The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The imperial moth, Eacles imperialis imperialis (Drury, 1773), is one of our largest and most beautiful moths. It is also the most variable in appearance and the most widely distributed of our large eastern United States saturniid moths.

Credit: Donald W. Hall, UF/IFAS
Synonymy
Synonyms for Eacles imperialis include Phalaena imperatoria and Basilona imperialis. For complete lists of synonyms, see Heppner (2003) or Ferguson (1971). Imperial moths have also been known by other common names (e.g., great-plane tree moth [Smith 1797] and yellow emperor [Stratton-Porter 1921]).
Distribution
Members of the imperial moth complex are found from Canada to Argentina (Goldstein 2003 [2010]), Janzen et al. 2012). Both larvae and adults are highly variable in coloration. This variation has led to designation of some of the variants as subspecies or even species (Tuskes et al. 1996). DNA barcoding studies are now being conducted that should shed light on the relationships of the different populations (Janzen et al. 2012). Only the eastern United States population, Eacles imperialis imperialis (Drury), will be considered here and, for simplicity, it will be referred to as the imperial moth or Eacles imperialis.
The imperial moth is found from southern New England south to the Florida Keys and west through the southern Great Lakes region to eastern Nebraska and central Texas (Rutkowski 1971, Wagner 2005) (Figure 2). Historically, its distribution extended farther north, but it has retreated from these areas beginning in the middle of the twentieth century. It may no longer occur in Massachusetts except for Martha's Vineyard. Several reasons for its disappearance from these northern areas have been proposed—increased usage of attractive artificial light sources, wide-spread use of insecticides and introduction of parasitoids for control of the gypsy moth (Goldstein 2003 [2010]). It is more common in the southern part of its distribution.
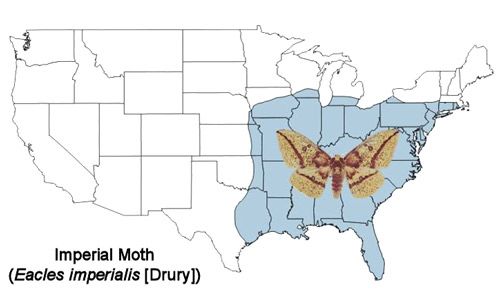
Credit: Donald W. Hall, UF/IFAS (based on map from Tuskes et al. 1996)
Description
Eggs
The eggs are flattened elliptical and 3 mm (1/8 in) in length (Packard 1905; Peterson 1965). Newly laid eggs are white (later becoming yellow), and the chorion (egg shell) is transparent. The chorion is covered with minute pits. As the embryo matures, its head capsule and segmentation are visible through the chorion. Immediately prior to hatching, the larval scoli (horns) are also visible through the chorion and in some specimens have already assumed their dark color (Eliot & Soule 1902; Soule 1902).

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS
Larvae
First instar larvae are orange with transverse black bands. They have two long, black, forked scoli tipped with thin white filaments on the second and third thoracic segments and a single one on the eighth abdominal segment. The other abdominal segments and the first thoracic segment have shorter scoli.

Credit: Donald W. Hall, UF/IFAS
Second instar larvae are dark and the scoli are shorter in relation to body length. There are also fine hairs on the body.

Credit: Donald W. Hall, UF/IFAS
The scoli of third instar larvae are even shorter in relation to body length and pigmentation of the head is darker.

Credit: Donald W. Hall, UF/IFAS
The scoli of fourth instar larvae continue the progression of shortening in relation to body length and the hairs on the body are much longer. Color variants often appear in this instar.

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS
Full-grown (fifth instar) larvae are 75–100 mm (approx. 3–5½ in.) in length (Godfrey et al. 1987). They are highly variable in color ranging from light to dark brown, burgundy, or green. The dark brown forms may be marked with burnt-orange patches dorsally and surrounding the spiracles. The area immediately around the spiracles is white in brown larvae and yellow in green larvae. The scoli of green larvae are yellow. Brown larvae are more common than green ones (Donahue 1965, Goldstein 2003 [2010]).

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS

Credit: Lyle Buss, UF/IFAS
For detailed descriptions of each instar, see Lintner (1869) and Packard (1893 & 1905).
Goldstein (2003 [2010]) reared imperial moths from Martha's Vineyard, Massachusetts and reported that both maternity and host plant affected whether larvae were green or brown with most of the green larvae appearing in larvae reared on pine. A few of the larvae he reared even switched from brown to green at one molt and then switched back to brown at the next molt.
Pupae
Pupae are dark brown with backward pointing spines on the posterior margins—presumably to assist in emergence from soil (Godfrey et al. 1987) and flanges on the anterior margins of the abdominal segments (Mosher 1914 & 1916). The abdominal segments are moveable but are prevented from telescoping by the flanges. There is a bifurcated cremaster at the tip of the abdomen (Mosher 1914 & 1916) (Figures 14 and 15). Female pupae have a notched posterior margin of the fourth abdominal segment behind the segment that is partially covered by the developing wings. Also, the female gonopores (genital openings of the adult) consist of two longitudinal slits (Figure 15 [inset]).

