Introduction
Dragon fruit, also known as pitahaya, pitaya, and strawberry pear, is a group of vine-like, climbing cacti originating from Mexico and Central and South America. Dragon fruit has been introduced and cultivated in Vietnam for more than 120 years. Between the 1990s and 2000s, dragon fruit was introduced into other Asian countries, the Middle East, Australia, and the United States as an exotic fruit crop (Figure 1). The short time period from planting to first harvest (= 2 years), relatively low maintenance required, high nutrient value of the fruit, and increasing demand in the market have made dragon fruit a welcomed fruit crop in the United States. In particular, growers in Florida consider dragon fruit as a potential alternative fruit crop to avocado and citrus, two economically important fruit crops heavily impacted and compromised by laurel wilt and huanglongbing, respectively. In south Florida, production of dragon fruit has been steadily increasing since the 2000s, and according to a recent estimate, the total acreage has exceeded 720 acres.

Credit: Cheng-Fang Hong, UF/IFAS
Stem and Fruit Canker of Dragon Fruit
Dragon fruit has been a relatively low-maintenance crop, but several diseases have arisen with significant impact, likely due to increases in planting acreage and the consequent buildup of pathogen inoculum. Based on a recent survey, stem and fruit canker caused by the fungal pathogen Neoscytalidium dimidiatum is a prevailing disease on dragon fruit in south Florida. The pathogen has also been reported in Southeast Asia, the Middle East, North America, Ecuador, Puerto Rico (South America and Caribbean) and India, causing canker-like symptoms on cladodes (stems) and fruits. Symptoms on cladodes start with sunken chlorotic spots, which sometimes have a tiny orange center (Figure 2A). Over time, the spots turn into larger, convex, orange to reddish-brown spots, which often coalesce to form large brown lesions. In many cases, the reddish-brown spots are surrounded by yellow halos and/or dark brown, water-soaked tissue (Figure 2B-C). The pathogen produces small black fruiting bodies (pycnidia) embedded in pale yellow to gray necrotic tissues (Figure 2C). These fruiting bodies harbor massive amounts of spores (conidia) that may be dispersed onto young, healthy tissues and start new infections. The lesions eventually dry out, perforate the cladodes, and form "shot-hole" symptoms (Figure 2D).

Credit: Cheng-Fang Hong, UF/IFAS
Once the spores are splashed by external forces, such as overhead irrigation water or windblown rain, they may land on, germinate and infect susceptible young cladodes, flower calyx, and developing fruits. Initial symptoms of N. dimidiatum infection on the calyx and developing fruits are similar to the initial symptoms seen on cladodes. Whitish-yellow sunken spots often appear scattered or as clusters on one side of the fruit (Figure 3A), particularly when the fruits are under or adjacent to the diseased cladodes. In many cases, the small, yellowish spots coalesce and form larger brown, dry lesions, which sometimes crack the fruit's peel (Figure 3B–D). In south Florida, Neoscytalidium infection is limited to the fruit's peel and usually does not invade the pulp. Diseased fruits seem to be more prone to the infection of other postharvest pathogens such as Colletotrichum. In other dragon fruit production areas, such as Israel and China, N. dimidiatum has been documented causing interior black rot of the fruit, potentially via flower tube infection. Because the grove can sustain the infection during the vegetative growing season, maintaining low levels of the pathogen inoculum is imperative to minimize the risk of stem and fruit damage.

