The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Sabethes cyaneus is an ornate and iridescently scaled mosquito found in Neotropical forests (Santos-Mallet et al. 2013). It is a peculiar mosquito in that both males and females possess striking paddles made of elongate scales on the midlegs (second pair of legs) (Figure 1). In addition, this species has an interesting mating behavior that occurs on a substrate where the males use elaborate courtship displays to attract and mate with females (Hancock et al. 1990). Females from this species have been known to preferentially land and feed on noses when utilizing a human host. In the larval stage, this mosquito is known to be a facultative(under certain conditions) predator (Boyd & MacFadden 2020, Hancock et al. 2022).

Credit: James Gathany, Centers for Disease Control and Prevention
Synonymy
Culex cyaneus Fabricius, 1805
Sabethes locuples (Robineau-Desvoidy, 1827)
Culex cyaneus Weidmann, 1828
Culex remipes Weidmann, 1828
Culex longipes Macquart, 1838
Sabethes remipes (Giles, 1900)
Haemogogus cyaneus (Blanchard, 1905)
Sabettus remipes (Blanchard, 1905)
Sabethes remipes (Peryassu 1908)
Sabethes cyaneus (Knab, 1909)
Distribution
Sabethes cyaneus can be found in forests throughout the Neotropics, from Mexico in the north, to Argentina, where the southernmost collections of this species have been made (Figure 2) (Gaffigan et al. 2020, Santos-Mallet et al. 2013). The type locality (place where the specimen used to describe the species was first collected) is Cayenne, French Guiana (Gaffigan et al. 2020). This species occurs at elevations from sea level up to 640 meters and in almost every rainforest layer, though it appears to prefer the canopy (Galindo et al. 1951).
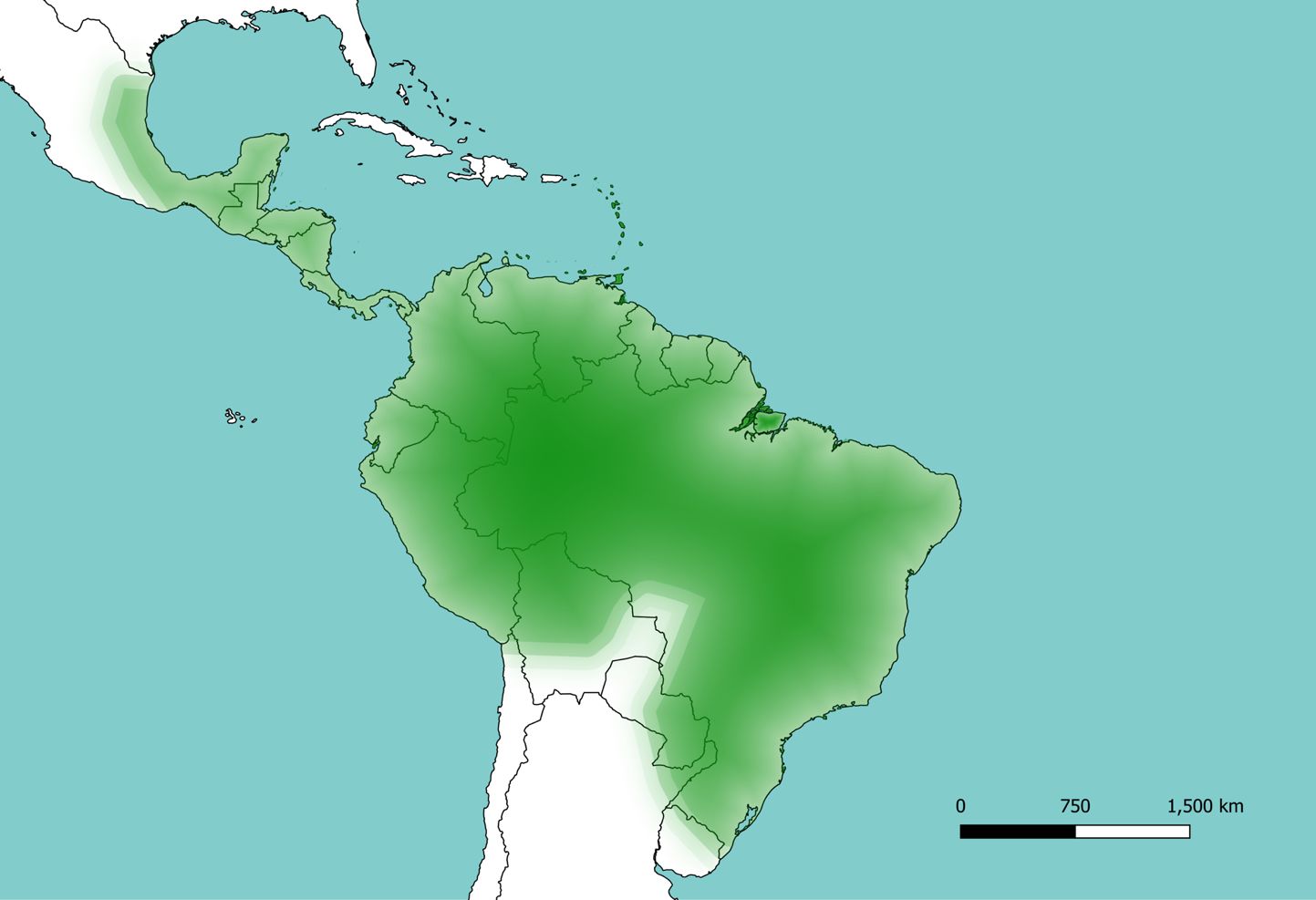
Credit: Hans Gripkey (hfgripkey@ucdavis.edu), University of California, Davis
Description
Egg
The egg of Sabethes cyaneus is biconical (shape of two cones with the flat sides meeting) from above (Figure 3) (Santos-Mallet et al. 2013). From the side, there are two distinct surfaces (Figures 3 & 4). One is smooth and convex, the other is less convex and covered with a network of projections of varying size (~1.5-5 μm) called tubercles (Santos-Mallet et al. 2013). Like most mosquito species, Sabethes cyaneus lay their eggs in water (Merritt et al. 2009), with the smooth, more convex side resting on the surface of the water (Santos-Mallet et al. 2013). The rough, tuberculate side of the egg is thought to help with flotation and orientation of the egg in water (Santos-Mallet et al. 2013).

