The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Esser (1981) discussed land planarians in Florida. He stated almost every month of the year, specimens of gray to brown, long flat worms, with several dark stripes running down the back, were submitted to the Nematology Bureau for identification and information concerning their biology. These worms were land planarians, included in the phylum Platyhelminthes. Almost all specimens submitted belonged to the genus Bipalium.
The land planarian Bipalium kewense Moseley was first described from a greenhouse at Kew Botanical Gardens near London, England, in 1878. This species is believed to be native to Indo-China and has been commonly found in American greenhouses since 1901.
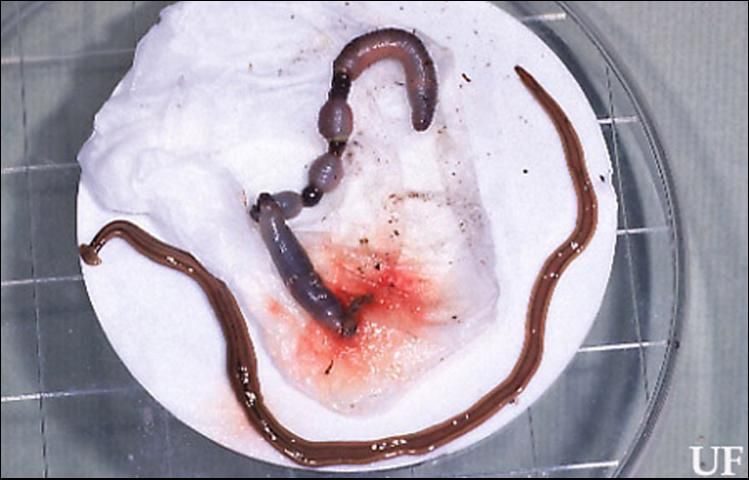
Credit: P. M. Choate, UF/IFAS

Credit: P. M. Choate, UF/IFAS
Distribution
Bipalium kewense exhibits a disjunct distribution pattern. In Australia and in the US a similar pattern of occurrence is evident. Records also include Barbados, Colombia, Costa Rica, El Salvador, Indonesia, Madagascar, Malaysia, Mauritius, Mexico, Puerto Rico, Tahiti, Taiwan, and Zimbabwe. The species appears to be dispersed with rooted plants. The natural range of Bipalium kewense extends from Vietnam to Kampuchea, possibly extending to Malaysia. Elsewhere it has been introduced by humans. Land planarians thrive in high temperature and humidity; thus, they are widely distributed in tropical and subtropical areas. They have been detected in natural habitats in Florida, Louisiana, and most recently in California, Georgia, North and South Carolina, and Texas. They have been reported in greenhouses in Alabama, California, Georgia, Illinois, Kentucky, Massachusetts, Mississippi, New Jersey, New York, North Carolina, Ohio, Oklahoma, South Carolina, and Tennessee. Land planarians had not previously been detected in mountain or desert areas. However, in October 2003, a sample of land planarians was brought to the Cochise County, Arizona, Cooperative Extension Service office for identification. Cochise County is a high desert (4000 ft+) grassland area between the Chihuahuan and Sonoran deserts (J. Gay, personal communication). This species is spreading northward along the Atlantic Coast and is able to endure freezing when in protected sites such as under boards (Hyman 1940).
It is unknown whether a September 2004 discovery in an outdoor habitat in Massachusetts indicates an established population or a temporary one that will die off during the winter (L. Novak, personal communication). In May 2005, they were reported from Nashua, New Hampshire (McClarin 2005). In April 2006, at least one specimen was reported from Novato (northern), California (Guinard 2006).
The widespread occurrence of land planarians is a result of horticultural practices and dispersion of potted plants in commerce. In tropical and subtropical areas, once planarians establish themselves in a greenhouse, they disperse to the adjacent environment.
