The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Sweetpotato Whitefly B Biotype, Bemisia tabaci (Gennadius) (Insecta: Hemiptera: Aleyrodidae)
Heather J. McAuslane and Hugh A. Smith
Distribution
Bemisia tabaci is primarily a pest of cultivated plants in tropical and warm temperate regions of the world. It is found throughout the southern United States and can overwinter outdoors as far north as South Carolina. It is found infesting greenhouses in more northern latitudes in the United States and Canada. It is widely distributed throughout the Caribbean Islands, Central and South America, and Mexico. It is present throughout tropical and subtropical regions of the world including many island groups in the Pacific. It is considered a major pest of horticultural, agronomic, and ornamental crops throughout the Mediterranean, Africa, India, and Australasia.

Credit: Yasmin Cardoza, University of Florida
Description and Life History
Infestations of whiteflies, especially in greenhouses, may occasionally be a mixture of Bemisia tabaci and Trialeurodes vaporariorum (Westwood), the greenhouse whitefly. They can be differentiated at the adult stage based on the position in which the wings are held over the body while alive, close to the body and tent-like in Bemisia, and more loosely in T. vaporariorum. Another way to differentiate adult Bemisia and T. vaporariorum is to examine the compound eyes using a microscope. The upper and lower compound eyes of T. vaporariorum are completely divided whereas they are joined by one ommatidium in Bemisia. Adult Bemisia are soft and whitish-yellow when they first emerge from their nymphal exuviae. Within a few hours, their two pairs of wings become iridescent white due to the deposition of a powdery wax. The body remains light yellow with a light dusting of wax. The body of the female measures 0.96 mm from the tip of the vertex (head) to the tip of the abdomen, while the male is somewhat smaller at 0.82 mm.
Adult whiteflies emerge through a T-shaped slit in the integument of the last nymphal instar. The remaining white, transparent shell is called the exuviae. If the exuviae have a round hole in it rather than a slit, an adult parasitoid emerged. Adults that emerge may simply fly up the same plant or over to another plant. These are called trivial flights. Some individuals, however, are primed for short-distance migration of up to several kilometers. Migrating individuals usually develop on plants that are senescing. These migrations can often be massive and can lead to severe infestation of newly planted crops.

Credit: Lyle Buss, UF/IFAS
Credit: Jane C. Medley, UF/IFAS
As far as we know, the sexes do not rely on pheromones to locate one another. Mating can occur as soon as the whiteflies have expanded and hardened their wings, usually within a few hours. Bemisia has arrhenotokous parthenogenetic reproduction where virgin females can only lay haploid eggs which give rise to males. Mated females can produce haploid and diploid (female) eggs. There is usually a pre-oviposition period of less than a day to a few days, depending on the temperature. The female lands on a plant, attracted to it by visual stimuli such as plant color, and walks or flies to the lower surface of a leaf. She tests the suitability of the plant by probing the leaf with her piercing-sucking mouthparts and ingesting a small amount of sap.
Chemoreceptors on the tip of her mouthparts and within her mouthparts sense the leaf’s chemical composition. If the host plant is deemed acceptable, she will insert her mouthpart stylets into the phloem from where she sucks plant sap. While she is feeding she may lay eggs, often in a semi-circular arrangement as she swivels her body around her feeding site. Female longevity can range from 10 to 24 days during which time she can lay between 66 and 300 eggs, depending on host plant and temperature.
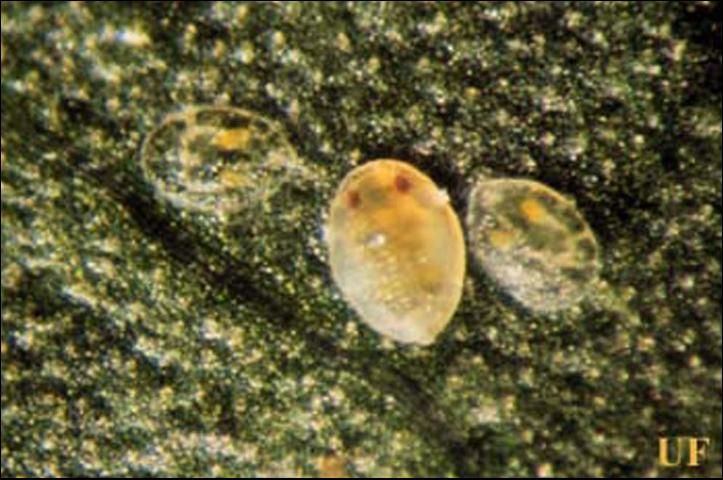
Whitefly eggs are oval in shape and somewhat tapered towards the distal end. The broader end has a short stalk, 0.024 mm, that is inserted by the ovipositing female into the leaf. The egg obtains moisture through this stalk. The egg is approximately 0.21 mm in length and 0.096 mm in width. The egg is pearly white when first laid but darkens over time. The distal end of the egg becomes dark brown just before the first nymphal instar ecloses. At 25° C, the eggs will hatch in six to seven days.

