida for emerald ash borer using this method (Douglas Restom Gaskill, pers. comm.).

Credit: Kansas Department of Agriculture
Several strategies discussed below show an integrated approach to slowing the mortality of ash trees but are not expected to eradicate the pest. Even though the emerald ash borer would not be eradicated, the lowered populations can allow for a long-term management solution to be developed through research (Herms and McCullough 2014).
Biological Control
Quickly after emerald ash borer was detected in North America, researchers began trying to find natural enemies as a control method (Liu et al. 2003, Bauer et al. 2004). Studies found that the greatest factor affecting emerald ash borer mortality was woodpeckers, which feed on late instar larvae (Cappaert et al. 2005, Duan et al. 2010). In 2007, Cappaert and McCullough (2009) discovered a high level of emerald ash borer larvae at a heavily infested site in southeast Michigan that were parasitized by a native wasp, Atanycolus cappaerti (Hymenoptera: Braconidae). Parasitism by this species has been observed frequently and appears to be present at high levels where the emerald ash borer is well established. Rates of parasitism by other native parasitoid species is typically low (Duan et al. 2009).
In an effort to establish a classical biological control program, researchers began to identify parasitoids of emerald ash borer in China, with the goal of mass producing and releasing parasitoid species in the United States. There are currently three species being released for control: Oobius agrili Zhang and Huang (Hymenoptera: Encyrtidae), an egg parasitoid, Tetrastichus planipennisi Yang (Hymenoptera: Eulophidae), and Spathius agrili Yang (Hymenoptera: Braconidae), a larval endo- and ectoparasitoid, respectively (USDA APHIS 2010). Areas with low population densities of emerald ash borer are more likely to be successfully managed with biological control by either native or introduced parasitoid species (Herms and McCullough 2014).
Insecticidal Control
In addition to biological control, chemical control is also used. A variety of insecticides are available, such as sprays and products for injection into tree trunks and soil (Poland and McCullough 2006). Despite the range of existing insecticidal products, early studies found that treatment with these products has yielded inconsistent results (Herms and McCullough 2014). However, control of emerald ash borer can be consistent when variables such as application rates and methods, timing of application, and tree condition are taken into consideration (Herms et al. 2014).
Selected References
Anulewicza AC, McCullough DG, Cappaert DL, Poland TM. 2008. Host range of the emerald ash borer (Agrilus planipennis Fairmaire) (Coleoptera: Buprestidae) in North America: Results of multiple-choice field experiments. Environmental Entomology 37: 230–241.
Bauer LS, Liu HP, Haack RA, Petrice TR, Miller DL. 2004. Natural enemies of emerald ash borer in southeastern Michigan. In Emerald Ash Borer Research and Technology Development Meeting: pp. 33–34.
Cappaert D, McCullough DG. 2009. Phenology of Atanycolus cappaerti (Hymenoptera: Braconidae), a native parasitoid of emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae). Great Lakes Entomology 41: 141–154.
Cappaert D, McCullough DG, Poland TM, Siegert NW. 2005. Emerald ash borer in North America: A research and regulatory challenge. American Entomologist 51: 152–165.
Duan JJ, Fuester RW, Wildonger J, Taylor PB, Barth S, Spichiger SE. 2009. Parasitoids attacking the emerald ash borer (Coleoptera: Buprestidae) in western Pennsylvania. Florida Entomologist 92: 588–592. (1 March 2022)
Duan JJ, Ulyshen MD, Bauer LS, Gould J, van Driesche R. 2010. Measuring the impact of biotic factors on populations of immature emerald ash borer (Coleoptera: Buprestidae). Environmental Entomology 39: 1513–1522.
EAB Timeline. USDA Forest Service and Michigan State University. (5 September 2019)
Emerald Ash Borer Information Network. (2016). USDA Forest Service and Michigan State University. (5 September 2019)
Haack RA, Jendek E, Liu H, Marchant KR, Petrice TR, Poland TM, Ye H. 2002. The emerald ash borer: A new exotic pest in North America. Newsletter of the Michigan Entomological Society 47: 1–5.
Herms DA, McCullough DG. 2014. Emerald ash borer invasion of North America: History, biology, ecology, impact and management. Annual Review of Entomology 59: 13–30.
Herms DA, McCullough DG, Smitley DR, Clifford CS, Cranshaw W. 2014. Insecticide options for protecting ash trees from emerald ash borer. North Central IPM Center Bulletin. Second Edition. 16 pp.
Jendek E. 1994. Studies in the East Palearctic species of the genus Agrilus Dahl, 1823 (Coleoptera: Buprestidae). Entomological Problems 25: 9–25.
Kovacs KF, Haight RG, McCullough DG, Mercader RJ, Siegert NW, Liebhold AM. 2010. Cost of potential emerald ash borer damage in U.S. communities, 2009-2019. Ecological Economics 69: 569–578.
Liu H, Bauer LS, Gao R, Zhao T, Petrice TR, Haack RA. 2003. Exploratory survey for emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae), and its natural enemies in China. The Great Lakes Entomologist 36: 191–204. (26 September 2022)
MacFarlane DW, Meyer SP. 2005. Characteristics and distribution of potential ash tree hosts for emerald ash borer. Forest Ecology and Management 213: 15–24.
Poland TM, McCullough DG. 2006. Emerald ash borer: Invasion of the urban forest and the threat to North America's ash resource. Journal of Forestry 104: 118–124.
Rutledge CE, Keena MA. 2012. Mating frequency and fecundity in the emerald ash borer Agrilus planipennis (Coleoptera: Buprestidae). Arthropod Biology 105: 66–72.
Sydnor TD, Bumgardner M, Todd A. 2007. The potential economic impacts of emerald ash borer (Agrilus planipennis) on Ohio, U.S., communities. Arboriculture & Urban Forestry 33: 48–54.
USDA APHIS. 2010. Emerald ash borer, Agrilus planipennis (Fairmaire), biological control release and recovery guidelines. USDA-APHIS-ARS-FS, Riverdale, Maryland. https://www.aphis.usda.gov/plant_health/plant_pest_info/emerald_ash_b/downloads/EAB-FieldRelease-Guidelines.pdf (5 September 2019)
Wang XY, Yang ZQ, Gould JR, Zhang YN, Liu GJ, Liu ES. 2010. The biology and ecology of the emerald ash borer in China Journal of Insect Science 10: 1–23.