Credit: Donald W. Hall, UF/IFAS
In male pupae, the posterior margin of the fourth abdominal segment behind the segment that is partially covered by the developing wings is entire, and the male gonopores appear as two short tubercles (Figure 16 [inset]).

Credit: Donald W. Hall, UF/IFAS
Adults
The imperial moth has a wingspan of 80–174 mm (approx. 3–7 in.) (Beadle & Leckie 2012; Covell 2005). Females are larger than males, and males tend to be more heavily marked than females—especially in the south (Covell 2005). Moths from more northern areas tend to have fewer dark markings, but both light and dark forms are found in both northern and southern areas (Tuskes et al. 1996).

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS

Credit: Donald W. Hall, UF/IFAS
For more photographs of adults, see the North American Moth Photographers Group website (References Cited section below).
Male and female imperial moths may be differentiated by the antennae. The antennae of males are quadripectinate for the basal two-thirds and simple for the remaining length. Female antennae are simple throughout their entire length.

Credit: Donald W. Hall, UF/IFAS
Schmidt (1994) reported that males have at least some purple on the ventral aspect of the ninth abdominal segment that was lacking in all females he examined. Females are usually larger than males.
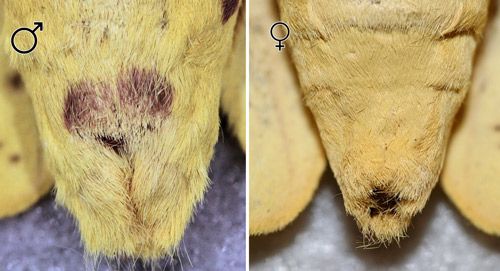
Credit: Donald W. Hall, UF/IFAS
Life Cycle
In Florida and other southern areas, a few adults emerge in spring or early summer, but most emerge in late summer. This has led various authors (Covell 2005; Ferguson 1971; Heitzman & Heitzman 1987) to report that there are two generations per year. However, Tuskes et al. (1996) believe that there is a staggered emergence with only a single generation per year.
Females of many species of moths are reported to be less common at lights than males (Williams 1939). This is true for imperial moths and other large saturniids (Worth 1979) and is probably the result of females not flying as far as males from the vicinity of host plants where they developed. The sex ratio bias against females of some species at lights has been found to be reduced when lights are in close proximity to host plants (Frank 2006). Adults emerge before sunrise and mate after midnight (Tuskes et al. 1996). Males tend to emerge several days before females. Females lay eggs singly or in small groups on both sides of leaves (Butterflies and Moths of North America web page, Tuskes et al. 1996). Adults do not feed and are short-lived.
Eggs hatch in 10 days to two weeks depending on temperature. Newly hatched larvae eat their egg shells (Stratton-Porter 1921). They swallow air and expand to a length of 7–8 mm (1/4–1/3 in) within about five minutes of hatching. At least in captivity, they wander for several days before settling down to feed on foliage. After molting and sclerotization, larvae eat their exuviae (Figure 22, inset).

Credit: Donald W. Hall, UF/IFAS
Often the presence of feeding late instar larvae of imperial moths and other large saturniids can be detected by the presence of their characteristic fecal pellets on surfaces (particularly on pavement) under the host trees (Figure 23).

Credit: Donald W. Hall, UF/IFAS
When full grown, larvae cease feeding, crawl down from the host plant, and wander along the ground searching for a suitable site to burrow into the soil for pupation. During this time they may undergo a slight color change, become shortened, and are known as prepupae (Figure 24).

Credit: Donald W. Hall, UF/IFAS
After burrowing into the soil, the prepupa forms a pupation cell in the soil. According to Tuskes et al. (1996), the surfaces of the cell are formed with larval secretions. However, Stratton-Porter (1921) was unable to detect any evidence of larval secretions and believed that the surfaces were formed by forceful packing of the soil by the prepupa. The pupa escapes the prepupal exuviae by splitting the top of the thorax (Figure 25).