Credit: James Castner, UF/IFAS
The first nymphal instar is capable of limited movement and is called the crawler. It is oval in shape and measures approximately 0.27 mm in length and 0.14 mm in width. The dorsal surface of the crawler is convex while the ventral surface, appressed to the leaf surface, is flat. The crawler has three pairs of well-developed four-segmented legs, three-segmented antennae, and small eyes. It is whitish- green in color and has two yellow spots, the mycetomes, visible in the abdomen through the integument (skin). The mycetomes house several species of endosymbiotic bacteria that may play an important role in whitefly nutrition. The crawlers usually move only a few centimeters in search of a feeding site but can move to another leaf on the same plant. They initiate feeding on the lower surface of a leaf, also feeding in the phloem. After they have begun feeding, they will molt to the second nymphal instar, usually two to three days after eclosion from the egg.
The second, third and fourth nymphal instars are immobile with atrophied legs and antennae, and small eyes. The nymphs secrete a waxy material at the margins of their body that helps adhere them to the leaf surface. Nymphs are flattened and oval in shape, greenish-yellow in color, and range from 0.365 mm (second instar) to 0.662 mm (fourth instar) in length. The mycetomes are yellow. The second and third nymphal instars each last about two to three days.

Credit: James Castner, UF/IFAS
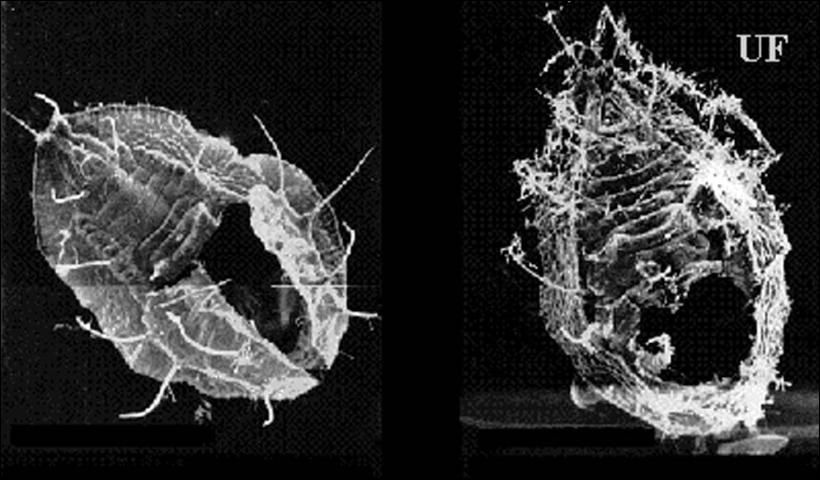
The red-eyed nymphal stage is sometimes called the “pupal stage.” There is no molt between the fourth nymphal instar and the red-eyed nymphal stage but there are morphological differences. The fourth and red-eyed nymphal stages combined last for five to six days. The stage gets its name from the prominent red eyes that are much larger than the eyes of earlier nymphal instars. The red-eyed nymphal stage is also less flattened and more convex in shape. This stage is more yellow than the fourth instar and the mycetomes are less visible. The red-eyed nymphal stages of Bemisia and T. vaporariorum can be distinguished easily in a mixed infestation. The sides of Bemisia red-eyed nymphs are slightly convex while those of T. vaporariorum are perpendicular to the leaf surface.
Hosts
