Introduction
Tamarixia radiata (Waterston) (Hymenoptera: Eulophidae) is an effective ectoparasitoid of the Asian citrus psyllid, Diaphorina citri Kuwayama (Hemiptera: Psyllidae). Diaphorina citri is one of the most serious pests of citrus worldwide because it vectors the bacterial pathogen causing huanglongbing (HLB) disease in citrus. In addition, both nymphs and adults are obligate phloem feeders that cause chlorosis on infested leaves and excrete honeydew that promotes the growth of sooty mold. HLB affects plant phloem, causing yellow shoots, mottling, chlorosis, and twig die back which cause rapid tree decline and may ultimately cause tree death. Fruit on diseased trees do not color properly, and can be bitter tasting and misshapen as well as reduced in size (Capoor 1963; Halbert and Manjunath 2004; Bové 2006).
In the United States, Diaphorina citri was first discovered in Palm Beach County, Florida on orange jasmine, Murraya paniculata (L.) Jack. (Rutaceae) in 1998 (Halbert 1998). It is now a serious pest of citrus in Florida (Michaud 2002; Halbert and Manjunath 2004), and specimens have been found throughout the US, including Florida, Texas, Hawaii, Louisiana, Alabama, Georgia, Mississippi, South Carolina, and California (National Invasive Species Information Center).
Currently all possible vector and disease control methods are being employed to manage HLB in Florida including biological control with Tamarixia radiata (Waterston) (Hymenoptera: Eulophidae) and Diaphorencyrtus aligarhensis (Shafee, Alam and Agarwal) (Hymenoptera: Encyrtidae) (Hoy et al. 2006; Qureshi et al. 2009). Tamarixia radiata is presumed superior to Diaphorina aligarhensis based on previous reports of high psyllid parasitization rates and rapid establishment in new areas (Aubert 1987, Skelly and Hoy 2004). Release of Tamarixia radiata revived the citrus industry in Reunion Island after its introduction from India in 1978 (E tienne et al. 2001) and the parasitoid caused substantial decline in Diaphorina citri populations in Guadeloupe Island within one year of release (Aubert and Quilici 1984; E tienne et al. 2001).
Distribution
Tamarixia radiata was discovered in the area of northwestern India (Punjab) which is now within present-day Pakistan (Waterston 1922). Because of the reported high parasitism efficiency and establishment rate of Tamarixia radiata, the parasitoid was imported into several countries, including the United States, to control Diaphorina citri.
Currently, the parasitoid is found in Brazil, China, Guadaloupe, Indonesia, Mauritius Mexico, Pakistan, Philippines, Nepal, Taiwan, Vietnam, Puerto Rico, and the United States (Waterston 1922; Chien et al. 1991a; Hoy and Nguyen 2001; E tienne et al. 2001; Halbert and Manjunath 2004; Pluke et al. 2008; León and Setamou 2010).
Description and Life Cycle
Adults
Tamarixia radiata adults are small black wasps (0.92 to 1.04 mm long) with widely separated eyes. The adult's head is slightly larger in width than length. The wings are hyaline with pale yellow veins. Sexual dimorphism is marked between the male and female adults. The male antennae are 1.5 times longer than those of females (Onagbola et al. 2009). Males are slightly smaller than females in total length and wing expanse. The female ovispositor is barely protruding (Waterson 1922).

Credit: Angel Hoyte and Jamie D. Yates, UF/IFAS
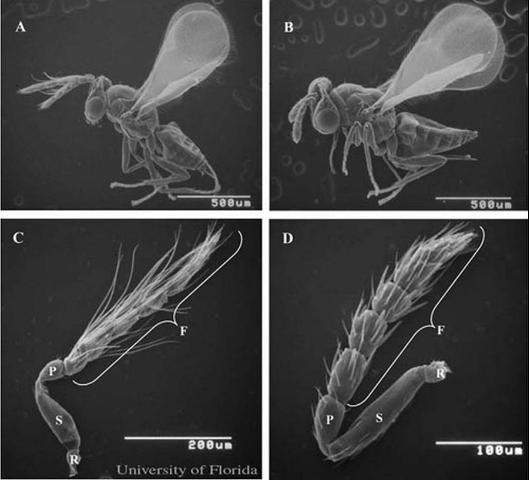
Credit: Ebenezer Onagbola and Diann Achor, UF/IFAS
Eggs
The adult female Tamarixia radiata lays one or occasionally two eggs beneath a Diaphorina citri nymph. Although more than one egg may sometimes be laid beneath a nymph, only one parasitoid larva usually reaches the adult stage and thus Tamarixia radiata is regarded as a solitary parasitoid. An adult female Tamarixia radiata can deposit up to 300 eggs (Hoy et al., 2006).
