The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The southern green stink bug, Nezara viridula (Linnaeus), is in the order Hemiptera or "true bugs." Stink bugs are in the family Pentatomidae, and adults are recognized by their shield-shape, five-segmented antennae, and their malodorous scent. The southern green stink bug is a highly polyphagous feeder, attacking many important food crops.

Credit: James Castner, UF/IFAS
Distribution
The southern green stink bug is believed to have originated in Ethiopia. Its distribution now includes the tropical and subtropical regions of Europe, Asia, Africa, and the Americas. In South America, it is expanding its range to Paraguay, south Argentina, and toward the northeast of Brazil, due to expanding soybean production (Panizzi 2008). In North America, it is limited primarily to the southeastern United States, Virginia to Florida in the east, Ohio and Arkansas in the midwest, and to Texas in the southwest. It is also established in Hawaii and California (Capinera 2001).
Description
Adult
The adult is shield-shaped with an overall dull green color. The eyes are dark red or black. Small black dots can be found along the sides of the abdomen. The wings completely cover the abdomen. The males average 12.1 mm (~½ in) in length and females 13.15 mm (~½ in) in length. Copulation may last a few minutes to a few days. Females can lay eggs three to four weeks after becoming adults. The average female lays one egg mass but production of two egg masses is not uncommon. A female southern green stink bug could lay as many as 260 eggs over her life span.

Credit: Paul Choate, UF/IFAS
This species should not be confused with the green stink bug Chinavia halaris (Say). The range of Chinavia halaris extends farther north than Nezara viridula and there are physical differences. The shape of the stink gland pore, located on the sternum between the second and third leg, is short and broad in the southern green stink bug, but long and curved in the green stink bug.
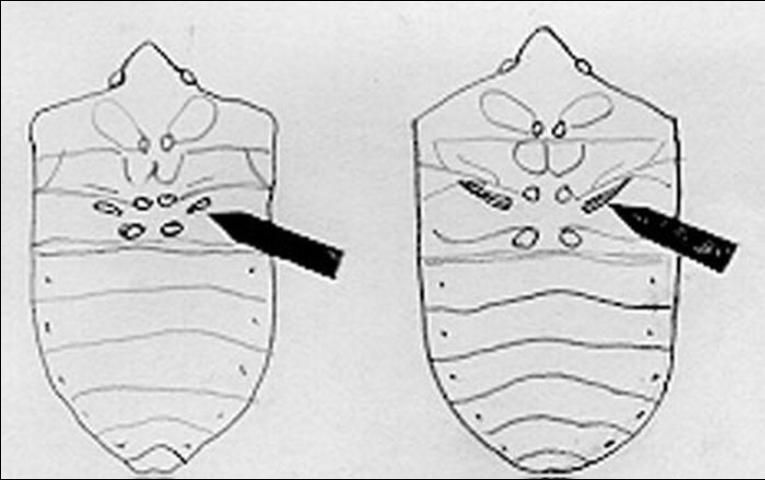
Credit: Jason M. Squitier, UF/IFAS
Eggs
Eggs have been found as early as the second week of April and as late as December 12th. The eggs are deposited in masses that range from 30 to 130 eggs per mass. The female oviposits on the undersurface of leaves in the upper portions of canopied crops and weeds. Weeds that are favored by the southern green stink bug include beggarweed, rattlebox, Mexican clover, wild blackberry, and nut grass. The eggs are firmly glued to each other and to the substrate. The eggs are white to light yellow in color and barrel shaped with flat tops with a disc shaped lid. There are 28 to 32 finger-like projections around the lid called chorial processes. The egg is 1/20 of an inch in length and 1/29 inch wide. The incubation time for the eggs is five days in the summer and two to three weeks in early spring and late fall. As incubation continues the eggs turn pinkish in color.

Credit: James Castner, UF/IFAS
Nymph
The nymph hatches from the egg by opening the disc shaped cap. The nymph slowly wiggles out of the shell. Each hatchling takes five to six minutes to escape from the egg, and the entire pod hatches in 1.5 hours. The first instars aggregate by the empty eggs and do not feed. The possible benefits of aggregation are to deter predation from the pooling of their chemical defenses. The nymphs are light yellowish in color with red eyes and transparent legs and antennae. The time until the next molt is three days. Feeding begins with the second instar. The second instar has black legs, head, thorax, and antennae. The abdomen is red and so are the spaces between the second, third, and fourth antennal segments. The thorax has a yellow spot on each outer side. The second instar lasts five days. The third and fourth instars differ from the second in size and an overall greenish color is becoming apparent. Each of these instars lasts seven days. Wing pads mark the arrival of the fifth instar. The abdomen is yellowish green with red spots on the median line. The southern green stink bug usually spends eight days as a fifth instar before the final molt to an adult.