Bemisia is widely polyphagous, feeding on over 500 species of plants in 74 families. Its hosts include vegetable, field, and ornamental crops. Of the important vegetable crops grown in Florida, B. tabaci is a major pest of tomato, peppers, squash, cucumber, beans, eggplant, watermelon, and cabbage. The Florida-grown field crops of potato, peanut, soybean and cotton are heavily attacked by B. tabaci. The ornamental host plants of Bemisia are too numerous to list, but include poinsettia, hibiscus, and chrysanthemum.
Credit: James Castner, UF/IFAS
Damage
Bemisia can cause economic damage to plants in several ways. Heavy infestations of adults and their progeny can cause seedling death or reduction in vigor and yield of older plants due simply to sap removal. When adult and immature whiteflies feed, they excrete honeydew, a sticky excretory waste that is composed largely of plant sugars. The honeydew can stick cotton lint together, making it more difficult to gin and therefore reducing its value. Sooty mold grows on honeydew-covered substrates, obscuring the leaf and reducing photosynthesis, and reducing fruit quality grade.
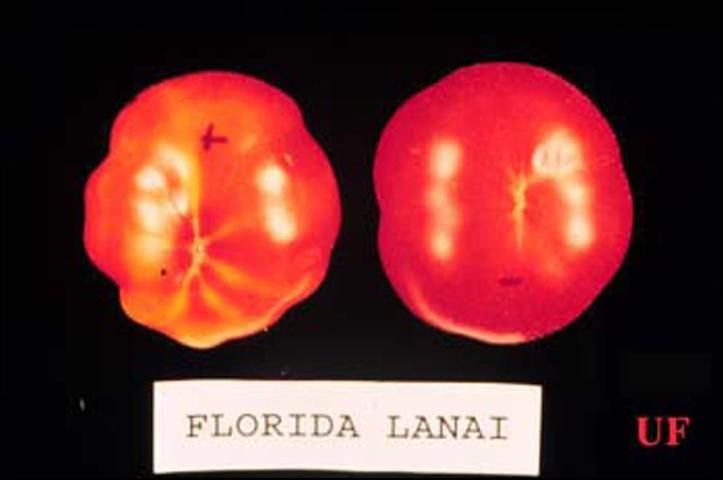
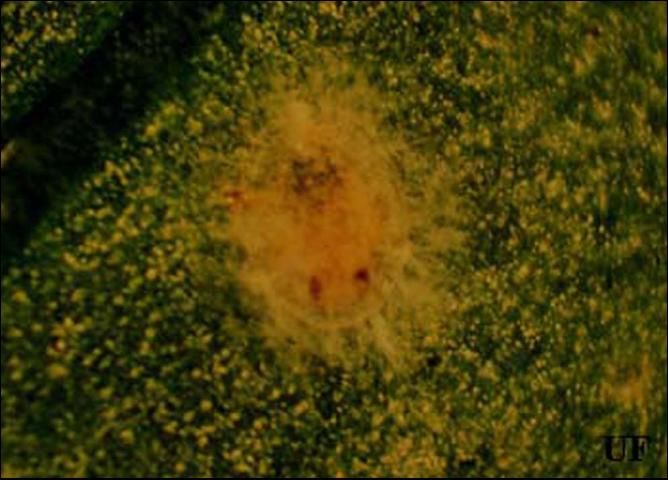
Feeding by immature Bemisia, but not adults, has been associated with several developmental physiological disorders of plants. Tomato irregular ripening was first noted in Florida tomatoes in 1987. Tomatoes that develop on plants that are heavily infested with whiteflies may incompletely develop external color, resulting in streaking. Even if fruits appear normal externally, the internal tissue may be white, hard, and unripe. It has been estimated that, in 1989, Florida tomato growers lost $25 million to tomato irregular ripening. Squash silverleaf disorder is another developmental disorder caused by feeding of immature whiteflies, also first noted in Florida in 1987. This disorder affects many Cucurbita species, including the squashes and pumpkins of C. pepo, C. moschata, and C. mixta. Feeding by immature whiteflies causes newly developing leaves, but not the leaves on which they are feeding, to take on a silvery appearance due to the separation of the upper epidermis from the underlying cell layer. The resultant air space reflects light, causing the silvery color. Fruits that develop on silvered plants may be bleached and are of lower quality grade. Other physiological disorders caused by Bemisia include lettuce leaf yellowing and stem blanching, carrot light root, pepper streak, Brassica white stem, and chlorosis of new foliage of many plants.