Credit: Santos-Mallet et al. (2013)
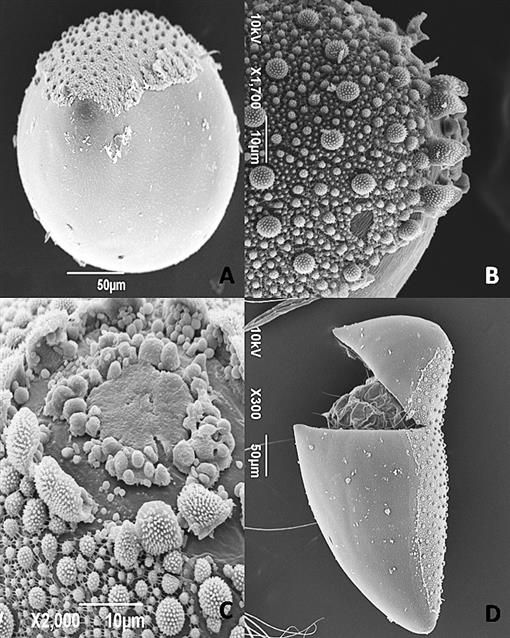
Credit: Santos-Mallet et al. (2013)
Larva
The Sabethes cyaneus larva has a typical immature mosquito body plan (Figure 5). The head is round, dorsoventrally flattened (flattened from the top and bottom), and bears short antennae, eyes, and mouthparts. Like other predatory species in the tribe Sabethini, the larvae bear toothed maxillae and mandibles (mouthpart structures) used for prey capture (Figure 6) (Hancock et al. 2022, Talaga et al. 2016). All segments making up the thorax are fused and can be differentiated by distinct groupings of hair-like setae (Merritt et al. 2009). The abdomen is composed of ten segments with the first seven being similar in form. The eighth segment bears the siphon (an abdominal structure used for breathing) (Merritt et al. 2009) which is very slender and five times as long as it is wide (Lane 1953). The ninth segment is reduced and the tenth segment, or anal segment, bears the saddle, the ventral brush (distinct pattern of hairs used for identification), and the anal papillae (similar in function to gills) which are long and rounded at the ends (Lane 1953).

