The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
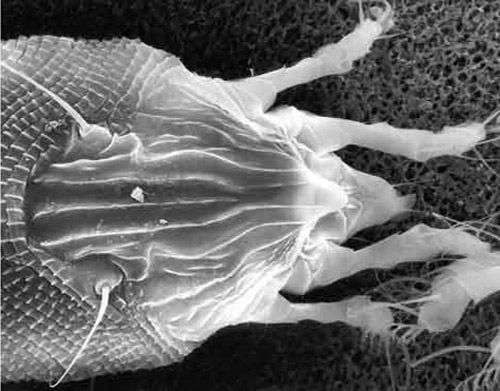
The coconut mite, Aceria guerreronis Keifer, attacks young fruits of the coconut palm, Cocos nucifera L. Although the mites are small (the largest stage is around 250 µm in length), populations can be extremely large and their feeding can cause scarring and distortion of fruit, which may cause premature fruit drop. It is one of the most serious arthropod pests of coconut palm, whether grown as a crop tree or as an ornamental. The coconut mite is distributed in many tropical countries where coconuts grow. In Florida, it is very prevalent on coconut palms in the Florida Keys, and occurs sporadically on the mainland.
Credit: Greg Erdos, University of Florida.
Three additional eriophyid mites occur on coconut palms in Florida, including Acathrix trymatus (Keifer), Acrinotus denmarki Keifer, and Amrinus coconuciferae (Keifer). These are found principally on the leaves, usually in low populations that do not cause significant damage. At least 12 eriophyid mite species are associated with coconut palms.
The vernacular name coconut mite has also been applied to both Acathrix trymatus and Raoiella indica Hirst (Tenuipalpidae) in addition to Aceria guerreronis. The latter species, which is highly destructive to coconut palm foliage, is native to southern Asia but was recently found on several Caribbean islands and could be a threat to coconut palms in Florida and throughout the region.
Distribution
The coconut mite was described by the eminent acarologist Hartford Keifer in 1965 from specimens collected in Guerrero, Mexico. The same year it was found near Rio de Janeiro, Brazil. Subsequently, it was found in many countries of Tropical America and in West Africa. It is controversial whether it is native to the Eastern or Western Hemisphere. Botanists have evidence that the coconut palm evolved in the South Pacific region and was spread along the coastal regions of Asia and into Africa. In the 1500s, Spanish and Portuguese colonists brought the coconut palm from West Africa to the American Tropics.
It seems unlikely that the coconut mite was introduced into the New World with the coconut itself and remained undetected for over four centuries. However, some coconut growers and Caribbean residents believe that damage typical of coconut mite was observed long before the mite was discovered. In 1984, when the species was positively identified for the first time in the continental United States by H. A. Denmark from specimens collected by F. W. Howard from coconuts on Sugarloaf Key, Florida, coconut mite damage was common in the Florida Keys and many residents had been familiar with it for years.
The most dramatic extension of the range of coconut mite in recent years occurred in the late 1990s, when it was found for the first time on coconuts in Tanzania (East Africa), India, and Sri Lanka.
Curiously, the coconut mite has not been reported in the South Pacific Region, which is the original home of the coconut palm.
Description
The adult female coconut mite is 205 to 255 µm long and 36 to 52 µm wide. These minute arthropods cannot be seen with the naked eye. Massive colonies of the mites and individual mites can be detected with a 10X hand lens. At this magnification, the colonies appear as vague silvery patches. Individual coconut mites appear small even when viewed under standard stereoscopic microscopes. Like typical eriophyid mites, they are elongate and have two pairs of legs, instead of four pairs as is typical of most mites. They are white and translucent.
Coconut mite infestations are generally diagnosed by their damage and confirmed by finding specimens on the fruits. Positive identification of the mite requires a microscope.
Biology and Ecology
The mites infest the abaxial (lower) surfaces of the perianth and the part of the fruit surface covered by the perianth. They penetrate between the tepals of the perianth and fruit surface a month after the fruit begins development; prior to this the tepals are too tightly appressed to allow entry of the mites. Presumably, a population is initiated by one or more fertilized females on the same or nearby plants.
The mites feed by piercing the superficial plant tissue and sucking the juices. A coconut mite develops from egg to adult in 10 days, so populations can build up rapidly, often producing thousands of mites in several aggregations on the same fruit. Massive populations of coconut mites may be present among the tepals and on the fruit surface beneath the perianth until about the sixth month of the coconut's development, after which populations decline. Coconuts mature in about 12 months, at which time few if any coconut mites are present, even in coconuts with extensive damage by these mites.
Credit: F. W. Howard, University of Florida.