Credit: James Castner, UF/IFAS
Bemisia tabaci transmits several serious plant-pathogenic viruses in the United States. In Florida, our main concerns are Tomato yellow leaf curl virus (TYLCV), Bean golden mosaic virus (BGMV), Squash vein yellowing virus, Cucurbit leaf crumple virus, and Cucurbit yellow stunting disorder virus. Tomato mottle virus (TMoV), the first whitefly- transmitted virus to impact Florida agriculture in the late 1980s, is rarely seen today. Advice for home gardeners to manage TYLCV includes destroying and disposing of symptomatic tomato plants so that they cannot become a source of inoculum for healthy plants, and managing Bemisia populations with insecticides, if necessary.
Credit: Shahab Hanif-khan, UF/IFAS
Cultural Control
Several cultural controls can reduce Bemisia tabaci populations and lessen their impact on crops. One important control method is sanitation. During the growing season, virus-infected plants can be rogued out and destroyed.
After harvest, crop residue (tomato plants, in particular) should be removed to reduce the availability of virus hosts between cropping periods.

Israeli researchers have had success with the use of barriers to keep viruliferous Bemisia from invading greenhouses.
Greenhouses are screened with very fine mesh plastic screen. Ventilation must be increased however, to reduce the likelihood of infection by plant pathogens. Whitefly infestations have also been reduced with the use of UV-absorbing greenhouse plastic films. Whiteflies do not enter greenhouses or areas covered with this type of plastic as frequently as they do greenhouses covered in non-UV- absorbing material. Reflective metalized plastic mulches are used in Florida to repel whiteflies from the crop. In Costa Rica, living mulches (e.g., perennial peanut and cilantro) may reduce somewhat the spread of geminivirus within tomato fields.