Description
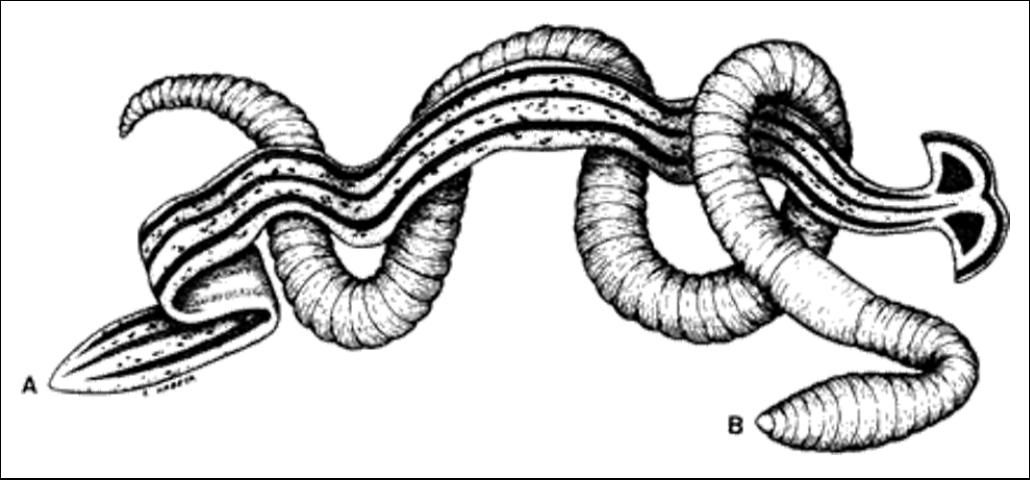
Land planarians are soft, bilaterally symmetric, acoelomate, dorsally-ventrally flattened worms, measuring 3 to 50 cm (1 1/8 in to 1 ft 7 2/3 in) long by 0.2 to 0.5 cm (~1/16 to 1/8 in) wide. They lack a respiratory and circulatory system, a skeleton, and an anus. The heads of many land planarians are expanded lunate or tapered to a blunt point. Eyespots may be present on the head. Colors of Florida species range from greenish-gray to brown with dark narrow stripes on the dorsal side. A mouth, which also serves as an anus, is present near the mid-body on the ventral surface. A protruding muscular plicate pharynx serves as a feeding organ and is attached to a three-branched intestine. The space between organs is filled with parenchyma (the functional parts of an organ). Circular and longitudinal muscles are present. A cerebral ganglion serves as a brain, innervating a ladder-shaped nervous system. Excretion of fluid wastes is accomplished with a primitive proto-nephridial system (Esser 1981).
Hyman (1943) described Dolichoplana striata Moseley: "Length up to 120mm (4 ¾ in). Form very elongated and flattened. Ground color yellowish brown with 6 longitudinal dark stripes, paired median, lateral, and marginal. Median stripes are very narrow and may be absent in young specimens. Lateral stripes are very conspicuous, black, sharply defined, beginning at level of eyes and continuing to posterior tip. Marginal stripes are less dark, diffuse, ill defined, fading away posteriorly. Creeping sole white, bordered on each side by diffuse pigmentation."
Bipalium kewense is recognized by its pale color, dorsal dark median line, dark patch in neck region, and expanded lunate head. This species may attain lengths of up to 30 cm (11 7/8 in) (Chandler 1974).
Life History
Reproduction and Development
Reproduction principally ensues by fragmentation at the posterior end. Lateral margins pinch in about 1 cm (~1/3 in) from the tail tip. Severance occurs when the posterior fragment adheres to the substrate and the parent worm pulls away. The posterior fragment is immediately motile, and within seven to 10 days a lightly pigmented head begins to form. One to two fragments are released each month.
Bright red eggs are deposited in 0.6 to 9.7 cm (1/4 to 3 7/8 in) cocoons. Within 24 hours the cocoons turn black. Planarians emerge in approximately 21 days.
Habitat
Because land planarians are photo-negative during daylight hours and require high humidity, they prefer dark, cool, moist areas under objects, such as rocks, logs, in debris, or under shrubs, and on the soil surface following heavy rains. Land planarians are also found in caves but are rare in rural sites. Movement and feeding occur at night. High humidity is essential to survival. They can survive desiccation only if water loss does not exceed 45 percent of their body weight. Land planarians are most abundant in spring and fall.
Dundee and Dundee (1963) reported Bipalium kewense as being plentiful enough in New Orleans to be used as demonstration material in zoology classes.
Locomotion
Land planarians glide smoothly on the substrate by the action of powerful, closely spaced cilia in a special medial ventral strip (creeping sole), on a thin coat of mucus secreted by glands opening into the creeping sole. Land planarians that migrate on plants or objects sometimes regain the ground by lowering themselves down by a string of mucus.
Nutrition
Land planarians devour earthworms, slugs, insect larvae, and each other. Prey are located by chemoreceptors located in a single ciliated pit under the head or in a ciliated ventral groove. Struggling prey are held to the substrate and entangled in slimy secretions from the planarian. The pharynx is protruded from the mouth and into the prey. Food is reduced to small particles prior to entering the gastrovascular cavity. The food particles are taken by epithelial cells in amoeboid fashion and formed into food vacuoles. Planarians store food in digestive epithelium and can survive many weeks by shrinking slowly in size without feeding. They can utilize their own tissues, such as reproductive tissue, for food when reserves are exhausted.
Credit: Phyllis Habeck, FDACS-DPI
Planarian Predators
Other animals rarely devour land planarians, since surface secretions appear distasteful, if not toxic. Protozoans, including flagellates, ciliates, sporozoans, and nematodes have been detected in land planarians. Because of their cannibalistic habit, land planarians may be their own worst enemy.
Economic Importance
Planarians are voracious predators of earthworms, and two species, Bipalium kewense and Dolichoplana striata Moseley, have been reported as nuisances in the southern United States in earthworm rearing beds (Hyman 1954; Dunn personal observation 1997). Two additional flatworm species, Artioposthia triangulata and Geoplana sanguinea, were accidentally imported to Ireland and England. They were reported as being capable of eradicating entire earthworm populations on farms. In greenhouses, although some collectors believe the flatworms might damage plants, they are considered harmless.
