Family
Rhizophoraceae is the family of flowering trees and shrubs distributed along the intertidal zone within subtropical and tropical regions.
Genus
The genus name, Rhizophora, is the combination of the Greek terms rhiza, as in root, and phoros, as in bearing (Austin 2004). The genus is referencing the aerial roots of the red mangrove.
Specific epithet
The species name, mangle, translates to thicket in Spanish.
Common Name: Red Mangrove
The origins of the term "mangrove" are disputed among both scholars and laypersons. Some assign the origin to the Malaysian term for the trees: mangi-mangi. Others believe that it originates from the Spanish term mangle or "thicket," first used by Gonzalo Fernández de Oviedo y Valdés in 1535 A.D. to describe the tangled thicket of branches and roots mangroves form as they grow along shorelines. The “red” in red mangrove is said to come from the bright red color of the wood under the bark and the reddish color of the prop roots.
Description
Red mangrove is a salt-tolerant, small-to-medium sized evergreen tree restricted to coastal areas along the southern portions of North America, the Caribbean as well as Central America and tropical West Africa (Figure 1). In Florida, it is found as far north as Levy County on the Gulf coast and St. Johns County on the Atlantic coast as they are sensitive to freezing conditions. Monospecific forests of red mangroves often form on coastal islands, shores, lagoons, creeks, and rivers where the water is saline or brackish. Farther inland, in freshwater areas, red mangroves will grow alongside other mangrove species creating a mixed mangrove forest of red, black mangrove (Avicennia germinans) and white mangrove (Laguncularia racemosa).
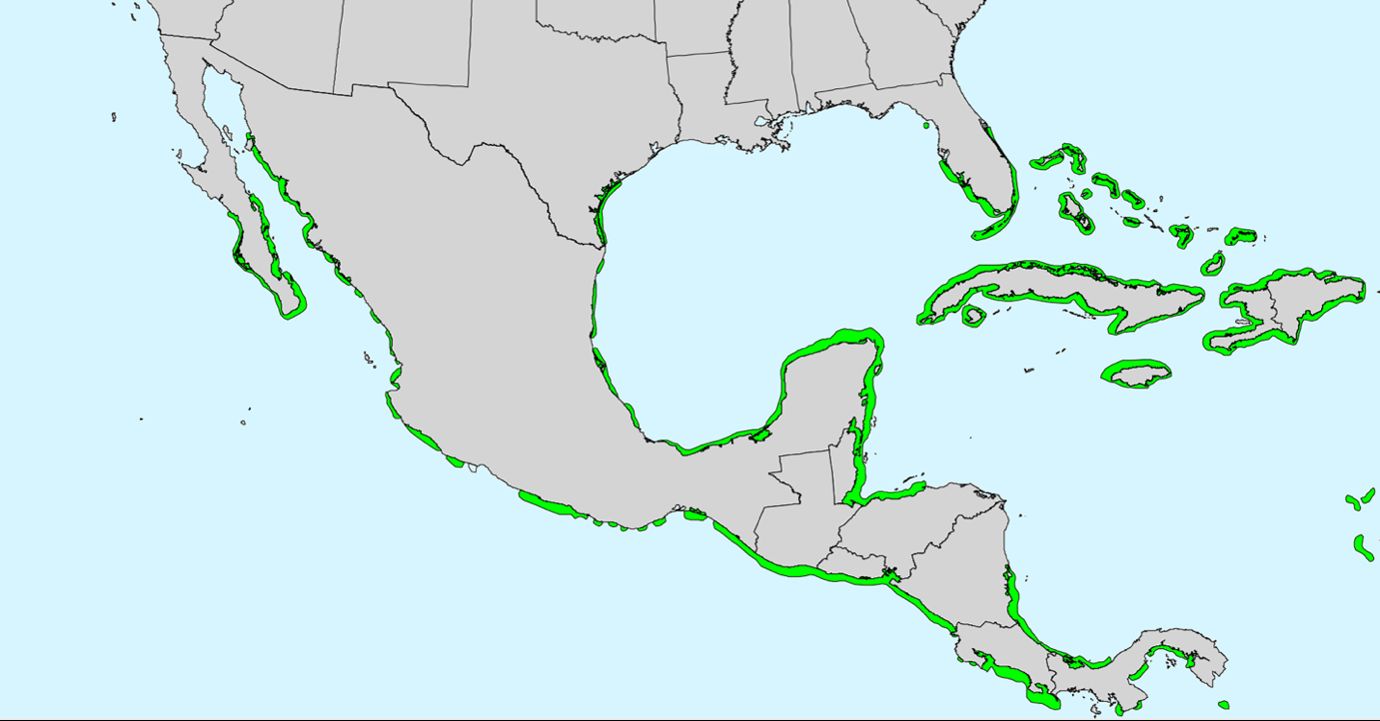
Credit: https://dendro.cnre.vt.edu/dendrology/syllabus/factsheet.cfm?ID=394
In Florida, heights of the red mangrove can reach 60 ft. In highly productive parts of its range with infrequent storms, red mangroves can reach heights above 130 ft (Allen 2002). Red mangrove leaves are medium to dark green while paler green on the underside, simple, opposite, elliptic to oblong in shape, 1.5-6 in long, and 0.5-2 in broad (Godfrey 1988). Leaves are thick and leathery with entire margins and a short petiole (Figure 2). Mature bark on the red mangrove is gray, irregularly furrowed, and thick while smooth on smaller trunks (Figure 3). The inner bark, roots, and small twigs have a reddish-to-pinkish tone that is especially noticeable when wet (Figure 4). The most recognizable characteristics of the red mangrove are the adventitious prop roots, also known as stilt roots, aerial roots, and formally referred to as rhizophores. Rhizophores create a mass entanglement of exposed, arching roots that make them impenetrable to humans but great nursery and habitat for fish (Figure 4). Red mangrove flowers are small, pale yellow turning brown, and can be present year-round in tropical regions. The fruit of the red mangrove are viviparous, meaning the fruit germinate while still attached to the parent plant. The fruit is small, 1-2 in and the green bean-like propagule that emerges from it can get quite long (7-15 in) before dropping off the tree (Figure 5). The seedling can remain viable for up to a year while floating in the ocean until reaching suitable substrate. Figure 6 shows established red mangrove seedlings.

