Within the Annonaceae, sugar apple (Annona squamosa) and atemoya (A. cherimola x A. squamosa hybrid) fruit have the greatest potential for commercialization in Florida. The major constraints to high productivity of these include key pests and the lack of pollinators.
Two hundred and ninety-six species of arthropods are associated with Annona spp. in the Neotropics. The families most frequently observed are Coccidae (Homoptera), Noctuidae, Oecophoridae (Lepidoptera), and Eurytomidae (Hymenoptera). The most common species in Florida are Bephratelloides cubensis, (Hymenoptera: Eurytomidae), Cocytius antaeus (Lepidoptera: Sphingidae), and the papaya scale Philephedra tuberculosa. Larvae of the moths Gonodonta nutrix and G. unica feed on leaves, but they are heavily parasitized by braconid wasps.
Insects on Flowers and Fruits
Pollinators and Pollination
Atemoyas and sugar apples are most often pollinated by nitidulid beetles (sap beetles), which breed and feed in decaying fruits or sap flows. The beetles are attracted to the fruity, fermenting odor of Annona flowers. The activities of beetles in the flowers, including feeding and mating, result in prolonged visits from several hours to a few days while the flowers advance from the female to the male phase. The number of beetles per flower affects the likelihood of fruit set, and also the quality of the fruit in some cases. Studies provide evidence for increased fruit set as numbers of visiting beetles increase (Nagel and Peña 1989). Trials to increase fruit set in atemoya orchards by augmenting sap beetle populations have yielded mixed results. Pollination by sap beetles can be improved by using chemical lures. Peña (2002) reported increased fruit set using nitidulid-pheromone bait stations on sugar apple and atemoya in south Florida.
In Florida, flowering of atemoyas begins in April, and sugar apples in May, and continues until early August. Nine species of native and exotic nitidulids visit the flowers (Figure 1), but Carpophilus mutilatus is the most important pollinator regarding efficacy and abundance in flowers, followed by C. fumatus and Haptoncus luteolus (Nagel and Peña 1989).

The Annona Seed Borer, Bephratelloides Species
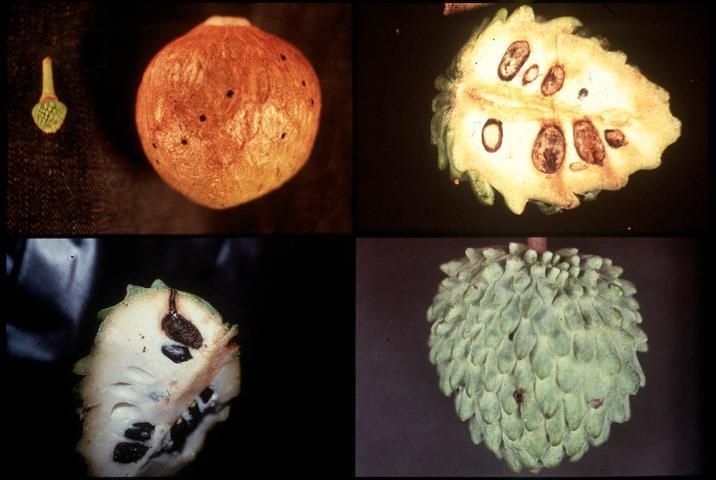
Bephratelloides spp. develop strictly in Annona seeds (Figure 2). Economic damage occurs when the adults chew their way out of the fruit, creating a 2 mm dia. tunnel that provides entry for other insects and decay organisms. Bephratelloides cubensis (Figure 3) is thelytokous, reproducing without males. It has approximately 4–5 generations per year. The egg stage lasts 12 to 14 days, the larval stage 6–8 weeks, the pupal stage 12–18 days, and the adult rarely lives beyond 15 days.


Bephratelloides cubensis prefers to oviposit in fruits ranging from 1.5–5.5 cm (0.6–2 inches) in dia., which corresponds to 3–7 week-old fruit after bloom (Figure 4). Although fruits larger than 5.5 cm dia. (2 inches) are probed, when B. cubensis populations are high, most of these attacks do not result in infestation. Preferred fruit sizes presumably correspond with seeds that have not yet hardened and are easy to penetrate with the ovipositor, while the seeds of older fruits are probably too hard to penetrate. Larger fruits may be less preferred because the distance from the fruit surface to the seed may exceed the length of the ovipositor. The probes in young sugar apple and atemoya fruits look like dark pinpricks surrounded by a round whitish patch and are visible for about two weeks; in older fruits the whitish patch does not appear and the probe marks are permanent and often ooze sap.
Oviposition activity by B. cubensis begins at about 900 h (9:00 a.m.) and continues throughout the daylight hours with peaks in activity around 1200–1300 h (12:00 p.m.–1:00 p.m.). The wasps spend the night on the underside of leaves on their host trees and move to the upper surface at sunrise. Flying individuals can be observed soon afterward and throughout the day.
Monitoring and Sampling
In Florida, custard apple (A. reticulata) sets fruit in September–November, and fruits remain on the trees as late as May. Atemoyas set fruit from April–August and sugar apples from May–August. Bephratelloides cubensis populations in Florida overwinter mainly in custard apple and then move to atemoyas that begin to set fruit during April. After a developmental time of 9 weeks, the adults emerge from the early atemoyas and infest younger atemoya and also sugar apple fruits. Second and third peaks of adult emergence and infestation occur in atemoya and sugar apples until young fruits are no longer available and the wasps switch to custard apple fruits in September. Wasp populations increase throughout the warm months because of the availability of large concentrations of atemoya trees, and are reduced in winter because custard apple is grown only sporadically as a dooryard tree in Florida. Low infestations in sugar apples may be due to low movement from atemoya to sugar apple orchards when plentiful fruits are available. No evidence of diapause (suspended development) has been found in mummified atemoya and sugar apple fruits that overwinter on trees or on the ground.
Peaks of adult B. cubensis activity are observed between 1100–1700 h (11:00 a.m.–5:00 p.m.), and numbers are higher during the warmer part of the summer. Monitoring sould concentrate on the outer layer of the canopy at the middle third of tree height and should be conducted during hours of peak activity.
Control Tactics
Fruit bagging is considered one of the best methods to prevent infestation by B. cubensis by several researchers. However, bagging encourages the growth of mealybugs on some fruits, probably because natural enemies are excluded. Although the method is effective, a cost comparison must be made between bagging and other potential control methods to determine its economic feasibility.
Biological Control
No significant native parasitization or predation of Bephratelloides spp. has been reported. In Florida, the fungus Beauveria bassiana caused high mortality on B. cubensis adults under laboratory conditions.
Mealybugs
Some species of mealybugs (Figures 5A & B) can cause significant crop losses, although they seldom approach major pest status. Factors that may contribute to increases in mealybug populations include certain ants, which reduce the effectiveness of natural enemies and transport mealybugs from one place to another. Most parasites of these mealybugs are hymenopterans of the family Encyrtidae and the most effective predators are coccinellid beetles.