Credit: Donald W. Hall, UF/IFAS
Pupae work their way to the surface prior to emergence of the adults (Stratton-Porter 1921, Tuskes 1996). Stratton-Porter (1921) reported that the pupae emerge from the soil abdominal tip first.
Hosts
Imperial moth larvae are polyphagous with many recorded hosts. However, there are probably regional differences in food preferences (Ferguson 1971). All of the larvae pictured here were raised on laurel oak, Quercus laurifolia Michaux, except the green larva (Figure 13) which was collected by Lyle Buss on live oak, Quercus virginiana Mill.
Tuskes et al. (1996) listed the following plant species as being the most commonly reported hosts for the imperial moth.
Rearing
Most females collected at lights have already mated and readily lay eggs in captivity. Females are reluctant to mate in small cages, but ones reared in captivity may be tethered outside to attract males. Worth (1980) designed a harness for tethering large moths that he used to successfully obtain mating of reared female imperial moths.
Caterpillars will feed on a variety of plant species in captivity (Tuskes et al. 1996). Villiard (1975) reported having the best success with pines.
When larvae are full grown and cease feeding, loose soil should be provided as a substrate for pupation.
Natural Enemies
Predators
The imperial moth probably has a variety of predators during its life cycle including birds, mammals, and insects. The long scoli of young instars may provide protection from some insect predators. When threatened, late instar larvae swing the thorax back and forth striking the potential predators with the thoracic scoli. Stratton-Porter (1921) observed larvae successfully repelling small birds in this manner.
Pupae probably gain protection from birds by pupating in underground cells. However, they may serve as prey for mammals that dig in the soil.
Adults remain motionless much of the time (Tuskes et al. 1996) and mimic the dead yellow leaves that are common in forests about the time the moths are emerging. A number of plant species (e.g., Vitis spp. and Cercis canadensis Linnaeus) have leaves that are similar in shape to an imperial moth in its typical resting position (Figure 26). Many of the leaves even have purple or brown patterns that enhance the mimicry of the moths.

