Calcium (Ca) is the dominant cation in all soils of agronomic importance. Florida soils are naturally high in Ca because much of the parent material that formed Florida soils is composed of limestone (calcium carbonate, CaCO3). Consequently, Ca deficiency in Florida turfgrasses is extremely rare, and the probability of observing a Ca response on sod farms, golf courses, lawns, or athletic fields is very low (Hull 1997). Because Ca uptake is genetically controlled (Havlin et al. 1999), applying Ca fertilizers to artificially increase soil Ca above the level necessary for proper plant growth normally does not result in an increase in plant uptake. Regardless, many turfgrass managers purchase and apply Ca in both granular and liquid forms. The objective of this document is to explain the function of Ca in turfgrasses, to describe situations where the Ca applications would or would not be of value in turfgrass management, and to identify Ca sources.
Function of Calcium in Turfgrasses
Calcium is absorbed by turfgrass as Ca2+ (a positively-charged divalent cation) and, under normal conditions, turfgrass receives sufficient quantities of Ca2+ from the soil without the need to apply Ca fertilizers. Calcium is required for cell membranes to function properly and likely serves to bind phospholipids or membrane proteins. Most Ca in turfgrasses is found in cell vacuoles or serves as a structural component of cell walls (Kinzel 1989). Calcium activates some enzymes but inhibits others (Salisbury and Ross 1992). Translocation of Ca into phloem tissues is limited, which is why deficiency symptoms, although exceedingly rare, will appear on newer leaves/tissues first. Deficiency may appear as twisted or deformed tissue in roots, stems, or leaves where cell division occurs. Leaf blades may appear rose to brown and the lead tips and/or margins may wither (Carrow et al. 2001).
Soil Testing for Calcium
The decision to apply Ca should not be based upon soil test Ca levels. Soil testing for Ca is of little value because the extractant dissolves Ca compounds, which would not otherwise be plant available (FDEP 2010). In addition, correlations between Ca soil test values and turfgrass response to applied Ca has not been demonstrated in Florida soils. Furthermore, applying Ca to balance soil cations (base cation saturation ratio) is not supported by evidence and has been widely rejected by land grant institutions (Kopittke and Menzies 2007; Kreuser 2015; Sartain 1993). In fact, the application of Ca has been shown to provide no beneficial effect on turfgrass grown on calcareous putting greens (St John et al. 2003).
Soil pH and Bicarbonates
Management of soil acidity (pH) and bicarbonate (also called alkalinity, HCO3-) affects the decision on whether to apply a fertilizer containing Ca to Florida turfgrasses. Gypsum and limestone are both sources of Ca. The application of gypsum does not increase pH, whereas limestone increases pH. The counterion in limestone (carbonate, CO32-) increases pH, and can be applied if pH is below the target level for turfgrass (Figure 1).
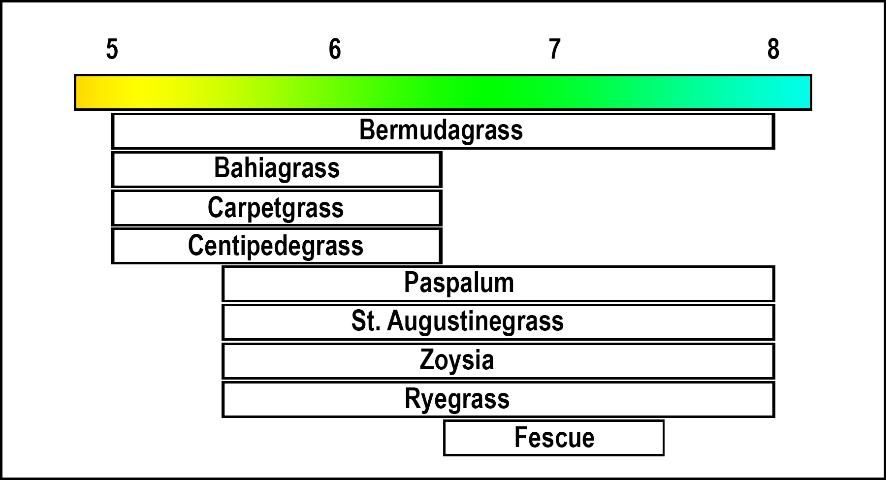
Credit: Travis Shaddox, UF/IFAS
In contrast, gypsum contains sulfate (SO42-) rather than carbonate. Gypsum does not increase pH, and can actually lower pH and neutralize bicarbonate in situations where the soil pH is initially high due to the presence of sodium (Na+) (Mylavarapu et al. 2016). The turfgrass response, if any, would likely be due to the increased biological activity that typically accompanies a reduction in pH (i.e. increased microbial activity and increased micronutrient availability). However, a turfgrass response to such a scenario has not been documented in Florida.