Credit: F.W. Howard, University of Florida.
Coconut mites probably disperse from one palm to another on air currents or by phoresy (e.g., carried on insects or birds that visit palm flowers). Where coconut palms are dense, they can crawl from palm to palm. Their inefficient host-finding capabilities seem to be compensated for by a high reproductive rate.
In Florida, coconut mite infestations are generally more prevalent on the Florida Keys than on the Florida mainland. In a survey conducted from 1986 to 1987, 98% of the coconuts on some sites in the Keys were infested, while 0 to 8% of the coconut palms were infested on the mainland. This is probably due to environmental differences in the regions. The Keys receive about half the annual precipitation that southern Florida does, and some reports suggest the coconut mite prefers dry conditions. However, some researchers refute this; studies conducted at Bahia Honda in the Keys and Añasco, Puerto Rico, revealed no correlation between seasonal rainfall patterns and coconut mite populations.
Hosts
Coconut palm appears to be the only host of coconut mite, although there is an isolated confirmed record from Brazil on the fruit of Lytocaryum weddellianmum (H. A. Wendland), a palm native to South America that is related to coconut.
Coconut palm varieties differ in their susceptibility to coconut mite. Almost all varieties probably have some level of susceptibility.

Credit: F.W. Howard, University of Florida.
Damage and Economic Importance
The meristematic zone from which the growing coconut fruit expands is a circular whitish area covered by the perianth. The young fruits of about 2.5 to 3.0 cm (~1 to 1 1/8 in) in diameter develop to the mature coconut of up to 25 cm (~10 in) during the period of about one year. As the damaged surface expands from beneath the perianth and becomes exposed to air, it develops a brown cork-like surface with deep fissures. If mite feeding is concentrated on one side of the fruit meristem, growth of the fruit may be uneven. Severe damage results in the stunting of fruits.
Copra, a major product of the coconut industry, is the white kernel, or coconut "meat," after it is dried. In one study, coconut mite damage was found to cause a loss of up to 30% of the copra. Other researchers have reported a less serious impact on copra production. In many tropical countries, coconut water is a principal product. Coconut water is the clear liquid in the coconut and is sometimes erroneously called coconut milk (which is actually the paste made by grinding the kernel). In Puerto Rico about 10 million fresh coconuts are sold each year for coconut water. Data is not available on the possible impact of the coconut mite on the production of coconut water, but where this product is served in fresh coconuts, the unappealing appearance of mite-damaged coconuts adversely affects sales. A favorite motif in advertising tourist destinations in Florida and other tropical locales is a view of a beach with gracefully swaying coconut palms stretching towards the sea. Coconut palms are present in the designed landscapes of resort areas in these regions and are treasured by many homeowners. Damage by coconut mite is not noticeable from a distance and does not affect the aesthetic appearance of palms. As a pest of ornamental plants, coconut mite is most important to homeowners or managers of areas where palms are seen up close, such as in the landscaping around hotel swimming pools.

Credit: F.W. Howard, University of Florida.
Detection
Early-stage infestation of a young coconut is often detectable as a small, pale triangular area extending distally on the fruit surface from beneath the perianth. In other cases, a broader pale zone extends from the perianth. The pale areas turn brown in a matter of days. As infested coconuts develop, the damaged area continues to extend from beneath the perianth, eventually covering a large portion of the surface. Older damaged tissue is suberized (cork-like), with deep longitudinal fissures that may be intersected by horizontal cracks.

Credit: J. V. DeFilippis, University of Florida.