Credit: Cheng-Fang Hong, UF/IFAS
Epidemic of Stem and Fruit Canker
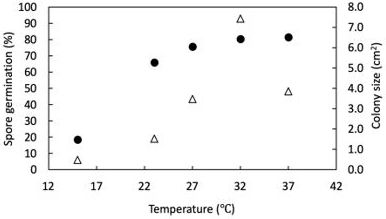
In recent studies, N. dimidiatum was reported to germinate and grow faster when the ambient temperature rises from 68°F to 95°F (20°C to 35°C) (Figure 4). Based on a model developed by a research team at the University of Florida, 50% of N. dimidiatum spores may germinate within 24 hours when the temperature is above 72°F (22°C), suggesting that the disease pressure could be high if the spores of the pathogen (inoculum source) are abundant and the temperature is rising. Hence, growers are recommended to consider implementing major pruning and sanitation programs after harvest and maintaining cleanliness in the groves before the following production season. The goals of the sanitation program are 1) to keep the pathogen population at a low level by removing and destroying diseased cladodes, and 2) to increase airflow and fungicide penetration within the canopy. It is crucial to maintain the groves as disease-free as possible during the winter and early spring, when young, susceptible cladodes are emerging and developing and the weather is cool and dry in south Florida. Otherwise, managing the disease becomes challenging as the temperature rises in late spring and remains high during the rainy season (May to October).
Management of Stem and Fruit Canker
Suggested preventative programs, including scouting, pruning, and sanitation during winter and spring are based on a recent study showing that the percentage of infected fruits in a grove was positively associated with disease incidence and severity on cladodes in the early season. If stem canker disease is high during the vegetative growth of the crop, a high percentage of fruit infection may be expected. In addition, N. dimidiatum kills plant tissues as it expands, causing irreversible spots or lesions on fruits after infection. The aesthetic damage on fruit peels could lead to 60%–80% or more loss of the market value, based on local growers' experience.
Fullerton et al. (2018) reported that, in Vietnam, it was possible to restore a highly diseased and unproductive dragon fruit grove to a healthy and high-yielding one, as long as a combination of strict field sanitation and effective fungicide programs was implemented. In south Florida, however, trying to restore a grove from a highly diseased and unproductive condition may be very costly, considering the extended period of the hot and humid season, the cost of labor, and limited fungicides registered for dragon fruit disease management.
Only a few conventional fungicides and some organic fungicides are currently registered for dragon fruit disease management (Table 1). The fungicides with the active ingredients azoxystrobin (FRAC group 11) and cyprodinil + fludioxonil (FRAC group 9 + FRAC group 12) are limited to a total of four applications per year, making it impossible to develop a disease management program that effectively protects dragon fruit throughout the season. Although the date for the start of different phenological stages of dragon fruit may vary slightly from year to year, the main idea of managing stem and fruit canker is the same: conducting sanitation (i.e., pruning and removing infected plant parts) during the cool and dry seasons, maintaining clean groves, and rotating fungicides among different FRAC groups during reproductive growth to better protect the flowers and fruits. More fungicide options are desperately needed, not only as additional tools for disease management, but also to prevent the development of fungicide-resistant pathogen populations by rotating the fungicides of different FRAC groups. This work is currently underway in collaboration with the USDA Minor Use Pesticide Program (Inter-regional Project No. 4). In short, a key component for managing dragon fruit diseases will be reducing the disease pressure by sanitation and applying fungicides during the appropriate time, which is always recommended for sustaining dragon fruit production in south Florida.
Concluding Remarks
In south Florida, stem and fruit canker, caused by the fungus N. dimidiatum, is a major disease of dragon fruit, causing up to 60%–80% losses of sale value due to the symptoms on the fruit. Management of this disease is challenging because of the prevailing/year-round warm, moist climate, the cost of labor, limited information on the pathogen, and the low numbers of registered chemical management options. Ongoing research is focusing on further understanding the biology of the pathogen, epidemiology of the disease, optimization of fungicide application, and efficacy evaluation of new chemical options for future registration on dragon fruit. Recommendations for management will be updated accordingly as further information is developed.
References
CDMS. 2019. "Crop Data Management Systems." Crop Data Management Systems, Inc. Las Vegas, Neveda. http://www.cdms.net/
Crane, J. H. 2018. "Tropical Fruit Production in Florida – Trials, Tribulations and Opportunities." Proc. Fla. State Hort. Soc. 131:ix-xii. https://journals.flvc.org/fshs/article/view/114701/110028
Crane, J. H., and C. F. Balerdi. 2006. Pitaya Growing in the Florida Home Landscape. HS1068/HS303, 11/2005. EDIS 2006 (21). https://doi.org/10.32473/edis-hs303-2005.
Espinoza-Lozano, L., Sumba, M., Calero, A., Jimenez, M. I., & Quito-Avila, D. 2022. First Report of Neoscytalidium dimidiatum Causing Stem Canker on Yellow Dragon Fruit (Hylocereus megalantus) in Ecuador. Plant Dis (ja). https://doi.org/10.1094/PDIS-06-22-1403-PDN
Evans, E. A., J. Huntley, J. H. Crane, and A. F. Wysocki. 2010. 2010 Cost Estimates of Establishing and Producing Pitaya (Dragon Fruit) in South Florida. FE888/FE888, 12/2010”. EDIS 2011 (1). https://doi.org/10.32473/edis-fe888-2010
Ezra, D., O. Liarzi, T. Gat, M. Hershcovich, and M. Dudai. 2013. First Report of Internal Black Rot Caused by Neoscytalidium dimidiatum on Hylocereus undatus (Pitahaya) Fruit in Israel. Plant Dis. 97: 1513. https://doi.org/10.1094/PDIS-05-13-0535-PDN
Fullerton, R. A., P. A. Sutherland, R. S. Rebstock, T. H. Nguyen, N. A. T. Nguyen, T. L. Dang, T. K. T. Ngo, and V. H. Nguyen. 2018. The Life Cycle of Dragon Fruit Canker Caused by Neoscytalidium dimidiatum and Implications for Control. In Proceedings of Dragon Fruit Regional Network Initiation Workshop. 71–80. Taipei: FFTC. http://www.fftc.agnet.org/upload/files/activities/20180713134846/Paper_Dr_Bob_Fullerton.pdf
Hong, C. F., R. Gazis, J. H. Crane, and S. Zhang. 2019. Prevalence and Epidemics of Neoscytalidium Stem and Fruit Canker on Pitahaya (Hylocereus spp.) in South Florida. Plant Dis. https://doi.org/10.1094/PDIS-10-19-2158-RE.
Ni, H.-F., C.-W. Huang, S.-L. Hsu, S.-Y. Lai, and H.-R. Yang. 2013. Pathogen Characterization and Fungicide Screening of Stem Canker of Pitaya. J. Taiwan Agricul. Res. 62: 225–234. https://www.tari.gov.tw/english/form/index-1.asp?Parser=20,15,101,82,,,3680,225,,,,18
Salunkhe, V. N., Bhagat, Y. S., Chavan, S. B., Lonkar, S. G., & Kakade, V. D. 2022. First Report of Neoscytalidium dimidiatum Causing Stem Canker of Dragon fruit (Hylocereus spp.) in India. Plant Dis. (ja). https://doi.org/10.1094/PDIS-04-22-0909-PDN
Serrato-Diaz, L. M., & Goenaga, R. 2021. First report of Neoscytalidium dimidiatum causing stem canker on dragon fruit (Hylocereus spp.) in Puerto Rico. Plant Dis. 105(9), 2728. https://doi.org/10.1094/PDIS-10-20-2265-PDN