Credit: Natalia Medina Irizarry, UF/IFAS

Credit: Natalia Medina Irizarry, UF/IFAS

Credit: Natalia Medina Irizarry, UF/IFAS

Credit: undefined

Credit: Natalia Medina Irizarry, UF/IFAS
Red mangroves are commonly mistaken for other types of mangroves. However, they can be distinguished by the site in which they appear, the root structures, and leaf size. Red mangroves can be found in higher energy sites (wave action) with longer flooding longer inundation periods along the shore, compared to black and white mangroves (Figure 7). The most distinctive feature between the tree species are the roots. Red mangrove roots are large and exposed branching structures while black mangroves have peg roots (pneumatophores) and white mangroves have no visible roots. Of the three species, red mangroves tend to have the largest leaves compared to the 1-3 in long black and white mangrove leaves.
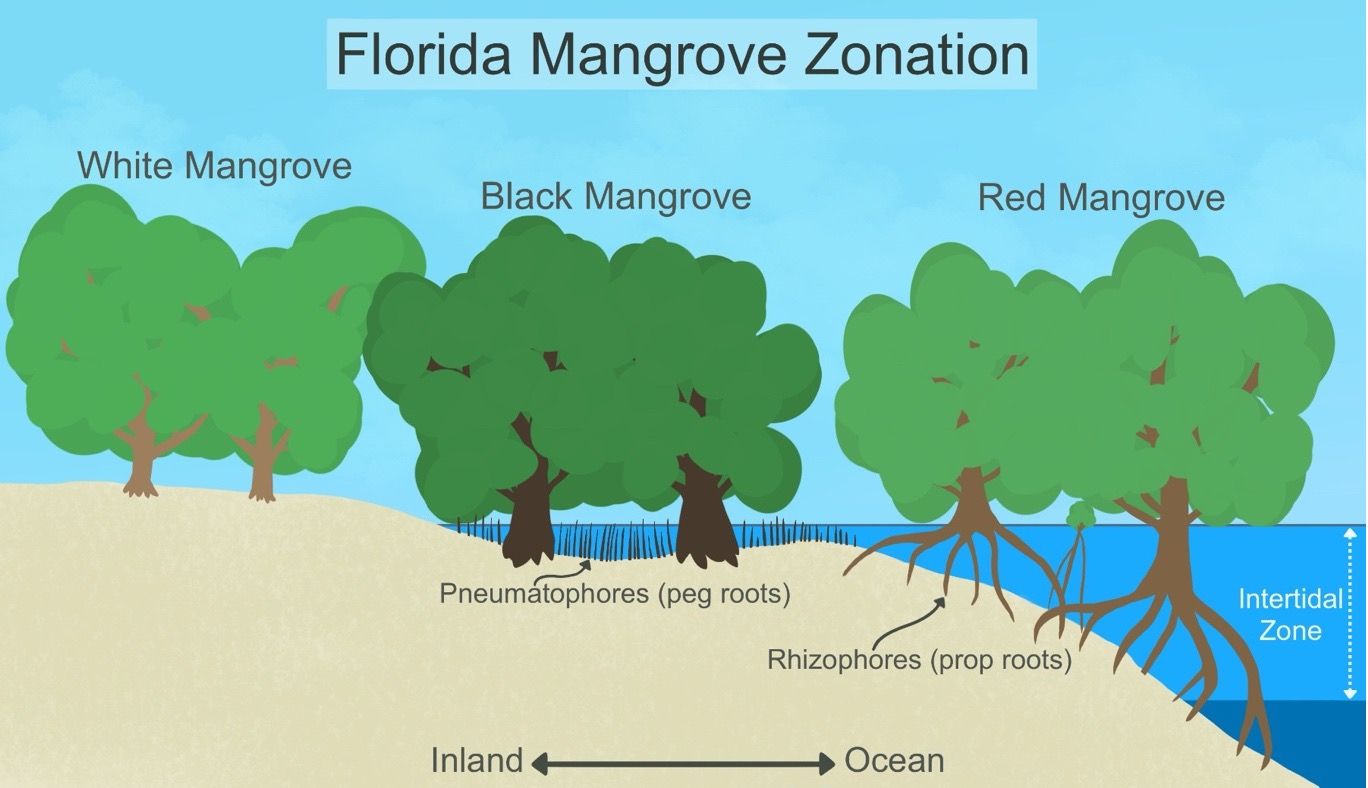
Credit: Natalia Medina Irizarry, UF/IFAS
Storm Tolerance
As a coastal species, red mangroves are important for protecting the coast from strong winds, storm surge, and erosion. In conjunction with its root system, the low and dense canopy reduces wave action significantly (McIvor et al. 2012). The dense canopy also protects the coast by acting as wind buffer. Altogether, red mangroves are the first line of defense against destructive storms and incredibly effective in reducing storm damages.
Threats
Mangroves are facing the threats of climate change, invasive species, and unsustainable management across the globe. Climate change related sea-level rise and increases in frequency of storms are expected to continue negatively impacting red mangrove productivity. Reduced productivity due to these stressors creates a ripple effect on the degradation of mangrove forests and ecosystem services provided. Invasive species with tolerance to saline environments also present a threat to red mangroves in Florida. Species such as Brazilian peppertree (Schinus terebinthifolius) and Australian pine (Casuarina spp.) have intensely misplaced Florida mangroves. Although red mangroves are not harvested in Florida for commercial products, they remain a valuable natural resource in other countries. Red mangroves in these regions may lack protection or enforcement of protection which often results in unsustainable management of the resource. There are efforts across the globe dedicated to mangrove restoration. These projects entail planting mangroves where they have previously existed to restore biodiversity and ecosystem services mangroves provide.
Applications
Commercial/Practical