Bicarbonate dissolved in irrigation water is a concern because of its ability to raise soil pH (above pH 8), and its ability to bind Ca and magnesium (Mg) which can leave behind soluble Na. If water high in bicarbonates increases pH to unacceptable levels (>8.0), gypsum may be one of several ways to acidify the soil and reduce bicarbonates through the process shown in Figure 2.
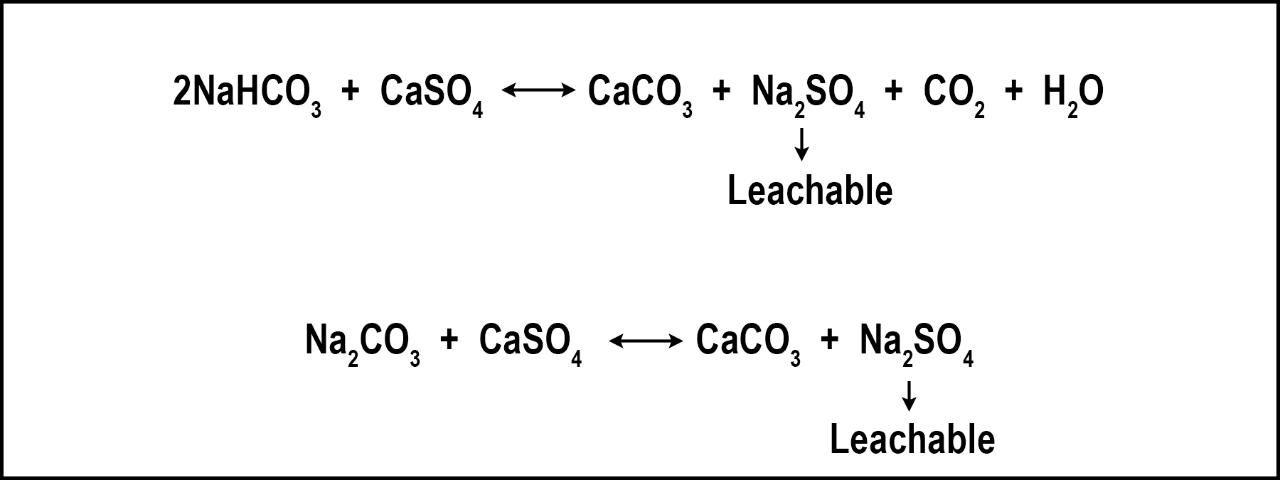
Credit: Travis Shaddox, UF/IFAS
Salt Remediation
Gypsum should not be used for salt remediation. Gypsum is a salt and, therefore, the application of gypsum does not remediate salt-affected turfgrass and, in fact, may exacerbate salt-related problems.
Calcium Sources
Irrigation Water
One of the most common Ca sources for irrigated turfgrass is the irrigation water itself. Because much of our water is sourced from the aquifer, ponds, and lakes, Ca concentration of irrigation water is commonly in excess of 40 parts per million (ppm; Adamski and Knowles Jr. 1999; Swancar 1996).
Lime
When most turf managers think of lime, they probably think of limestone. Limestone is comprised of calcium carbonate and it is one of the least expensive Ca sources available for turfgrass use. However, lime may also be purchased as burned lime, hydrated lime, or dolomitic lime. Each lime source has a different capacity to neutralize acidity and increase pH. For more information on these sources, refer to 'Fertility Considerations for Sod Production' (https://edis.ifas.ufl.edu/ss164). Lime should be used as a Ca source when soil pH falls below the suggested pH range for the turfgrass being grown (Fig 2). If soil pH is already greater than the suggested range, gypsum should be the preferred Ca source in place of lime.
Gypsum
The application of gypsum may enhance soil permeability in clay-textured saline soils. High Na levels cause dispersion of clays and often result in poor drainage. Gypsum can alleviate sodic soils by replacing Na with Ca on the soils exchange sites, which enhances soil structure (Carrow and Duncan 2012) and increases soil permeability. However, Florida soils naturally have little to no structure due to their high sand (very low clay) content. Florida's sandy soils and high rainfall also enable Na to be easily leached without the application of gypsum. Thus, the application of gypsum to turfgrasses grown on Florida soils would have little to no influence on soil structure; therefore, it should come as no surprise that the enhancement of soil structure in Florida as a result of a gypsum application has not been documented.