Credit: Conrad Brooks, New Mexico State University

Credit: Hancock et al. (2022)
Pupa
There is no formal taxonomic description of the Sabethes cyaneus pupa (Figure 7), though the mosquito pupa in general is composed of two main body sections: the cephalothorax (fused head and thorax), and the abdomen (Merritt et al. 2009). The fusion of the head with the thorax gives the pupa its comma shape. Paddles used for swimming can be found at the end of the abdomen. Horn-like projections called trumpets are located on the dorsal side of the head and are used for breathing (Merritt et al. 2009).

Credit: Conrad Brooks, New Mexico State University
Adult (female)
Head
The Sabethes cyaneus female has a proboscis (piercing-sucking mouthparts) two-thirds as long as the forefemur (first main leg segment of the forelegs) (Lane 1953). The proboscis is dark blue, curved, and the quarter section furthest from the head is thickened (Figure 8). The palps (paired structures found directly above the proboscis) are dark blue and 2.5 times as long as the clypeus (lobe-like structure found above the palps and below the antennae). The clypeus and torus (second antennal segment) are nearly black (Lane 1953). The antennae are almost as long as the proboscis and the occiput (area behind the eyes) has bluish scales with a violet sheen (Figure 9) (Lane 1953). In comparison to females, males have more densely plumose antennae and shorter palps (Lane 1953).

Credit: Walter Reed Biosystematics Unit (wrbu.si.edu), Smithsonian Institution
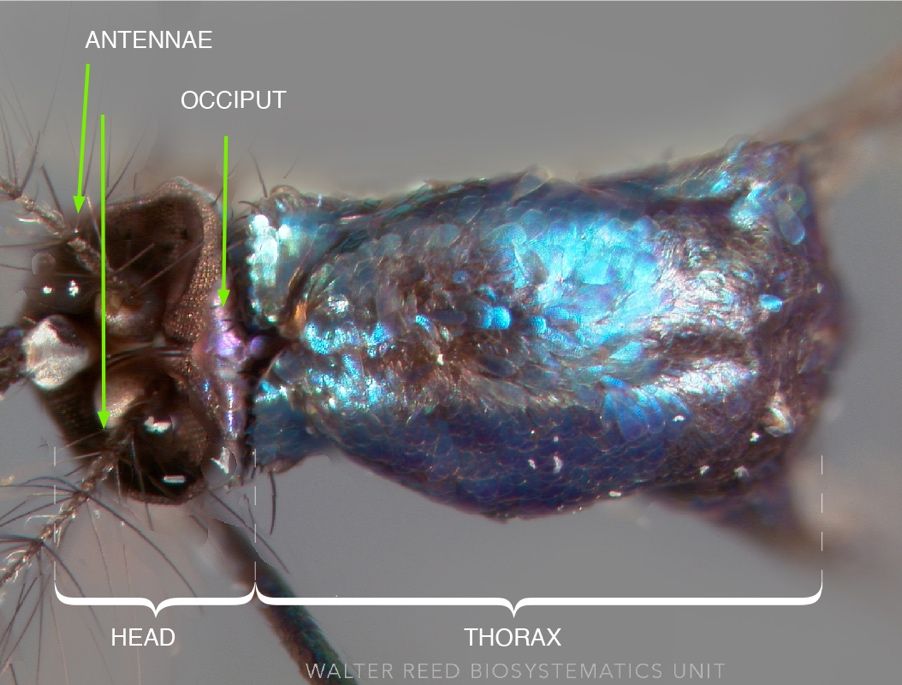
Credit: Walter Reed Biosystematics Unit (wrbu.si.edu), Smithsonian Institution
Thorax
The exoskeleton is blackish (Lane 1953). The dorsal (upper) side of the thorax has metallic blue scales and hairs near the base of the wing (Lane 1953). The pleuron (lateral section of thorax) is covered with silver scales (Figure 10) except behind the pronotum (first dorsal thorax segment) and between the fore- and midleg, where the scales are like that of the dorsal side of the thorax, but slightly lighter (Lane 1953).