Survey and Detection
In daylight look for flatworms (sometimes with expanded heads) under rocks and logs where cool, damp areas exist. Slime trails are telltale evidence of land planarians but might also indicate slugs or snails. In worm beds, look for land planarians attached to earthworms by mucus membranes.
Disease Transmission
None reported.
Management
None reported.
Selected References
Boag B, Evans KA, Yeates GW, Johns PM, Neilson R. 1995. Assessment of the global potential of the predatory land planarian Artioposthia triangulata (Dendy) (Tricladida: Terricola) from ecoclimatic data. New Zealand Journal of Zoology 22: 311–318.
Buchsbaum R. 1948. Animals without backbones. University of Chicago Press. 405 p.
Chandler CM. 1974. The land planarian Bipalium kewense Moseley in middle Tennessee. Journal of the Tennessee Academy of Science 49: 128–129.
Cole ME. 1969. A record of the occurrence of the exotic land planarian, Bipalium kewense, in Calloway County, Kentucky. Transactions of the Kentucky Academy of Science 30: 71.
Connella JV, Stern DH. 1969. Land planarians: Sexuality and occurrence. Transactions of the American Microscopical Society, 88: 309–310.
Daly J, Matthews HM. 1982. Effect of weight and temperature upon oxygen consumption of the land planarian Bipalium kewense. Physiological Zoology 55: 148–154.
Dundee DS, Dundee HA. 1963. Observations on the land planarian Bipalium kewense Moseley in the Gulf Coast. Systematic Zoology 12: 36–37.
Edwards CA, Lofty JR. 1972. Biology of earthworms. Chapman and Hall Limited, London. 283 p.
Esser RP. 1981. Land planarians (Tricladida: Terricola). Contribution no. 227, Bureau of Nematology, Florida Department of Agricultural and Consumer Services, Division of Plant Industry, Gainesville, Fl.
Guinard D. (7 April 2006). Fw: Shovel Head Worm. (6 April 2006).
Harrel RC, Bechler DL, Ogren RE. 1994. First Texas record of Geoplana arkalabamensis (Turbellaria: Tricladida: Terricola) with a review of other land planarians reported in the state. Texas Journal of Science 46: 45–49.
Hyman LH. 1940. Native and introduced land planarians in the United States. Science 92: 105–106.
Hyman LH. 1943. Endemic and exotic land planarians in the United States with a discussion of necessary changes in the Rhynchodemidae. American Museum Novitates 1241: 1–21.
Hyman LH. 1951. The Invertebrates: Platyhelminthes and Rhynchocoela the acoelomate Bilateria, McGraw-Hill, New York, USA. 550 p.
Klots AB. 1960. A terrestrial flatworm well established outdoors in the northeastern United States. Systematic Zoology 9: 33–34.
Landsperger WJ, Peters EH, Dresden MH. 1981. Properties of a collagenolytic enzyme from Bipalium kewense. Biochimica et Biophysica Acta 661: 213–220.
MacKenzie D. 1991. Where earthworms fear to tread. New Science 10: 31–34.
McClarin J. (7 May 2005). Fw: Land planaria. (9 May 2005).
Moseley HN. 1877. Notes on the structures of several forms of land planarians, with a description of two new genera and several new species, and a list of all species at present known. Microscopy Journal 7: 273–292.
Neck RW. 1987. A predatory terrestrial flatworm, Bipalium kewense, in Texas (USA) feral populations and laboratory observations. Texas Journal of Science 39: 267–272.
Pennak RW. 1953. Fresh-water invertebrates of the United States. Ronald Press Company, New York, USA. 769 p.
Riser NW, Morse MP. 1974. Biology of the Turbellaria. McGraw-Hill, New York, USA. 530 p.
Sluys R, Joffe B, Cannon LRG. 1995. An alien flatworm in Australian waters. Memoirs of the Queensland Museum 38: 642.
Sun CN, Chew SBC, White HJ. 1979. The fine structure of smooth muscle in Bipalium kewense, its possible relation to evolution of skeletal muscle. Cytologia (Tokyo) 44: 181–190.
von Graf L. 1899. Monographie der Turbellarien II. Trichladida. In Bronn HG (ed.), Klassen und Ordnungen des Tier-Reichs, Bd. IV, Apt. pp. 1733–3369.
Walton LB. 1907. Land planarians in the United States. Science 25: 732–733.
Walton LB. 1912. The land planarians of North America with a note on a new species. Science 35: 940.
Winsor L. 1983. A revision of the cosmopolitan land planarian Bipalium kewense (Turbellaria: Tricladida: Terricola). Zoological Journal of the Linnean Society 79: 61–100.
Winsor L. 1997. The biodiversity of terrestrial flatworms (Tricladida: Terricola) in Queensland: A preliminary report. Memoirs of Museum Victoria 56: 575–579.