Calcium Chelates
Some lime and gypsum sources may contain a chelating or complexing agents such as lignosulfates, citrates, gluconates, or plant extracts. These additives are designed to increase the solubility and availability of Ca for plant uptake. When the soil pH is low (< 5.5), the use of Ca chelates on Florida soils may increase pH more effectively than non-chelated Ca sources (Adams and Pearson 1969). However, very few Ca chelates have been tested and confirmed to increase pH more efficiently than non-chelated Ca. Under normal Florida conditions, Ca is naturally solubilized from the soil and remains soluble in the soil for extended periods. Therefore, fertilizer additives intended to increase Ca solubility are normally unnecessary.
Calcium Nitrate
Calcium nitrate has a guaranteed nitrogen (N) and Ca analysis of 15% and 19%, respectively. Calcium nitrate is normally applied based upon the N component and, consequently, any turfgrass response is likely due to N. However, if the soil pH is below a suggested turfgrass range (Figure 1), calcium nitrate will increase pH and supply N in a single application.
Summary
Under normal Florida conditions, Ca should be used when pH increases are necessary, when soil bicarbonate levels result in unacceptable turfgrass, or when soil Na levels are resulting in unacceptable turfgrass. Applications of Ca for other purposes will not harm the turf or the environment, but will increase maintenance costs and are of little to no value for Florida turfgrasses.
References
Adams, F. and R.W. Pearson. 1969. "Neutralizing soil acidity under bermudagrass sod." Soil Sci. Soc. Am. Pro. 33: 737‒742.
Adamski, J.C. and L. Knowles Jr. 1999. "Ground-water quality of the surficial aquifer system and the upper floridan aquifer, Ocala National Forest and Lake county, Florida." Accessed Feb. 24, 2017. https://fl.water.usgs.gov/PDF_files/wri01_4008_adamski.pdf
Carrow, R.N. and R.R. Duncan. 2012. Best management practices for saline and sodic turfgrass soils: assessment and reclamation. Boca Raton: CRC Press.
Carrow, R.N., D.V. Waddington and P.E. Rieke. 2001. Turfgrass soil fertility and chemical problems: assessment and management. Chelsea, Michigan: Ann Arbor Press.
Florida Department of Environmental Protection. 2010. "Florida friendly best management practices for protection of water resources by the green industries." Tallahassee, FL: Florida Department of Environmental Protection.
Havlin, J.L., J.D. Beaton, S.L. Tisdale and W.L. Nelson. 1999. Sulfur, calcium, and magnesium. Soil fertility and fertilizers: an introduction to nutrient management. Upper Saddle River, New Jersey: Prentice Hall.
Hull, R. 1997. "Calcium usage by turfgrasses: the nutrient forgotten by turf managers." Turfgrass Trends 6: 6‒13.
Kinzel, H. 1989. "Calcium in the vacuoles and cell walls of plant tissue." Flora 182: 99‒125.
Kopittke, P.M. and N.W. Menzies. 2007. "A review of the use of the basic cation saturation ratio and the "ideal" soil." Soil Sci. Soc. Am. J. 71: 259‒265. doi:10.2136/sssaj2006.0186.
Kreuser, W.C. 2015. "Simplifying soil test interpretations for turf professionals." Accessed April 2025. https://turf.unl.edu/sites/unl.edu.ianr.agronomy-horticulture.turf/files/media/file/Simplifying-Soil-Test-Interpretations-g2265.pdf
Mylavarapu, R., G. Hochmuth, C. Mackowiak, A. Wright and M. Silveira. 2016. Lowering soil pH to optimize nutrient management and crop production. SL437. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://edis.ifas.ufl.edu/ss651
Salisbury, F.B. and C.W. Ross. 1992. Plant physiology. 4th ed. Belmont, California: Wadsworth Pub. Co.
Sartain, J.B. 1993. "Interrelationships among turfgrasses, clipping recycling, thatch, and applied calcium, magnesium, and potassium." Agron. J. 85: 40‒43.
St John, R.A., N.E. Christians and H.G. Taber. 2003. "Supplemental calcium applications to creeping bentgras established on calcareous sand." Crop Sci. 43: 967‒972.
Swancar, A. 1996. "Water quality, pesticide occurrence, and effects of irrigation with reclaimed water at golf courses in Florida." Accessed Feb. 24, 2017. https://fl.water.usgs.gov/PDF_files/wri95_4250_swancar.pdf