The pink hibiscus mealybug Maconellicoccus hirsutus (Green) may cause severe damage to soursop (Annona muricata) (Figure 6). It attacks many plant species and affects at least 74 plant families and about 144 genera. Some major hosts include mango, hibiscus, palms, coffee, grape, citrus, and Annona spp.

The development of this species occurs in 3-4 weeks and each female can produce approximately 500 eggs. In the summer, females may not seek shelter to lay eggs. Eggs hatch in 3-8 days, and the nymphal stage lasts 10–22 days.
Several insecticides have been tested against this mealybug. However, most researchers agree that chemical and cultural control provides relief for a short period of time, or they are ineffective control methods.
Insects and Mites on Foliage
Soft Scales
In Florida, the scales Parasaissetia nigra, Saissetia coffeae, S. oleae, and Philephedra tuberculosa (Figure 7A & B) cause damage by feeding on sap. Sudden increases in populations of these insects coincide with plant stress and absence of effective natural enemies.

Armored Scales
Armored scales are uncommon and include coconut scale (Aspidiotus destructor Signoret), white peach scale (Pseudaulacaspis pentagona [Targioni-Tozetti]), Florida red scale (Chrysomphalus aonidum [L].), dictyospermum scale (Chrysomphalus dictyospermi [Morgan]), and mining scale (Howardia biclavis [Comstock]).
Lepidopterous Leaf Feeders
In Florida, larvae of the fruit piercing moths, Gonodonta nutrix (Figure 8) and G. unica, feed on tender leaves. They do not cause significant damage because they are heavily parasitized by a braconid wasp (Figure 9).


Mites
Brevipalpus spp. can affect the epidermal surface of unripe soursop fruit. The fruit becomes bronze colored with darker epidermal cracking and lighter striations, resembling rust. The eriophyid Aceria annonae infests leaves of soursop and pond apple (Figure 10). There are no records of its economic damage or control.

Aphids
The cotton aphid, Aphis gossypii Glover and the black citrus aphid, Toxoptera aurantii (Boyer de Fosncolombe), may infest young leaf shoots (Figure 11).

Control, Precautions, and Restrictions
Annonas are subject to attack by several insects. However, only a few have populations at levels high enough to be considered pests. Below is a website that contains control measures to use on an as-needed basis. However, the user must read the pesticide label carefully and adhere to regulations. The local UF/IFAS Extension office may be able to provide more information on pest control materials.
References
Habeck DH, Nagel J, Peña JE. 1989. Colopterus posticus (Erichson), a Nitidulid Beetle New to the United-States. Florida Entomologist 72(1):89–90.
Nadel H. 1990. Beetle Pollination of Sugar Apples and Atemoyas or: Why a Rotten Fruit is not all bad. Tropical Fruit News. pp. 4–5.
Nadel H, Peña JE. 1991. Seasonal Oviposition and Emergence Activity of Bephratelloides cubensis (Hymenoptera: Eurytomidae), a Pest of Annona Species in Florida. Environmental Entomology 20(4):1053–1057.
Nadel H, Peña JE. 1994. Identity, Behavior, and Efficacy of Nitidulid Beetles (Coleoptera: Nitidulidae) Pollinating Commercial Annona Species in Florida. Environmental Entomology 23(4):878–886.
Nagel J, Peña JE, Habeck D. 1989. Insect Pollination of Atemoya in Florida. Florida Entomologist 72(1):207–211.
Peña JE. 2013. Potential Invasive Pests of Agricultural Crops. Wallingford, Oxfordshire: CABI. 440 p.
Peña JE. 2002. Effect of Pheromone Bait Stations in Improving Fruit Set of Annona spp., by Sap Beetles in Florida. International Society for Horticultural Science: Acta Horticulturae 575: 509–517.
Peña JE, Baranowski RM, Litz RE. 1987. Life-History, Behavior and Natural Enemies of Philephedra tuberculosa (Homoptera: Coccidae). Florida Entomologist 70(4):423–427.
Peña JE, Bennett FD. 1995. Arthropods Associated with Annona spp. in the Neotropics. Florida Entomologist 78(2):329–349.
Peña JE, Castineiras A, Bartelt R, Duncan R. 1999. Effect of pheromone bait stations for sap beetles (Coleoptera: Nitidulidae) on Annona spp. fruit set. Florida Entomologist 82(3):475–480.
Peña JE, Glenn H, Baranowski RM. 1984. Important insect pests of Annona spp. in Florida. Proc Fla State Hort. Soc. 97:337–340.