The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Nylanderia fulva was previously confused with Nylanderia pubens (Synonym: Paratrechina pubens). Nylanderia fulva is native to South America where the type locality is Brazil (Gotzek et al. 2012), whereas Nylanderia pubens may have originated from the St. Vincent Island and the Lesser Antilles (Trager 1984, Forel 1893). Samples of Nylanderia pubens collected in Coral Gables and Miami, Florida, date from 1953 (Trager 1984). Klotz et al. (1995) report infestations in Boca Raton, Homestead, and Miami and state that "in 1990, hundreds of these ants were found on the second floor of a large Miami hospital." Deyrup et al. (2000) report that it "is abundant on the campus of the University of Miami, where it resembles a pale Nylanderia bourbonica, foraging on sidewalks and running up and down tree trunks." L. Davis, Jr. (2003 personal communication) has seen these ants from Everglades National Park, Fort Lauderdale, Jacksonville, and Port St. Lucie. Specimens from Sarasota (F. Santana 2003, personal communication) were also confirmed. These ants seem to have large populations where they occur and are considered a pest in Colombia (Davis 2003, personal communication).

Credit: Lyle J. Buss, University of Florida
In the West Palm Beach area, two heavily infested sites were observed about three miles west of the Intracoastal Waterway. These sites are adjacent to a utility right-of-way running to the port of Palm Beach (site of a Florida Power and Light electrical generating plant). The port may have been the point of debarkation of Nylanderia pubens into Palm Beach County. In 2002, a significant infestation of an ant was found around Houston (Harris County), Texas, that is being referred to by the media and others as the "Rasberry crazy ant" (Nylanderia nr. pubens) because it was discovered by a pest control operator named Tom Rasberry. The Texas ants were slightly larger than those from Florida, so the Texas ants were called Paratrechina sp. near pubens (MacGown and Layton 2010). However, Gotzek et al. (2012) hypothesized that pest outbreaks attributed to Nylanderia pubens in most of the southern United States may actually be Nylanderia fulva. The ants from South Florida have not been studied but are believed to be Nylanderia fulva as well.
Nylanderia pubens and Nylanderia fulva workers are morphologically similar, but the two species can be differentiated by examining the paramere of males or by genetic comparisons (Gotzek et al. 2012). The paramere of Nylanderia fulva is sparsely pilose and uneven whereas the paramere of N. pubens has dense fringe of macrochaetae.
Distribution
Nylanderia fulva has been reported from 27 counties of Florida and 27 counties of Texas. The ant infestation has also been reported from Louisiana, Mississippi, Alabama and Georgia.
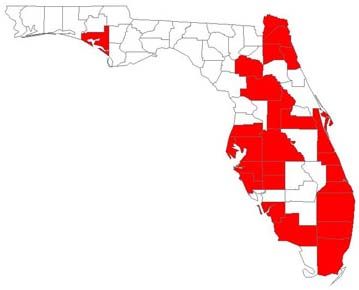
Credit: D. H. Oi, USDA-ARS
Description
Nylanderia fulva is part of a group of ants referred to as "crazy ants" due to their quick and erratic movements.
The tawny crazy ant is a medium-small, monomorphic, golden-brown to reddish-brown ant. Workers are 2.0–2.3 mm (~1/16–3/32 in) in length, males are slightly larger (2.4–2.7 mm (~3.32 in–1/8 in)), and queens are ~4.0 mm (~3/16 in) or longer (MacGown and Layton 2010). The body surface is smooth and glossy, and covered with dense pubescence (hairs). After feeding, the ant's gaster (rear portion of the abdomen) will appear to be striped due to stretching of the light-colored membrane connecting segments of the gaster. Antennae have 12 segments with no club. The antennal scape is nearly twice the width of the head. This ant has one petiolar segment and an acidopore is present at the tip of gaster in place of the stinger. Males have 13-segmented antennae (Zenner-Polania 1990) and both male and female reproductives have three ocelli on top of their head, whereas ocelli are absent in workers. Other than the size of the reproductive females, other features are similar to the workers (Zenner-Polania 1990).
Similar Ants
Nylanderia guatemalensis (Forel 1885), the Guatemalan crazy ant, has pale middle and hind coxae, shorter, stouter, and darker pilosity (Davis 2003, personal communication), and is notably smaller than Nylanderia fulva, total length 2.0 to 2.5 mm (~1/16 to 3/32 in) as compared to 2.6 to 3.0 mm (~3/32 to 1/8 in) for Nylanderia fulva (Trager 2004 personal communication). Deyrup et al. (2000) state that Nylanderia guatemalensis is a common species in Dade, Broward and Monroe Counties, but is less common farther north to Hillsborough and Indian River counties, and Deyrup (2002) adds that it is found in south Florida, north to Sarasota and Indian River counties.
Prenolepis imparis (Say 1836), the false or small honey ant, has a severely constricted hourglass-shaped alitrunk (mesosoma), and is found in north Florida, south into Orange County (Deyrup 2002).