Credit: J. V. DeFilippis, University of Florida.
Young coconuts are green, yellow, bronze, light orange, or a blend of these colors, depending on variety. Mature coconut fruits are brown. Thus, the dark brown color of advanced coconut mite damage is most noticeable before the fruit has fully matured. Coconut mite damage can be spotted at a distance, but the diagnosis must be confirmed by closer examination. Browning of coconuts can be caused by factors other than coconut mites, including various forms of mechanical damage. For example, a petiole constantly rubbing against a coconut in the wind can cause browning over the affected area of the surface. A smooth brown surface may be the result of recent damage due to cold. In Florida and Puerto Rico, a second mite, Tarsonemus sp. (Acari: Tarsonemidae), causes damage similar in appearance to early coconut mite damage, but is rare. This mite occurs in populations of not more than a few hundred individuals per coconut and has been seen only on young coconuts. In a Florida study of coconuts with damage attributable to mites feeding beneath the perianth, 99% were found to be infested with coconut mite, and only 1% with Tarsonemus sp.
Management
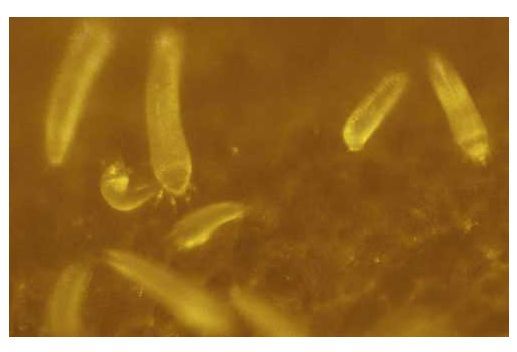
Predatory mites found beneath the coconut perianth in Florida and observed to prey on coconut mites include Amblyseius largoensis Muma, Neoseiulus mumai Denmark, and Neoseiulus paspalivorus DeLeon. In Puerto Rico, Bdella distincta Baker and Bablock preyed on coconut mite and on Steneotarsonemus furcatus (DeLeon) in the same habitat. In both of these localities, however, coconut mite infestations were heavy, implying that the effects of these predators were insignificant.
The fungus Hirsutella thomsonii (Fisher), which is widely distributed and known to attack various species of mites, has been isolated from coconut mites in various countries, as has Hirsutella nodulosa Petch in Cuba. Control of several species of mites with fungus has been developed and applied, but success has often depended greatly on environmental conditions. In general, these efforts have been most successful under humid conditions favoring the development of the fungi.
A simple mechanical form of control practiced by some farmers is to prune all of the coconuts in all stages of development. This is said to eliminate coconut mites at least temporarily, but obviously causes a disruption in production. The method may be a useful in situations such as in landscapes with high tourist activity, where coconuts are periodically pruned to prevent injury caused by falling coconuts.

Credit: F.W. Howard, University of Florida.
Field observations have indicated that in some situations there is an inverse relationship between water availability and damage levels of coconut mite. Other observations indicate that increased nutrient availability results in faster growth of coconuts so that they incur less coconut mite damage or tolerate it better. However, in other studies, increased nutrients seemed to increase the level of mite attack. More research is needed to determine an economically feasible cultural control of the coconut mite.
The coconut varieties most common in Florida and the Caribbean, viz., 'Jamaica Tall', 'Panama Tall', 'Malayan Golden Dwarf', 'Malayan Yellow Dwarf', and 'Malayan Green Dwarf' are all highly susceptible to coconut mite. Some varieties of coconut may be resistant to coconut mite. An apparently resistant Cambodian variety was reported in a research station in Africa. It was suggested that the round fruit of this variety resulted in a tighter perianth that excluded coconut mites. However, we have observed extensive damage of coconut mites on round-fruited coconut varieties in Florida and the Caribbean.
Acaricides have been tested for control of the coconut mite, and some have been shown to kill the mites. However, most chemicals applied topically had to be applied frequently and indefinitely to maintain control. Systemic acaricides might persist longer in the plant, but such chemicals could result in residues in the fruits, and coconuts are harvested throughout the year. Chemical control is perhaps the least viable option for control of coconut mite.
Selected References
Fernando LCP, Moraes GJ. Wickramananda IR. 2002. Proceedings of the International Workshop on Coconut Mite (Aceria guerreronis), Sri Lanka, 6–8 January 2000. Coconut Research Institute, Sri Lanka. 117 pp.
Howard FW, Abreu-Rodriquz E, Denmark HA 1990. Geographical and seasonal distribution of the coconut mite, Aceria guerreronis (Acari: Eriophyidae), in Puerto Rico and Florida, USA. Journal of Agriculture, University of Puerto Rico 74: 237–251.
Howard FW, Moore D, Giblin-Davis R, Abad R. 2001. Insects on palms. CABI Publications, Wallingford, UK.
Moore D. 2000. Non-chemical control of Aceria guerreronis on coconuts. Biocontrol News and Information, 21, 83N–87N.
Moore D, Howard FW. 1996. Coconuts. In Eriophyid mites—Their biology, natural enemies, and control. Lindquist EE, Sabelis MW, Bruin J. (eds.). Elsevier Science BV, pp. 561–569.