Credit: Donald W. Hall, UF/IFAS
The low density of imperial moth populations (Tuskes et al. 1996) combined with the high variability of patterns in the moths' wings may prevent predators from forming search images.
Parasitoids
At least five species of tachinid flies (Diptera: Tachinidae) (Arnaud 1978 [p. 620]) and one species of ichneumonid wasp (Hymenoptera: Ichneumonidae) (Krombein et al. 1979 [p. 538]) have been reported from Eacles imperialis.
Tachinid parasitoids of Eacles imperialis
Belvosia bifasciata (Fabricius)
Belvosia townsendi Aldrich
Drino incompta (van der Wulp)
Lespesia frenchii (Williston)
Winthemia citheroniae (Sabrosky)
Ichneumonid parasitoid of Eacles imperialis
Conocalama quebecensis (Provancher)
Acknowledgement
The author would like to acknowledge Howard Frank for reviewing this article and offering helpful suggestions.
Selected References
Arnaud PH. 1978. A Host-Parasite Catalog of North American Tachinidae (Diptera). United States Department of Agriculture Miscellaneous Publication 1319. Washington, D.C.
Beadle D, Leckie S. 2012. Petersen Field Guide to Moths of Northeastern North America. Houghton Mifflin. New York, N. Y. 611 pp.
Butterflies and Moths of North America. http://www.butterfliesandmoths.org/species/Eacles-imperialis
Covell CV. 2005. A Field Guide to Moths of Eastern North America. Special Publication Number 12. Virginia Museum of Natural History. Martinsville, Virginia. 496 pp.
Donahue RJ. 1965. Life cycle of seclusion. Natural History 74(5): 50–51.
Eliot IM, Soule CG. 1902. Caterpillars and their Moths. The Century Company, New York. 302 pp.
Ferguson DC. 1971. The Moths of North America. Fascicle 20.2A. Bombycoidea. Saturniidae (Part). Classey. Hampton, England. pp. 32–33.
Frank KD. 2006. Effects of artificial lighting on moths. In: Rich C, Longcore T. (ed.). Ecological consequences of artificial night lighting. Island Press. Washington, D.C. pp. 305–344.
Godfrey GL, Jeffords M, Appleby JE. 1987. Saturniidae (Bombycoidea). In Stehr FW. (ed.). Immature Insects. Kendall/Hunt Publishing Company. Dubuque, Iowa. pp. 513–521.
Goldstein PZ. 2003 (2010). Life history of the Imperial Moth Eacles imerpialis (Drury) (Saturniidae: Ceratocampinae) in New England, U.S.A.: distribution, decline, and nutritional ecology of a relictual islandic population. Journal of Research on the Lepidoptera 42: 34–49.
Heitzman JR, Heitzman JE. 1987. Butterflies and moths of Missouri. Missouri Department of Conservation. Jefferson City, Missouri. 385 pp.
Heppner JB. 2003. Lepidoptera of Florida. Part 1. Introduction and Catalog. Volume 17 of Arthropods of Florida and Neighboring Land Areas. Division of Plant Industry. Florida Department of Agriculture and Consumer Services. Gainesville, Florida. 670 pp.
Janzen DH, Hallwachs W, Harvey DJ, Darrow K, Rougerie R, Hajibabaei M, Smith MA, Bertrand C, Gamboa IC, Espinoza B, Sullivan JB, Decaens T, Herbin D, Chavarria LF, Franco R, Cambronero H, Rios S, Quesada F, Pereira G, Vargas J, Guadamuz A, Espinoza R, Hernandez J, Rios L, Cantillano E, Moraga R, Moraga C, Rios P, Rios M, Calero R, Martinez D, Briceño D, Carmona M, Apu E, Aragon K, Umaña C, Perez J, Cordoba A, Umaña P, Sihezar G, Espinoza O, Cano C, Araya E, Garcia D, Ramirez H, Pereira M, Cortez J, Pereira M, Medina W, Hebert PDN. 2012. What happens to the traditional taxonomy when a well-known tropical saturniid moth fauna is DNA barcoded? Invertebrate Systematics 26(5–6): 478–505.
Krombein KV, Hurd Jr.PD, Smith DR, Burks BD. 1979. Catalog of Hymenoptera in America North of Mexico. Volume 1. Symphyta and Apocrita (Parasitica). Smithsonian Institution Press. Washington, D.C. 1198 pp.
Lintner JA. 1869. Transformations of Eacles imperialis (Drury). Twenty-fourth report of the State Museum. XII. pp. 150-154. (From the twenty-third annual report of the New York State Cabinet of Natural History.) Appendix D. Entomological Contributions.
Mosher E. 1914. The classification of the pupae of the Ceratocampidae and Hemileucidae. Annals of the Entomological Society of America 7(4): 277–300.
Mosher E. 1916. A classification of the Lepidoptera based on characters of the pupae. Bulletin of the Illinois State Laboratory of Natural History 12:17–159.
North American Moth Photographers Group (Eacles imperialis page). http://mothphotographersgroup.msstate.edu/species.php?hodges=7704
Packard AS. 1893. The life histories of certain moths of the families Ceratocampidae, Hemileucidae, etc., with notes on the armature of the larvae. (pp. 157–163). Proceedings of the American Philosophical Society 31(141): 139–192.
Packard AS. 1905. Monograph of the bombycine moths of North America, including their transformations and origin of the larval markings and armature. Part 2. Family Ceratocampidae, subfamily Ceratocampinae. (pp. 119–126). Memoirs of the National Academy of Sciences Volume 9. 394 pp. + 57 plates. Available for free download here.
Plants Database. 2014. USDA Natural Resources Conservation Service. https://plants.sc.egov.usda.gov/home
Peterson A. 1965. Some eggs of moths among the Sphingidae, Saturniidae, and Citheroniidae (Lepidoptera). Florida Entomologist 48: 213–219.
Robinson GS, Ackery PR, Kitching IJ, Beccaloni GW, Hernández LM. HOSTS - a Database of the World's Lepidopteran Hostplants. (17 September 2014)
Rutkowski F. 1971. Notes on some south Florida Lepidoptera. Journal of the Lepidopterists' Society 25(2): 137–139.
Schmidt, MD. 1994. Eacles imperialis gender markings. Ohio Lepidopterist 16(4): 85.
Smith JE. 1797. The Natural History of the Rarser Lepidopterous Insects of Georgia. Vol. 2, Printed by Bensley T for Smith JE. London. 214 pp.
Soule CG. 1902. The hatching of Eacles imperialis. Psyche 9: 299–300.
Stratton-Porter G. 1921. Moths of the Limberlost. Doubleday, Page, & Company. Garden City, New Jersey. 369 pp.
Tietz HM. 1972. An Index to the Described Life Histories, Early Stages and Hosts of the Macrolepidoptera of the Continental United States and Canada. Part 1. The Allyn Museum of Entomology. Sarasota, Florida. (Distributed by Entomological Reprint Specialists. Los Angeles, California). 536 pp.
Tuskes PM, Tuttle JP, Collins MM. 1996. The Wild Silk Moths of North America. Cornell University Press. Ithaca, NY. 250 pp.
Villiard P. 1975. Moths and How to Rear Them. Dover Publications, Inc. New York, New York. 242 pp.
Wagner DL. 2005. Caterpillars of Eastern North America. Princeton University Press. Princeton, New Jersey. 512 pp.
Williams C. 1939. An analysis of four years of light trap captures of insects in a light trap. Part 1. General survey; sex proportion; phenology; and time of flight. The Transactions of the Royal Entomological Society of London 89(6): 79–131.
Worth CB. 1980. An elegant harness for tethering large moths. Journal of the Lepidopterists' Society 34(1): 61–63.
Worth CB, Muller J. 1979. Captures of large moths by an UV light trap. Journal of the Lepidopterists' Society 33: 261–264.