Larvae
The newly hatched parasitoid larvae feed on hemolymph from the site of attachment, eventually killing the hosts, Diaphorina citri nymphs. The first instar parasitoid larvae are about 0.28 mm long and 0.11 mm wide, while the fourth instar larvae are 1.14 mm long and 0.59 mm wide.
http://entnemdept.ifas.ufl.edu/creatures/beneficial/wasps/tamarixia_radiata06.avi video of Tamarixia radiata larva, just prior to pupation, inside Diaphorina citri nymph. (12 MB avi file courtesy of C. N. Rao, National Research Center for Citrus, Nagpur, India)
Pupae
Pupation occurs within the mummified Diaphorina citri nymphs and new adults emerge through a hole on the thorax or head of the parasitized mummy.

Credit: Angel Hoyte and Jamie D. Yates, UF/IFAS

Credit: Angel Hoyte, Jamie D. Yates, UF/IFAS
http://entnemdept.ifas.ufl.edu/creatures/beneficial/wasps/tamarixia_radiata07.wmv video of adult Tamarixia radiata emerging from mummified nymph of Diaphorina citri. (91 MB wmv file by Angel Hoyte and Jamie D. Yates, University of Florida)
Under experimental conditions (26±1°C, 70% RH), the total developmental period (egg to adult) for the wasps is completed in 11.4 days. The egg, larval, prepupal, and pupal stages are completed in 1.9, 4.0, 0.6 and 4.9 days, respectively.
The average longevity of the female adult (23.6 days) is greater than that of the male (11.4 days). Males are capable of multiple matings. However, mating has no effect on longevity of adults (Chien et al. 1991b). The female to male sex ratio is 1.8 to 3.2, depending upon the origin of the colony and the rearing conditions (Chien 1995; Skelley and Hoy 2004; Hall 2008).
The adult Tamarixia radiata parasitoid emerges from the Diaphorina citri nymph's thorax, leaving a round emergence hole visible without magnification. Tamarixia radiata females parasitize all immature stages of Diaphorina citri and show a significant preference for 5th-instar nymphs (Chien et al., 1991b, Hoy et al., 2006).
In addition to killing nymphs through parasitism, adult females feed on younger nymphs (Chein 1995, Skelley and Hoy 2004). Female Tamarixia radiata may obtain protein for egg development by feeding on hemolymph from psyllid nymphs, which is accessed through ovipositor-induced punctures (Hoy et al. 2006). A single Tamarixia radiata female is able to kill over 500 psyllids by a combination of host feeding and parasitism. Females also feed on honeydew excreted by psyllids (Hoy et al. 2006). Tamarixia radiata adults are strongly attracted to bright fluorescent lights (Skelley and Hoy 2004) and females primarily rely on olfactory cues for host location (Mann et al. 2010).
Hosts
Tamarixia radiata is not known to attack any other psyllid species than Diaphorina citri (Aubert and Quilici 1984).
Tamarixia radiata use in Classical Biological Control of Diaphorina citri in Florida
Classical biological control of Diaphorina citri was initiated in Florida in 1999. Colonies of Tamarixia radiata and Diaphorina aligarhensis were imported from Taiwan and Vietnam, respectively (Hoy and Nguyen, 2001). In total, 12,000, 16,800, and 8,000, adults of a mixed colony from the two origins were released in Florida in 1999, 2000, and 2001, respectively (Skelley and Hoy 2004). The parasitoid dispersed quickly and established throughout the major citrus-growing regions of the State (Hoy and Nguyen 2001).
Currently, the parasitoid is also found in Texas and Puerto Rico where it was not released intentionally. While Tamarixia radiata has a short generation time and high reproductive rate in the laboratory, its effectiveness in suppressing psyllid populations under field conditions has been variable in Florida and other adjoining regions (Michaud 2004; Pluke et al. 2008; Qureshi et al. 2009). Parasitism rates have averaged less than 20% during spring and summer, increasing to 39–56% in the fall as compared to 79 and 88% in Puerto Rico (Pluke et al. 2008). Parasitism rates in Florida are lower than observed in Reunion Island, Guadaloupe, and Puerto Rico.
No hyperparasitoid species attacking Tamarixia radiata have yet been observed in the United States (Hall 2008). However, 64–100% mortality of Tamarixia radiata has been reported from intraguild predation (killing and eating potential competitors) by coccinellid species in Florida (Michaud 2004; Qureshi et al. 2009). Coccinellids consume parasitized nymphs containing Tamarixia radiata larvae thus reducing populations of Tamarixia radiata. In addition, the more extreme climatic conditions of Florida and current intense use of insecticides to control Diaphorina citri populations are presumed to be responsible for low parasitoid populations in Florida.
Selected References
Aubert B. 1987. Trioza erytreae del Guercio and Diaphorina citri Kuwayama (Homoptera: Psylloidea), the two vectors of citrus greening disease: Biological aspects and possible control strategies. Fruits 42: 149-162.