Credit: Walter Reed Biosystematics Unit (wrbu.si.edu), Smithsonian Institution
Legs
The legs are dark with metallic blue or violet scales (Lane 1953). The midlegs have ornate “paddles” made of elongate scales located just past the base of the tibia (second major leg segment from the body) and on the first tarsal segment (first segment below tibia) (Figure 11). The second tarsal segment has a small paddle that is continuous with the larger leg paddle, but only on one side (Lane 1953). These paddles are thought to have a role in mate selection and are present on both males and females (South & Arnqvist 2008). The hind tibia is ciliated (has small hair-like scales) on the bottom half (Lane 1953).

Credit: South & Arnqvist (2009)
Wings
The wings have broad scales that are truncated at the ends (Lane 1953).
Abdomen
The abdomen has metallic blue scales on the dorsal side (Figure 12) and golden scales on the ventral (lower) side (Lane 1953). The male abdomen is heavily setose (covered in hair-like setae) and thickened near the end (Lane 1953).

Credit: Walter Reed Biosystematics Unit (wrbu.si.edu), Smithsonian Institution
Life Cycle/Biology
Adults are diurnal, forest canopy mosquitoes (Galindo et al. 1951, Howard et al. 1915). Like most mosquitoes, female Sabethes cyaneus must feed from the blood of another animal in order to produce eggs and do bite humans on occasion. When biting humans, they tend to bite on the nose (Figure 13) (O’Brien-Stoffa et al. 2021). However, they are believed to feed preferentially on other animals, due to the disparity between the low number of adults caught with human bait and the high frequency of Sabethes larvae observed during field surveillance of bamboo larval traps (Galindo 1951). Sabethes cyaneus females are tree hole mosquitoes that typically lay eggs about four to nine days after a bloodmeal (Hancock 1990) in water-holding tree holes and other types of phytotelmata (small pools of water held by flowers, leaves internodes, or other parts of terrestrial plants) (Santos-Mallet et al. 2013). Females can lay up to 45 eggs at a time under laboratory conditions (Galindo et al. 1951), and like other Sabethes species, female Sabethes cyaneus lay eggs by flinging each egg individually from the end of their abdomen while in flight (Figure 14) (Hancock 1990).

Credit: Jason Williams, UF/IFAS
The larval stages last about 10 days in total under laboratory conditions (Hansen 2017). Larvae are known to feed on detritus and microbes present in their aquatic phytotelma microhabitat, like most other mosquito species (Merritt et al. 2009). They have also been observed capturing and consuming other tree hole mosquito larvae as well as Culicoides biting midge larvae (Boyd & MacFadden 2020, Galindo et al. 1951). Due to this opportunistic shift in diet, this species is classified as a facultative predator, meaning they do not completely rely on insect prey for food (Boyd & MacFadden 2020). The larva has a very unusual mode of predation: it typically waits near the wall, edge, or bottom of its container habitat for prey to enter striking distance. Then, the larva opens its maxillae and mandibles (mouthparts) and arches its body, pinning it between its siphon and mouthparts (Figure 15) (Boyd & MacFadden 2020, Hancock et al. 2022). This method of predation is novel among predatory fly larvae in the infraorder Culicimorpha because of the utilization of the siphon to capture and handle prey (Hancock et al. 2022). Sabethes cyaneus larvae have been observed actively hunting for food and prefer to prey on other mosquito species, rather than cannibalize larvae of the same species (Hancock et al. 2022). Besides the way in which they catch prey, this species is also peculiar in that it does not always fully consume its prey, indicating that the attacks may also be an effort to reduce competition or to increase the concentration of micronutrients in their habitat (Hancock et al. 2022).

Credit: Vieira et al. 2020
Sabethes cyaneus, like all other mosquitoes, go through four larval instars (stages) before the pupal stage (Merritt et al. 2009). There is no description of unique Sabethes cyaneus pupal behaviors, though the mosquito pupa in general is highly mobile and responds to changes in light and to tactile stimuli (Merritt et al. 2009). This stage does not feed and typically floats just below the surface of the water (Merritt et al. 2009), where its trumpets are exposed to open air for respiration. The pupal stage lasts about four to eight days (Galindo et al. 1951, Hancock et al. 1990).