Host plant resistance offers hope for management of whitefly-transmitted viruses, although no varieties of host plants have been found to be highly resistant to whiteflies themselves. Consult the Florida Vegetable Production Handbook for information on commercially available varieties of tomato that are tolerant of TYLCV (https://edis.ifas.ufl.edu/publication/cv137). Some plant factors are not preferred by whiteflies. For example, smooth-leaved varieties of cotton and soybean are less preferred by ovipositing female Bemisia than are hairy-leaved varieties. This holds true for many plant species. Glossy (less waxy) crucifers, such as broccoli and collard, are less acceptable for oviposition than are varieties with a normal wax layer.
Biological Control
In Florida, Bemisia rarely reaches outbreak population levels in natural environments or in agricultural situations where no insecticides are applied. In these situations, natural biological control keeps Bemisia below economically injurious levels. Much mortality is caused by minute parasitic wasps (parasitoids) in the aphelinid family. Female parasitoids lay their eggs inside the whitefly nymph or between the whitefly and the leaf surface, depending on the genus of wasp. The immature parasitoids develop within the whitefly host, eventually consuming the entire host, except the integument. The immature parasitoid pupates within the integument of the host and the adult parasitoid emerges through a round hole. The most common parasitoids attacking Bemisia in Florida are in two genera, Encarsia and Eretmocerus. Encarsia pergandiella (Howard) and Encarsia sophia (Girault & Dodd) (formerly E. transvena (Timberlake)) are common throughout the state, while Encarsia nigricephala Dozier is common in north-central Florida.
Several species of Eretmocerus are also common throughout the state. Eretmocerus species cause mortality to whiteflies by host-feeding in addition to parasitism. Females stab the immature whitefly repeatedly with their ovipositor and then turn around and feed from the wound, obtaining valuable protein with which to provision their eggs.
Bemisia populations can also be reduced by predation. Many species of insects, mites, and spiders feed on immature and adult whiteflies. Some well-known generalist predators that prey on whiteflies include Chrysoperla species larvae (lacewings), Orius species (minute pirate bugs), and Geocoris species (bigeyed bugs). Several coccinellid species are specialist whitefly predators, such as Delphastus catalinae (LeConte) and Nephaspis oculatus (Blatchley).
Under certain climatic conditions, usually wet and warm, Bemisia populations can be drastically reduced by natural epizootics of entomopathogens. Beauveria bassiana and Verticillium lecanii, two fungal pathogens with efficacy against Bemisia and other insects, have been commercialized and are available for field and greenhouse application against Bemisia.
In protected plant culture, such as greenhouses, it is not possible to rely on natural population regulation.
Several effective species of parasitoids, such as Eretmocerus eremicus, and predators, such as the predatory mite Amblyseius swirskii and the coccinellid D. catalinae, are available commercially for control of Bemisia.

Credit: Heather McAuslane, UF/IFAS

Credit: null

Credit: James Castner, UF/IFAS

Credit: James Castner, UF/IFAS

Credit: Heather McAuslane, UF/IFAS

Credit: Jack Dykinga, USDA

Credit: Jack Dykinga, USDA

Credit: Jack Dykinga, USDA
Chemical Control
Bemisia is difficult to control with insecticides for two reasons; adults and immatures infest the lower surfaces of leaves, which are difficult to reach with insecticide sprays; and they have developed resistance to many insecticide chemistries. Commercial growers of horticultural and ornamental crops rely on systemic insecticides such as the neonicotinoids (imidacloprid, thiamethoxam, dinotefuran) and diamides (cyazypyr) to manage whiteflies. See ENY-478 and ENY-735 for more information on management of whiteflies in commercial production. Homeowners would be best advised to treat populations before they reach outbreak levels and to spray immature whiteflies with a mixture of insecticidal soap or insecticidal oil in water (see label for directions). Care should be taken to cover the lower surface of the leaves. Other chemical control recommendations can be found at the following sites:
- Florida Insect Management Guide for Vegetables
- Florida Insect Management Guide for Field Crops
- Florida Insect Management Guide for Ornamentals