Aubert B, Quilici S. 1984. Biological control of the African and Asian citrus psyllids (Homoptera: Psylloidea), through eulophid and parasites (Hymenoptera: Chalcidoidea) in Reunion Island. pp. 100-108. In Garnsey SM, Timmer LW, Dodds JA (eds.).1993. Proceedings of the 9th Conference of the International Organization of Citrus Virologists, University of California, 9-13 May, Riverside, CA. University of California, Riverside.
Bové JM. 2006. Huanglongbing: A destructive, newly-emerging, century-old disease of citrus. Journal of Plant Pathology 88: 7-37.
Capoor SP. 1963. Decline of citrus trees in India. Bulletin of National Institute of Science India 24, 48-64.
Chien CC, Chu YI, Ku SC. 1991a. Biological control of citrus psyllid, Diaphorina citri, in Taiwan. II. evaluation of Tamarixia radiata and Diaphorencyrtus diaphorinae for the control of Diaphorina citri. Chinese Journal of Entomology 11: 25-38.
Chien CC, Chu YI, Ku SC. 1991b. Parasite strategy, morphology and life history of Tamarixia radiata (Hymenoptera, Eulophidae). Chinese Journal of Entomology 11: 264-281.
Chien CC. 1995. The role of parasitoids in the pest management of citrus psyllid. Proceedings of the Symposium on Research and Development of Citrus in Taiwan, Taichung, Taiwan, pp. 245-261.
E´tienne J, Quilici S, Marival D, Franck A. 2001. Biological control of Diaphorina citri (Hemiptera: Psyllidae) in Guadeloupe by imported Tamarixia radiata (Hymenoptera: Eulophidae). Fruits (Paris) 56: 307-315.
Halbert SE. 1998. Entomology Section. Tri-ology. https://www.researchgate.net/publication/291292753_Entomology_section (28 February 2022).
Halbert SE, Manjunath KL. 2004. Asian citrus psyllids (Sternorrhyncha: Psyllidae) and greening disease in citrus: A literature review and assessment of risk in Florida. Florida Entomologist 87: 330-353.
Hall DH. 2008. Biological control of Diaphorina citri. Proceedings of the International Workshop on Huanglongbing of citrus (Candidatus Liberibacter) and the Asian Citrus Psyllid (Diaphorina citri) 7-9 May 2008, Hermosillo, Sonora, Mexico.
Hoy MA, Nguyen R. 2001. Classical biological control of Asian citrus psylla. Citrus Industry 81: 48-50.
Hoy MA, Nguyen R, Jeyparakash A. (2006). Classical biological control of Asian citrus psyllid in Florida. Florida IPM. https://ipm.ifas.ufl.edu/Agricultural_IPM/psyllid.shtml (15 May 2014).
León JH, Sétamou M. 2010. Molecular evidence suggests that populations of the Asian citrus psyllid parasitoid Tamarixia radiata (Hymenoptera: Eulophidae) from Texas, Florida and Mexico represent a single species. Annals of the Entomological Society of America 103: 100-120.
Mann RS, Rouseff RL, Smoot JM, Castle WJ, Stelinski LL. 2010. Sulfur volatiles from Allium spp. affect Asian citrus psyllid, Diaphorina citri Kuwayama (Hemiptera: Psyllidae) response to citrus volatiles. Bulletin of Entomological Research (In press).
Michaud JP. 2002. Invasion of the Florida citrus ecosystem by Harmonia axyridis (Coleoptera: Coccinellidae) and asymmetric competition with a native species, Cycloneda sanguinea. Environmental Entomology 31: 827-835.
Michaud JP. 2004. Assessment of cotton as an alternative host plant for the brown citrus aphid, Toxoptera citricida (Homoptera: Aphididae). Florida Entomologist 87: 105-111.
Onagbola EO, Boina DR, Hermann SL, Stelinski LL. 2009. Antennal sensilla of Tamarixia radiata (Hymenoptera: Eulophidae), a parasitoid of Diaphorina citri (Hemiptera: Psyllidae). Annals of the Entomological Society of America 102: 523-531.
Pluke RWH, Qureshi JA, Stansly PA. 2008. Citrus flushing patterns, Diaphorina citri (Hemiptera: Psyllidae) populations and parasitism by Tamarixia radiata (Hymenoptera: Eulophidae) in Puerto Rico. Florida Entomologist 91: 36-42.
Qureshi JA, Rogers ME, Hall DG, Stansly PA. 2009. Incidence of invasive Diaphorina citri (Hemiptera: Psyllidae) and its introduced parasitoid Tamarixia radiata (Hymenoptera: Eulophidae) in Florida citrus. Journal of Economic Entomology 102: 247-256.
Skelley LH, Hoy MA. 2004. A synchronous rearing method for the Asian citrus psyllid and its parasitoids in quarantine. Biological Control 29: 14-23.
Waterston J. 1922. On the chalcidoid parasites of psyllids (Hemiptera, Homoptera). Bulletin of Entomological Research 13: 41-58.