Introduction
Orange spiny whitefly, Aleurocanthus spiniferus Quaintance, is a native pest of citrus in tropical Asia. In the early 1920s, pest outbreak infestation levels caused Japan to begin a biological control program. Primarily, orange spiny whitefly affects host plants by sucking the sap but it also causes indirect damage by producing honeydew and subsequently promoting the growth of sooty mold. Sooty mold is a black fungus that grows on honeydew. Heavy infestations of orange spiny whitefly, or other honeydew-producing insects such as scales, mealybugs, aphids, and other whitefly species, can cause sooty mold to completely cover the leaf surface and negatively affect photosynthesis.
Distribution
The orange spiny whitefly has spread to Africa, Australia, the Caribbean and the Pacific Islands (Nguyen et al. 1993). In the Caribbean Islands, Gowdey (1922) recorded orange spiny whitefly as an occasional pest of citrus in Jamaica. For the Pacific Islands, it was first recorded in Guam in 1951 where it was observed not only on citrus, but also on rose, grape, peach, pear, and guava (Peterson 1955). Orange spiny whitefly was first detected on rose foliage in Honolulu, Oahu in 1974. Subsequent surveys discovered it on navel orange, lime, tangerine, and pear, but infestations were reportedly low (USDA 1974). In Africa, the first report of this pest was in 1987 in Swaziland and in 1988 in South Africa (Van den Berg et al. 1990). More recently, orange spiny whitefly was reported from Italy (2008), Croatia (2012), and Montenegro (2013) (Radonjic et al. 2014). Established populations of orange spiny whitefly are not yet known to occur in the continental US.
Description and Life History
Whiteflies have six developmental stages: egg, crawler (1st instar), two sessile nymphal instars (2nd and 3rd instars), the pupa (4th instar), and adult. Identification of the Aleyrodidae is largely based upon characters found in the pupal (4th instar) stage. The duration of the life cycle and the number of generations per year are greatly influenced by the prevailing climate. A mild temperature with high relative humidity provides ideal conditions for growth and development. About four generations per year have been recorded in Japan (Kuwana et al. 1927), while reports from Guam showed the presence of five or six generations (Peterson 1955). As many as seven generations occurred under ideal laboratory conditions. Kuwana et al. (1927) in Japan showed wide variability in life cycle duration, particularly in the 3rd and 4th instar: egg stage, 11 to 22 days; 1st instar, seven to 11 days; 2nd instar, five to seven days; 3rd instar, seven to 130 days; pupal stage, seven to 34 days.
Egg: The egg is minute (0.2 x 0.1 mm), yellow, stalked, curved, and marked with polygonal areas (Quaintance 1903, Quaintance and Baker 1917). The stalk is short and holds the egg in more or less an upright position on the leaf.
Larva: The larva is elliptical or oval-shaped, brown to black, and a short fringe of wax surrounds its body. As the orange spiny whitefly develops to the sessile nympal stage, the second instar is about 0.4 x 0.3 mm and has a distinctly crenulated marginal edge. Additionally, the second instar has short, acute incisions between the marginal wax tubes. Also, in the 2nd and 3rd instar stages, abdominal segments are more distinct than thoracic segments. The dorsal or upper side has a single row of eight abdominal and six thoracic strong, heavy spines on either side of the mid-dorsal line of the body.
Pupa: The oval-shaped pupa or 4th instar is jet black, convex, approximately 1.23 mm long and 1.88 mm wide with strong, dark dorsal spines. The marginal wax tubes produce a compact, short, cottony fringe that occurs on the pupal margin. The pupal stage of orange spiny whitefly is very similar to another exotic whitefly that is currently established in Florida, Alerocanthus woglumi Ashby or citrus blackfly. The main field characteristic difference between orange spiny whitefly and citrus blackfly is that the white wax fringe that surrounds their pupal case margins is generally twice as large for the orange spiny whitefly.

Credit: Division of Plant Industry

Credit: Division of Plant Industry
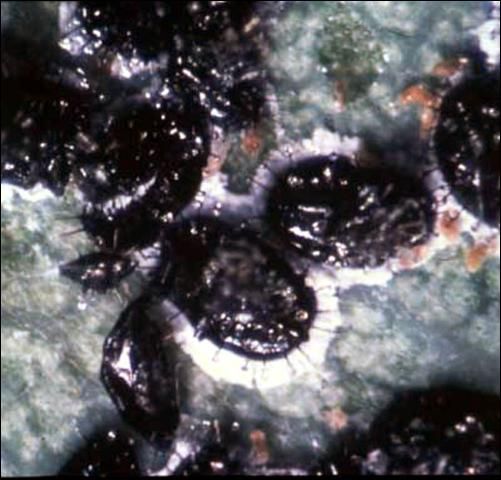
Credit: Division of Plant Industry
Adult: The adult orange spiny whitefly cannot be distinguished from citrus blackfly -- both are a slate blue color. More information on citrus blackfly in Florida is available in Nguygen and Hamon (1998).