Credit: Herb Pilcher, USDA-ARS

Credit: Herb Pilcher, USDA-ARS

Credit: Herb Pilcher, USDA-ARS

Credit: Herb Pilcher, USDA-ARS

Credit: Herb Pilcher, USDA-ARS
Life Cycle
The southern green stink bug can complete its life cycle in 65 to 70 days. It is most prevalent during the periods of October through December and again in March through April. The southern green stink bug is known to have up to four generations per year in warm climates. The southern green stink bug overwinters as an adult, and hides in the bark of trees, leaf litter, or other locations to obtain protection from the weather. As spring temperatures begin to warm, the southern green stink bug moves out of the winter cover and begins feeding and oviposition.
Damage
The southern green stink bug has piercing-sucking mouthparts. The mouth consists of a long beak-like structure called the rostrum. Salivary fluid is pumped down the salivary duct and liquefied food is pumped up the food canal. All plant parts are likely to be fed upon, but growing shoots and developing fruit are preferred. Attached shoots usually wither, or in extreme cases may die. The damage on fruit from the punctures is hard brownish or black spots. These punctures affect the fruit's edible qualities and decidedly lower its market value. Young fruit growth is retarded, and it often withers and drops from the plant. In addition to the visual damage caused by southern green stink bug feeding, the mechanical transmission of tomato bacterial spot may also result.

Credit: Ronald Smith, Auburn University, insectimages.org
Action Threshold
The economic threshold for southern green stink bug in soybeans is reported as 36 stink bugs per 100 swings of a net. For cowpea (southern pea), a population level of 5000 southern green stink bugs per hectare would be large enough to downgrade peas from Grade A to Grade B. In cotton an accepted threshold is three to four stink bugs per 100 swings with a sweep net.

Credit: James Castner, UF/IFAS

Credit: Commonwealth Scientific and Industrial Research Organization, Australia (CSIRO)
Management
Biological Control
Parasitoids, usually wasps and flies, provide biological control of the southern green stink bug. In Florida a tachinid fly, Trichopoda pennipes, parasitizes adults and nymphs; and a wasp, Trissolcus basalis, parasitizes eggs. These two parasites have also been introduced as biological control agents in other areas, such as Australia and Hawaii, to control the southern green stink bug. California used Trissolcus basalis in an effort to control its southern green stink bug population.
Chemical Control
The use of trap crops is not a widely accepted idea for control of the southern green stink bug, but it has excellent potential as a type of control. The choice for trap crops in the summer would be leguminous plants such as cowpeas and beans. In the late fall and early spring cruciferous plants are recommended. The trap crop should be sprayed or plowed under before the developing southern green stink bugs become adults to prevent them from migrating to the main crop. Insecticides are commonly applied at blossom and fruit formation.
Florida Insect Management Guide for okra
Florida Insect Management Guide for tomatoes, peppers, and eggplant
Selected References
Buschman LL. 1980. Parasites of Nezara viridula (Hemiptera: Pentatomidae) and other Hemiptera in Florida. Florida Entomologist 63: 154–162.
Capinera JL. 2001. Handbook of Vegetable Pests. Academic Press, San Diego. 729 p.
Drake CJ. 1920. The southern green stink bug in Florida. State Plant Board of Florida Quarterly Bulletin IV: 41–93.
Hoffmann MP et al. 1991. Imported wasp helps control southern green stink bug. California Agriculture 45: 20–22.
Jones TH. 1918. The southern green plant bug. United States Department of Agriculture Bulletin 689: 2–26.
Lye BH, Story RN, Wright VL. 1988. Southern green stink bug (Hemiptera: Pentatomidae) damage to fresh market tomatoes. Journal of Economic Entomology 81: 190–194.
McPherson JE. 1982. The Pentatomoidea (Hemiptera) of Northeastern North America. Southern Illinois University Press, Illinois. 240 pp.
McPherson RM, Newsom LD. 1984. Trap crops for control of stink bugs in soybean. Journal of the Georgia Entomological Society 19: 470–480.
Panizzi AR. 2008. Southern green stink bug, Nezara viridula (L.) (Hemiptera: Heteroptera: Pentatomidae). pp. 3471–3471. In Encyclopedia of Entomology. Capinera JL (editor). Springer, Heidelberg.
Slater JA, Baranowski RM. 1978. True Bugs. Wm. C. Brown Co. Publishers Dubuque, Iowa. 256 pp.
Stam PA, Newsom LD, Lambremont EN. 1987. Predation and food as factors affecting survival of Nezara viridula (L.) (Hemiptera: Pentatomidae) in a soybean ecosystem. Environmental Entomology 16: 1211–1216.
Todd JW. 1989. Ecology and behavior of Nezara viridula. Annual Review of Entomology 34: 273–292.