In Florida, there is essentially no commercial use for mangroves. Mangroves are protected from trimming under the 1996 Mangrove and Trimming and Preservation Act. Historically, the wood of the red mangroves has been used for pilings, posts, cabinetry, construction, and wharves. The bark of the red mangrove was also used as a source of tannin which was used for treating heavy leather and textiles as well as a stain for floors and furniture (Morton 1965). Live mangrove trees have been planted since the early 1900’s along the coast to reduce coastal erosion (Morton 1981). Today, planting red mangroves is still an effective method for reducing coastal erosion. The term “living shoreline” has been coined for such projects and has created a market for red mangrove nurseries. Additional benefits to these living shorelines offer include carbon sequestration and improvements to water quality. For additional horticultural related information: Rhizophora mangle: Red Mangrove.
Wildlife
A forest of red mangroves provides nursery and rookery habitat for many fishes, birds, reptiles, and mammals. Fishes such as the young sturgeon, snook, and snapper can be found living and feeding around mangrove forests. Birds including the roseate spoonbill, white ibis, little blue heron, and various egrets rely on mangrove forests as a nesting site. Many reptiles and mammals also depend on mangrove forests for their resources and habitat. This list includes the American crocodile, Kemp’s ridley sea turtle, loggerhead sea turtle, Florida panther, Florida saltmarsh vole, Keys marsh rabbit, and manatee.
Medicinal
Historically, parts of the red mangrove plant have been used to treat several medical conditions. The bark has been reported to help treat angina, boils, fungal infections, bruises, and has been used as plaster for fractured bones (Bandaranayake 1998). Both the bark and leaves have been used historically as an antiseptic and to treat tuberculosis, diarrhea, dysentery, elephantiasis, malaria, leprosy, and fevers (Bandaranayake 1998).
References
Allen, J. A. 2002. “Rhizophora mangle L.” in Tropical Tree Seed Manual: Part II, Species Descriptions. Agricultural Handbook 712. Edited by J. Vozzo, 690–692. Washington, DC: US Department of Agriculture.
Austin, D. F. 2004. Florida Ethnobotany. Boca Raton: CRC Press. https://doi.org/10.1201/9780203491881
Bandaranayake, W. M. 1998. “Traditional and Medicinal Uses of Mangroves.” Mangroves and Salt Marshes 2 (3): 133–148. https://doi.org/10.1023/A:1009988607044
Godfrey, R. K. 1988. Trees, Shrubs, and Woody Vines of Northern Florida and Adjacent Georgia and Alabama. Athens: University of Georgia Press.
McIvor, A. L., I. Möller, T. Spencer, and M. Spalding. 2012. “Reduction of Wind and Swell Waves by Mangroves.” Natural Coastal Protection Series: Report 1. Cambridge Coastal Research Unit Working Paper 40. ISSN 2050-7941.
Morton, J. F. 1965. “Can the red mangrove provide food, feed and fertilizer?” Economic Botany 19 (2): 113–123. https://doi.org/10.1007/BF02862823
Morton, J.F. 1981. Atlas of Medicinal Plants of Middle America: Bahamas to Yucatan. Springfield, IL: Charles C. Thomas.