Credit: Division of Plant Industry
Economic Importance
Citrus fruit quality and production could be adversely affected if this pest were introduced into Florida. It causes a general weakening of the infested trees due to sap loss and development of sooty mold. The leaves, fruit, and branches of infested trees are usually covered with sooty mold. Orange spiny whitefly is spread from one place to another through movement of nursery stocks and infested fruits. Orange spiny whitefly is also a potential threat to various ornamental plant hosts in Florida.
Management
Biological control: Orange spiny whitefly outbreaks in other countries have been successfully brought under control through biological control. For example, the Japanese introduced parasitoids Prospaltella smithi Silv and a Cryptognatha sp. from China in 1925 and achieved satisfactory control by killing more than 74% of the population (Kuwana et al. 1927). In Guam, the use of Prospaltella smithi and Amitus hesperidium achieved 80% to 95% parasitism (Peterson 1955). However, it was noted that the biocontrol agent did not perform as well on rose and grapes, an observation reported in India.
Classical biological control of A. spiniferus brought significant reduction within eight months of release of Encarsia cf. smithi (Hymenoptera: Aphelinidae) in the commercial orchards of southern Africa with a mean parasitism rate of about 72.9% parasitism (Van den Berg et al. 1997). It was also shown to be cost-effective with a benefit ratio of 2.8 (Van den Berg 2000).
Current established biological control options that are effective against citrus blackfly in Florida include Encarsi opulenta Huang & Polaszek and Amitus hesperidum Silvestri. Possibly these existing biological controls would also control orange spiny whitefly in Florida.

Credit: Division of Plant Industry
In additional to biological controls, whiteflies in general can be controlled by sprays applied primarily for scale insects. A low concentration petroleum spray oil (0.25% to 0.5%) has been found to provide acceptable control in China on sweet orange (Citrus sinensis L.) and pummelo (Citrus grandis L.) (Rae et al. 2000). As with any control option, effects may vary with the growing region, host plant or variety, and oil formulation.
Chemical control: In general, chemical control has not been shown to be effective on orange spiny whitefly or other whiteflies in crop systems.
Selected References
APHIS. 1975. Orange spiny whitefly, Aleurocanthus spiniferus (Quaintance). F.A.O. Plant Protection Bulletin 23:26–28.
Gowdey CC, 1922. Annual Report of the Government Entomologist. Review of Applied Entomology 11:3, 1923.
Kuwana I, Ishii T. 1927. On Prospaltella smithi Silv., and Cryptognatha sp., the enemies of Aleurocanthus spiniferus Quaintance, imported from Canton, China. Review of Applied Entomology 15:463.
Nguyen R, Sailer RI, Hamon AB. 1993. Catalog of Aleyrodidae on Citrus and their Natural Enemies (Homoptera - Aleyrodidae). Florida Department of Agriculture & Consumer Services, Division of Plant Industry. Occasional papers of the Florida State Collection of Arthropods. Contribution No. 730, Bureau of Entomology.
Peterson GD. 1955. Biological control of the orange spiny whitefly. Guam Journal of Economic Entomology. 48: 681–683.
Quaintance AL, Baker AC. 1917. Contribution to our knowledge of the white flies of the subfamily Aleyrodinae (Aleyrodidae). Proceedings of the United States National Museum 51: 352–353.
Radonjic S, Hrncic S, Malumphy C. 2014. First Record of Aleurocanthus spiniferus (Quaintance) (Hemiptera: Aleyrodidae) in Montenegro. CRA-Research Centre for Agrobiology and Pedology. 97: 141–154.
Rae DG, Watson DM, Huang MD, Cen YJ, Wang BZ, Beattie GAC, Liang WG, Tan BL, Liu DG. 2000. Efficacy and phytoxicity of multiple petroleum oil sprays on sweet orange (Citrus sinensis L.) and pummelo (C. grandis L.) in Southern China. International Journal of Pest Management 46:125–140.
USDA. 1974. New United States records - orange spiny whitefly Aleurocanthus spiniferus (Quaintance) - Hawaii. Cooperative-Economic-Insect-Report 24: 30, 585.
Van den Berg MA, Greenland J. 1997. Classical biological control of Aleurocanthus spiniferus (Hem.: Aleyrodidae), on citrus in Southern Africa. Entomophaga 42:459–465.
Van den Berg MA, Hoppner G, Greenland J. 2000. An Economic Study of the Biological Control of the Spiny Blackfly, Aleurocanthus spiniferus (Hemiptera: Aleyrodidae), in a Citrus Orchard in Swaziland. Biocontrol Science and Technology 10: 27–32.