Credit: Janine Razze, UF/IFAS
Credit: Heather McAuslane, UF/IFAS
Selected References
Bellows TS, Jr., Perring TM, Gill RJ, Headrick DH. 1994. Description of a species of Bemisia (Homoptera: Aleyrodidae). Annals of the Entomological Society of America 87: 195-206. https://doi.org/10.1093/aesa/87.2.195
Brown JK, Frohlich DR, Rosell RC. 1995. The sweetpotato or silverleaf whiteflies: biotypes of Bemisia tabaci or a species complex? Annual Reviews of Entomology 40: 511-534. https://doi.org/10.1146/annurev.en.40.010195.002455
Csizinszky AA, Schuster DJ, Kring JB. 1997. Evaluation of color mulches and oil sprays for yield and for the control of silverleaf whitefly, Bemisia argentifolii (Bellows and Perring) on tomatoes. Crop Protection 16: 475-481. https://doi.org/10.1016/S0261-2194(97)00013-6
Evans GA, Hamon AB. 5 November 2002. Whitefly taxonomic and ecological Website: an online interactive catalog of the whiteflies (Hemiptera: Aleyrodidae) of the world and their parasites and predators. (https://www.fsca-dpi.org/homoptera_hemiptera/Whitefly/whitefly_catalog.htm ).
Gerling D. (ed.). 1990. Whiteflies: their Bionomics, Pest Status and Management. Intercept, Ltd., Andover, UK.
Gerling D , Mayer RT (eds.). 1996. Bemisia: 1995. Taxonomy, Biology, Damage, Control and Management. Intercept, Ltd., Andover, UK.
Hamon AB , Salguero V. 1987. Bemisia tabaci, sweetpotato whitefly, in Florida (Homoptera: Aleyrodidae: Aleyrodinae). Entomology Circular No. 292, Florida Department of Agriculture and Consumer Services, Division of Plant Industry.
Liu, T-X , Stansly PA. 1999. Searching and feeding behavior of Nephaspis oculatus and Delphastus catalinae (Coleoptera: Coccinellidae), predators of Bemisia argentifolii (Homoptera: Aleyrodidae). Environmental Entomology 28: 901-906. https://doi.org/10.1093/ee/28.5.901
Lopez-Avila A. 1986. Taxonomy and biology, pp. 3 - 11. In M. J. W. Cock (ed.), Bemisia tabaci - A Literature Survey on the Cotton Whitefly with an Annotated Bibliography. CAB International Institute of Biological Control, Ascot, UK.
Maynard DN, Cantliffe DJ. 1990. Squash silverleaf and tomato ripening: new vegetable disorders in Florida. Vegetable Crops Fact Sheet VC-37, Gainesville: University of Florida Institute of Food and Agricultural Sciences.
McAuslane HJ, Johnson FA, Colvin DL, Sojack B. 1995. Influence of foliar pubescence on incidence and parasitism of Bemisia argentifolii (Homoptera: Aleyrodidae) on soybean and peanut. Environmental Entomology 24: 1135-1143. https://doi.org/10.1093/ee/24.5.1135
Momol T. 4 March 1999. Tomato yellow leaf curl virus on tomato in north Florida and south Georgia. UF/IFAS Pest Alert. http://entomology.ifas.ufl.edu/pestalert/tmm-0304. htm.
Momol T, Funderburk J. 20 August 1999. Tomato yellow leaf curl virus on tomato: update from north Florida. UF/ IFAS Pest Alert. http://entomology.ifas.ufl.edu/pestalert/tmm-0820.htm.
Nyoike TW, Liburd OE, Webb SE. 2008. Suppression of whiteflies, Bemisia tabaci (Hemiptera: Aleyrodidae), and incidence of curcurbit leaf crumple virus, a whitefly-transmitted virus of zucchini squash new to Florida, with mulches and imidacloprid. Florida Entomologist 91:460-465. https://doi.org/10.1653/0015-4040(2008)91[460:SOWBTH]2.0.CO;2
Stansly PA, Smith HA, Seal DR, McAvoy E, Polston JE, Gilreath PR, Schuster DJ. 2015. Management of whiteflies, whitefly-transmitted plant virus, and insecticide resistance for vegetable production in southern Florida. ENY-735. Gainesville: University of Florida Institute of Food and Agricultural Sciences.
Tsai JH, Wang K. 1996. Development and reproduction of Bemisia argentifolii (Homoptera: Aleyrodidae) on five host plants. Environmental Entomology 25: 810-816. https://doi.org/10.1093/ee/25.4.810
Webb SE, Schuster DJ, Stansly PA, Polston JE, Adkins S., Baker C., Roberts P., Liburd O, Nyoike T., McAvoy E, Whidden A. 2011. Recommendations for management of whiteflies, whitefly-transmitted viruses, and insecticide resistance for production of cucurbit crops in Florida. ENY-478. Gainesville: University of Florida Institute of Food and Agricultural Sciences. https://doi.org/10.32473/edis-in871-2011
Wilkerson JL, Webb SE, Capinera JL. 2005. Vegetable Pests II: Acari - Hemiptera - Orthoptera - Thysanoptera. UF/ IFAS CD-ROM. SW 181. University of Florida.