Credit: Tim Linksvayer, Carleton College

Credit: M. B. Layton. Mississippi State University, Mississippi

Credit: J. A. MacGown. Mississippi State University, Mississippi

Credit: J. A. MacGown. Mississippi State University

Credit: J. A. MacGown. Mississippi State University
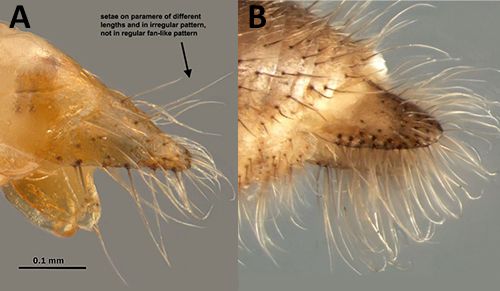
Credit: A by J. A. MacGown. Mississippi State University, Mississippi. Photograph B is modified from Gotzek et al. (2012)
Foraging and Feeding
Nylanderia fulva is omnivorous (MacGown and Layton 2010). As ambient temperatures increase (20°C), foraging trails become several ants wide (Sharma et al. 2013a). Colonies consist of a large number of ants with more than one queen (polygyne) and are polydomous (nesting in several locations). Nylanderia fulva nests occur in leaf litter, soil, rotten logs, under potted plants, and along underground electrical conduits. MacGown and Layton (2010) reported that Nylanderia fulva often occur in huge numbers and display supercolony characteristics such as yellow crazy ants, Anoplolepis gracilipes Smith (Hymenoptera: Formicidae), Argentine ants, Linepithema humile (Mayr) (Hymenoptera: Formicidae), and big headed ants, Pheidole megacephala (F.) (Hymenoptera: Formicidae), where multiple, interconnected nests could be hundreds of kilometers long (Giraud et al. 2002).
Observation of Nylanderia fulva in South Florida suggests that populations of this ant grow explosively and displace other ants within these epizootics. Infestations of these super colonies occupy large tracts such as golf courses or whole neighborhoods. Ants nest in and under rocks, logs, and other debris in overgrown lots or natural areas. These nesting areas are often hundreds of meters from foraging sites making it difficult or impossible to treat because of property ownership boundaries. As quickly as populations appear, they also suddenly disappear. Modest permanent populations are not a trademark of transient species. This "boom or bust" population pattern for Nylanderia fulva has also been observed in the West Indies (Wetterer et al. 2014)
Nylanderia fulva made shelters on the branches and trunks of magnolia trees and southern red cedar trees. In each tree, when the shelters were broken apart, tuliptree scales and juniper aphids were exposed, respectively (Sharma et al. 2013b). They are also attracted to sweet parts of plants, including nectaries and over-ripe fruit.