Credit: Hancock et al. 2022
Adult mosquito males do not feed on blood and are strictly nectar feeders (Yuval 2006). Sabethes cyaneus has been shown to be monandrous (females mate only once) and appears to participate in mutual mate selection (both sexes participate in mate selection) (South & Arnqvist 2008, South & Arnqvist 2011).
The most common method of mosquito mating is congregations or swarms of mosquitoes near emergence sites (Yuval 2006). The swarms are composed of males, and receptive females fly through these swarms and pair with a mate (Yuval 2006). Sabethes cyaneus mosquitoes differ in that they exhibit mutual mate choice (South & Arnqvist 2011, Yuval 2006). The males participate in elaborate courtship displays on a substrate before attempting to mate (Figure 16), which indicates that females choose their mates based off male displays (Hancock et al.1990, South & Arnqvist 2011). Interestingly, the size of the male’s paddles does not affect their mating success (South & Arnqvist 2011). However, the size of the female’s paddles is directly related to their mating success and the differences in leg paddle appearance between individuals are more pronounced in females than in males, indicating sexual selection by the male (South & Arnqvist 2009, South & Arnqvist 2011).
The mating ritual begins with a receptive female resting underneath a horizontal stick or branch that serves as a typical resting place for this species. When a receptive female is detected, the male outstretches its midlegs and approaches the female and flies in tight loop patterns nearby. The male then lands to face the female, with its wings outstretched and grips one of the female’s wings with its midtarsus (end segment of a midleg). The male then moves its free midleg in a sporadic fashion and eventually transitions into a rhythmic swinging motion of the body in conjunction with a flicking of the proboscis and raising of the mid- and hindlegs to a horizontal position (Figure 16A).
After a few repetitions of this motion, the female eventually begins to lower her abdomen to a vertical position (Figure 16B), at which point the male angles his abdomen toward the female and attempts to clasp her abdomen (Figure 16C), initiating “superficial coupling” (Figure 16D, 17). If the female does not fully lower her abdomen, the male will continue repeating the display until the female’s abdomen is in the proper position. After superficial coupling, the male releases its grip on the female wing and begins to wave its mid- and hindlegs in the form of an arc (Figure 16E). Once the male has successfully clasped to the female genitalia, it then begins to “waggle” and alternately raise and drop its mid- and hindlegs (Figure 16F). The waggling continues until either the female or male kicks off the other, in an action known as disengagement, after which the male flies off and leaves the female at rest on the substrate (Figure 16G) (Hancock et al. 1990).
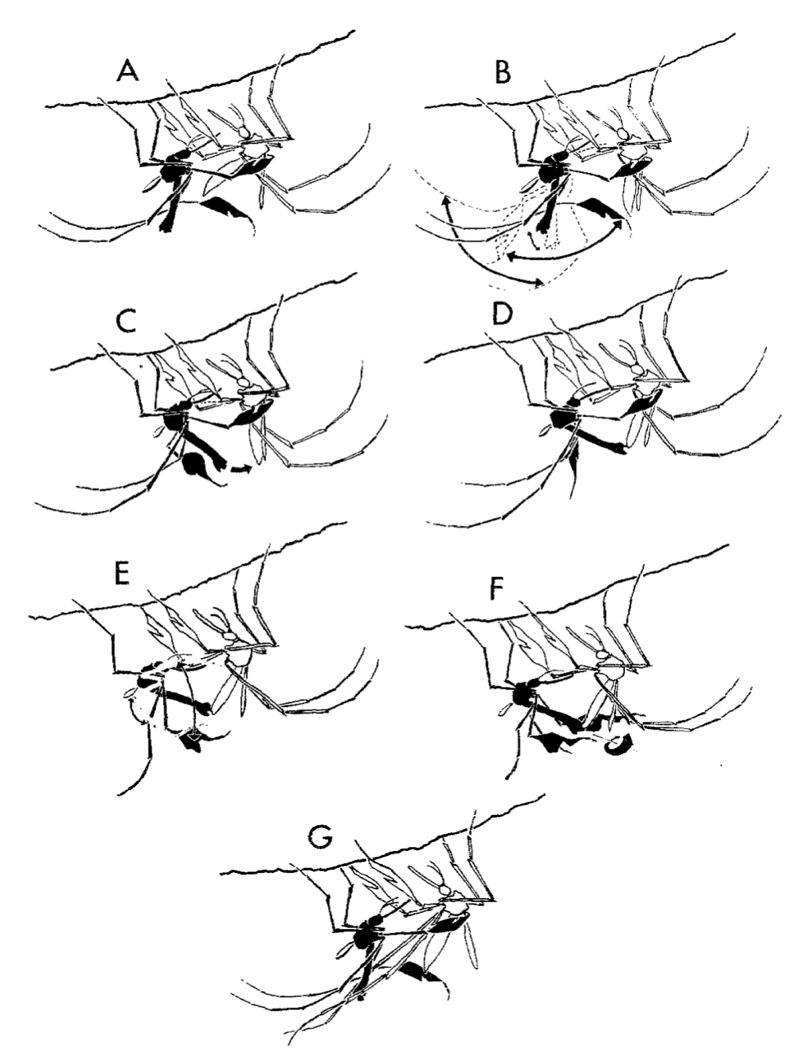
Credit: Hancock et al. (1990)