Credit: Eileen A. Buss, University of Florida

Credit: Shweta Sharma, University of Florida
Pest Status
Nylanderia fulva can be a nuisance. Huge number of workers in infested areas can make human activities uncomfortable and difficult (MacGown and Layton 2009). Pest control operators report infestations of sidewalks, buildings and gardens. Nylanderia fulva has caused electrical damage to phone lines, air conditioning units and computers (Meyers 2008). Nylanderia fulva has killed honey bee larvae and used the hives as their nests (Harmon 2009). The hemipteran population rises in some of the Caribbean Islands due to the tending by these ants, and the damage to fruit crops by hemipteran insects increased (Wetterer and Keularts 2008).
Introduction of Nylanderia fulva reduced the abundance of native ants in a natural reserve in Valle del Cauca, Colombia, by 74% (Aldana et al. 1995) and it has also caused reduction in diversity of ant species in Colombia (Zenner-Polania, 1990). It has also been reported by LeBrun (2014) that Nylanderia fulva is displacing red imported fire ants, Solenopsis invicta Buren, where the two populations overlap in Texas.
Management
Controlling hemipteran populations may reduce food sources utilized by Nylanderia fulva (Sharma et al. 2013b). Avoid transporting infested plant materials, mulches, pots, etc. to uninfested areas.
Granular baits over a large area could be applied by broadcasting. Given that a colony of Nylanderia fulva may contain =16,000 individuals (D. Calibeo, personal communication) contact insecticides are rendered ineffective when treated surfaces are covered by dead ants, preventing further contact with the insecticide.
Large Nylanderia fulva infestations can be difficult to control and may require a professional pest control service. More information on Nylanderia fulva control is available at: https://edis.ifas.ufl.edu/pdffiles/IN/IN88900.pdf.
Acknowledgements
We would like to thank Dr. David Oi, USDA-ARS, Gainesville, FL, for reviewing this article. We thank Mr. Joe MacGown, Dr. Blake Layton, Mississippi State University, Starkville, MS; and Dr. Eileen Buss and Mr. Lyle Buss, University of Florida, Gainesville, FL, for letting us use their pictures.
Selected References
Aldana RC, Baena ML, and Chacón de Ulloa L. 1995. Introducción de la hormiga loca (Paratrechina fulva) a la reserva natural Laguna de Sonso (Valle del Cauca, Colombia). Bol. Mus. Ent. Univ. Valle. 3: 15–28.
Calibeo D, Oi FM. 2011. Integrated pest management (IPM) of the Caribbean crazy ant, Nylanderia (= Paratrechina) pubens (Forel). ENY-2006 (IN889) ed. Gainesville: University of Florida.
Deyrup M. 2003. An updated list of Florida ants (Hymenoptera: Formicidae). Florida Entomologist 86: 43–48. (8 July 2013).
Deyrup M, Davis L, Cover S. 2000. Exotic ants of Florida. Transactions of the American Entomological Society 126: 293–326.
Fasulo TR, Kern WH, Koehler PG, Short DE. 2005. Pests In and Around the Home. UF/IFAS. SW-126.
Fasulo TR, Koehler PG. 2002. Cockroaches and Pest Ants—Computer-verified Training Tutorials. Bug Tutorials. UF/IFAS. SW-157.
Forel A. 1893. Formicides de l'Antille St. Vincent, récoltées par Mons. H. H. Smith. Transactions of the Entomological Society of London 1893: 333–418.
Giraud T, Pedersen JS, Keller L. 2002. Evolution of supercolonies: the Argentine ants of southern Europe. Proceedings of National Academy of Sciences U.S.A. 99: 6075–6079.
Gotzek D, Brady SG, Kallal RJ, Lapolla JS. 2012. The importance of using multiple approaches for identifying emerging invasive species: The case of the Rasberry crazy ant in the United States. PLoS ONE 7: e45314. doi:10.1371/journal.pone.0045314
Harmon K. 2009. Honeybees face new threat in Texas: "Crazy" ants. Online posting (https://blogs.scientificamerican.com/news-blog/honeybees-face-new-threat-in-texas-2009-08-07/), accessed 25 February 2022.
Klotz JH, Mangold JR, Vail KM, Davis LR Jr, Patterson RS. 1995. A survey of the urban pest ants (Hymenoptera: Formicidae) of peninsular Florida. Florida Entomologist 78: 109–118.
LaPolla JS, Brady SG, Shattuck SO. 2010. Phylogeny and taxonomy of the Prenolepis genus-group of ants (Hymenoptera: Formicidae). Systematic Entomology 35: 118–131.
LeBrun EG, Jones NT, Gillbert LE. 2014. Chemical warfare among invaders: A detoxification interaction facilitates an ant invasion. Science 1014–1017.
MacGown JA, Layton B. 2010. The invasive Rasberry crazy ant, Nylanderia sp. near pubens (Hymenoptera: Formicidae), reported from Mississippi. MidSouth Entomologist 3: 44–47.
Meyers JM. 2008. Identification, distribution and control of an invasive pest ant, Paratrechina sp. Hymenoptera: Formicidae, in Texas. Doctoral dissertation, Texas A&M University.
Say T. 1836. Descriptions of new species of North American Hymenoptera, and observations on some already described. Boston Journal of Natural History 1: 209–305.
Sharma S, Oi DH, Buss EA. 2013a. Honeydew-producing hemipterans in Florida associated with Nylanderia fulva (Hymenoptera: Formicidae), an invasive crazy ant. Florida Entomologist 96: 538–547.
Sharma S, Oi DH, Hodges GS, Gilman EF, Buss EA. 2013b. Associations between hemipteran honeydew producers and Nylanderia fulva (Mayr), with management implications for Florida landscapes. PhD dissertation submitted to University of Florida.
Trager JC. 1984. A revision of the genus Paratrechina (Hymenoptera: Formicidae) of the continental United States. Sociobiology 9: 51–162.
Wetterer JK, Davis O, Williamson JR. 2014. Boom and bust of the tawny Crazy Ant, Nylanderia fulva (Hymenoptera: Formicidae), on St. Croix, US Virgin Islands. Florida Entomologist 97: 1099–1103.
Wetterer JK, Keularts JLW. 2008. Population explosion of the hairy crazy ant, Paratrechina pubens (Hymenoptera: Formicidae), on St. Croix, US Virgin Islands. Florida Entomologist 91: 423–427.
Zenner-Polania I. 1990. Biological aspects of the "Hormiga Loca", Paratrechina (Nylanderia) fulva (Mayr), in Colombia, pp. 290-297. In: R. K. Vander Meer, K. Jaffe and A. Cedeno [eds.], Applied Myrmecology: A World Perspective. Westview Press, Boulder, Colorado.