Credit: Woodbridge A. Foster, Ohio State University
Economic/medical importance
Although closely related species in the same genus are involved in transmission of yellow fever (Walter Reed Biosystematics Unit 2020), Sabethes cyaneus is not known to be a significant vector of any pathogens that cause disease in humans (Gaffigan et al. 2020). Sabethes cyaneus is a competent vector of Zika virus, though much less so than that of the most prominent vector, the yellow fever mosquito, Aedes aegypti (Linnaeus) (Karna et al. 2018). In recent years, Zika virus has been recorded in Florida (Likos et al. 2016). Sabethes cyaneus could contribute to transmission of the virus if both the virus and the mosquito become established in Florida, though the contribution may only be marginal. Saint Louis encephalitis virus has also been isolated from a sample of locally trapped Sabethes mosquitoes in Panama, including Sabethes cyaneus (Galindo et al. 1959). This, however, is not conclusive evidence that Sabethes cyaneus is a competent vector of the virus.
Management
Despite occasional feeding on humans, Sabethes cyaneus is primarily a zoophilic (prefers to feed on animals other than humans) forest mosquito and is not considered a significant nuisance or contributor to disease transmission. Therefore, it is not the primary target of vector control efforts where it occurs.
Selected References
Boyd T, MacFadden S. 2020. Larval violence in Neotropical jungle mosquitoes: investigations of interspecific predation mechanics in two sabethine mosquitoes. Conference presentation abstract. AMCA 86th Annual Meeting, Portland, Oregon, United States.
Gaffigan T, Wilkerson R, Pecor J, Stoffer J, Anderson T. 2020. Systematic Catalog of Culicidae. Walter Reed Biosystematics Unit (WRBU). http://www.mosquitocatalog.org/default.aspx
Galindo P, Carpenter SJ, Trapido H. 1951. Ecological observations on forest mosquitoes of an endemic yellow fever area in Panama. The American journal of tropical medicine and hygiene 1(1): 98-137. https://doi.org/10.4269/ajtmh.1951.s1-31.98
Galindo P, De Rodaniche E, Johnson CM. 1959. St. Louis encephalitis in Panama. The American journal of tropical medicine and hygiene 8(5): 557-560. https://doi.org/10.4269/ajtmh.1959.8.557
Hamada N., Thorp JH, Rogers DC (Eds.). 2018. Thorp and Covich's Freshwater Invertebrates: Volume 3: Keys to Neotropical Hexapoda. Academic Press. Cambridge, MA. https://doi.org/10.1016/B978-0-12-804223-6.00001-9
Hancock RG, Foster WA, Yee WL. 1990. Courtship behavior of the mosquito Sabethes cyaneus (Diptera: Culicidae). Journal of Insect Behavior 3(3): 401-416. https://doi.org/10.1007/BF01052117
Hancock RG, Boyd T, MacFadden S, Sowders A, Foster WA, Lounibos LP. 2022. Mosquitoes eating mosquitoes: how Toxorhynchites amboinensis, Psorophora ciliata, and Sabethes cyaneus (Diptera: Culicidae) capture prey. Annals of the Entomological Society of America advance article. https://doi.org/10.1093/aesa/saac017
Hansen I. 2017. Sabethes cyaneus culture video [Video]. YouTube. https://www.youtube.com/watch?v=nDaBMsbcTUA&ab_channel=ImmoHansen
Howard LO, Dyar HG, Knab F. 1915. The mosquitoes of north and central America and the West Indies (Vol. 3). Carnegie institution of Washington, Washington D.C.
Karna AK, Azar SR, Plante JA, Yun R, Vasilakis N, Weaver SC, Hanley KA. 2018. Colonized Sabethes cyaneus, a sylvatic new world mosquito species, shows a low vector competence for Zika virus relative to Aedes aegypti. Viruses 10(8): 434. https://doi.org/10.3390/v10080434
Lane J. 1953. Neotropical Culicidae (Vol. 2). Neotropical Culicidae. Volumes I & II. University of São Paulo, São Paulo, Brazil.
Likos A, Griffin I, Bingham AM, Stanek D, Fischer M, White S, Hamilton J, Eisenstein L, Atrubin D, Mulay P, Scott B, Jenkins P, Fernandez D, Rico E, Gillis L, Jean R, Cone M, Blackmore C, McAllister J, Vasquez C, Rivera L Philip, C. 2016. Local mosquito-borne transmission of Zika virus—Miami-Dade and Broward counties, Florida, June–August 2016. Morbidity and Mortality Weekly Report 65(38): 1032-1038. https://doi.org/10.15585/mmwr.mm6538e1
Merritt RW, Cummins KW, Berg MB. 2009. An Introduction to the Aquatic Insects of North America 4th edition. Kendall/Hunt Publishing Company, Dubuque, IA.
O’Brien-Stoffa C, Ward S, Hancock R. 2021. The curious case of nasophilia in Sabethes mosquitoes: Mechanisms and cues of a successful blood meal, exclusively from blood host facial features, Conference presentation abstract. AMCA 86th Annual Meeting, Portland, Oregon, United States
Santos-Mallet J, Sarmento JS, Alencar J, Müller GA, Oliveira EM, Foster WA, Marcondes CB. 2013. Scanning electron microscopy of eggs of Sabethes cyaneus. Journal of the American Mosquito Control Association 29(1): 64-68. https://doi.org/10.2987/12-6264.1
South SH., Arnqvist G. 2008. Evidence of monandry in a mosquito (Sabethes cyaneus) with elaborate ornaments in both sexes. Journal of Insect Behavior 21(6): 451-459.
South SH, Arnqvist G. 2009. Morphological variation of an ornament expressed in both sexes of the mosquito Sabethes cyaneus. Evolutionary Ecology Research 11(1): 1-21. https://doi.org/10.1007/s10905-008-9137-0
South SH, Arnqvist, G. 2011. Male, but not female, preference for an ornament expressed in both sexes of the polygynous mosquito Sabethes cyaneus. Animal Behaviour 81(3): 645-651. https://doi.org/10.1016/j.anbehav.2010.12.014
Talaga S, Leroy C, Céréghino R, Dejean A. (2016). Convergent evolution of intraguild predation in phytotelm-inhabiting mosquitoes. Evolutionary Ecology 30(6): 1133-1147. https://doi.org/10.1007/s10682-016-9862-3
Vieira G, Bersot MIL, Pereira GR, de Abreu FVS, Nascimento-Pereira AC, Neves MSAS, Rosa-Freitas MG, Motta MA, Lourenço-de-Oliveira R. 2020. High speed video documentation of the mosquito Sabethes albiprivus egg-catapulting oviposition behavior (Diptera: Culicidae). Neotropical Entomology 49(5): 662-667. https://doi.org/10.1007/s13744-020-00782-x
Walter Reed Biosystematics Unit. 2021. Sabethes chloropterus species page. Walter Reed Biosystematics Unit Website, http://wrbu.si.edu/vectorspecies/mosquitoes/chloropterus
Yuval B. 2006. Mating systems of blood-feeding flies. Annual Review of Entomology 51: 413-440. https://doi.org/10.1146/annurev.ento.51.110104.151058