The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Most species of sap beetles are attracted to the wounds of trees where they feed on sap. However, the habits of the Nitidulidae are quite variable (Parsons 1943). Sap beetles have been found in various habitats feeding on flowers, fruits, sap, fungi, decaying and fermenting plant tissues, or dead animal tissue (Parsons 1943, Hayashi 1978).
Although there are many species of sap beetles, only several species are known agricultural pests of field and stored products. These include the dusky sap beetle Carpophilus lugubris Murray on field and sweet corn; the corn sap beetle, Carpophilus dimidiatus on field corn; the complex Carpophilus dimidiatus (F.), Carpophilus freemani Dobson, and Carpophilus mutilatus Erichson on stored maize (Arbogast and Throne 1997); the dried fruit beetle Carpophilus hemipterus (L.); the pineapple beetle, Urophorus humeralis; a picnic beetle, Glischrochilus quadrisignatus (Say); the strawberry sap beetle, Stelidota geminata (Say); and the yellowbrown sap beetle, Epuraea (Haptoncus) luteolus (Erichson) a pest of dried fruit. Sap beetles are often considered minor pests but the presence of large numbers of sap beetles on a host plant can prove economic in terms of crop damage caused by the feeding beetles, but impact on crop value is primarily due to the contamination of products ready for sale by adults and larvae. In addition to damage caused by feeding, sap beetles have also been recognized as vectors of fungi (Dowd 1991).

Credit: Ken Gray, Oregon State University

Credit: Keith Weller, USDA

Credit: Lyle J. Buss, University of Florida

Credit: Lyle J. Buss, University of Florida
Distribution
Sap beetles are widely occurring. More than half of the genera are cosmopolitan or nearly so (Parsons 1943). An equal number are American in distribution. The New World and two of the tropicopolitan genera appear to be relatively recent arrivals from the tropics into the United States. Six genera are endemic to North America. Twenty one genera have so far been reported in Florida. Agricultural pest species from the genera Carpophilus, Stelidota, and Glischrochilus are distributed throughout Florida. Epuraea luteolus is reported as an introduction to Florida and is widely distributed.
Description
Adults
Adult sap beetles are variable in size, 0.9 to 15 mm in length. The antennae are usually eleven segments with the distal three segments forming a club which makes them easily recognized. The clubbed portion, however, is quite variable within species, being quite distinct or only slightly developed (Parsons 1943). Antennal grooves are usually present. The elytra or wing covers are entire sometimes shortened to expose two or three abdominal segments. The dorsal surface usually has uniform punctation but sometimes punctures are of different sizes. The prosternum has a process produced between the front coxae. The tarsal formula is usually 5-5-5 and tarsi are five segmented. The first tarsal segment is of normal size, the fourth very small and the fifth the longest.
Abdominal segment VIII of males is heavily sclerotized, well raised and large. In females it is usually reduced and submembranous.
The dusky sap beetle adult, Carpophilus lugubris, is about 2.8 mm long with short elytra. The adult is uniform dull black in color.

Credit: Ken Gray, Oregon State University
Picnic beetles, Glischrochilus quadrisignatus and Glischrochilus fasciatus, are about 6.4 mm long black with four orange spots on the wing covers, or elytra.
The strawberry sap beetle adult, Stelidota geminata, is less than 2.8 mm, light to dark brown oval and somewhat flattened.
Eggs
Eggs are milky white, sausage shaped, and 0.8 mm long and 0.23 mm wide.
Larvae
Larvae of all three genera are white (pale yellow when mature) with a light brown head, opposing external mouthparts and three pairs of small true thoracic legs. The larval body is elongate, sub-cylindrical, somewhat dorsoventrally flattened, slightly sclerotized except for hard sclerotized epicranium. The body of the larvae bears few hairs and is equipped with hardened projections from the end of the abdomen that are species specific.

Credit: Ken Gray, Oregon State University
Pupae
Pupae are white, turning cream colored and later tan before adult emergence. The pupa is typical exarate (furrowed) averaging 4.4 mm in length and 2.0 mm in width (Parsons 1943).

Credit: Ken Gray, Oregon State University
Biology
Sap beetles are characterized by a rather short larval development and comparatively long lived adults. This allows the sap beetles to master and to adapt to extremely different types of substrates. In temperate regions most of the species hibernate beneath logs. In the tropics, multiple generations may occur, especially if there is available food material throughout the year. Information on sap beetle biology is known primarily from studies done in the north central United States, principally Illinois and Ohio where Carpophilus spp., Glischrochilus quadrisignatus, and Stelidota geminata are important pests.
The different species of Carpophilus are similar with respect to their biology. However, most work has been focused on the biology of Carpophilus lugubris. Investigations carried out by Sanford and Luckman (1963) showed that in Illinois, Carpophilus lugubris is very active in fall and overwinter as adults in soil or debris near the bases of trees or stumps. They become active around April or early May and are attracted to decomposing plant material or wounds in trees. The newly emerged females of the overwintering generation deposit eggs on or near decomposing plant material such as corn ears on or in the soil. Larvae feed on whatever is available when they emerge and eventually pupate in the soil. Adults of this first generation and those of the overwintering generation migrate in June to early sweet corn plantings shortly before or soon after it silks. Adults feed on the silk and fallen pollen from tassels. Wherever wet pollen collects on the plant such as the leaf whorls and axils they will feed and deposit eggs. The adults are also attracted to insect and plant volatiles produced by ear-wounding damage of the corn earworm Helicoverpa zea. It was observed that adults preferred to deposit eggs on earworm frass, earworm damaged corn ears, and even smut galls when compared with undamaged ears.

Credit: Gregg S. Nuessly, University of Florida
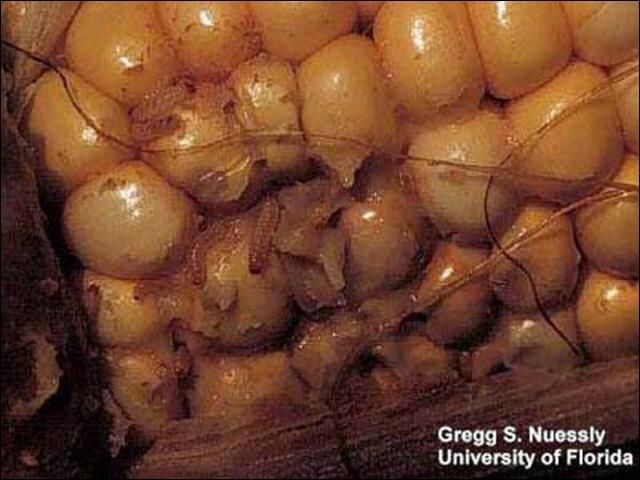
Credit: Gregg S. Nuessly, University of Florida
The dusky sap beetle is the predominant species on sweet corn. Larvae hatch within two to five days at 75°F (24°C). They pass through three instars in 14 days, drop to the ground where pupal cells are formed a few inches below the soil surface. In summer, 28 to 30 days elapse between egg deposition and adult emergence. From July to November all stages of the insect can be found in the fields of sweet and field corn and in some decomposing fruits and vegetables especially melons. Heaviest infestation occurred during the four days preceding harvest. If population densities are high adults are likely to oviposit in any corn ears that are available regardless of maturity or in the absence of damage.
The longevity and fecundity of Carpophilus lugubris adults varies. Male longevity was reported by Sanford (1963) to be 115.2 days on average. In the same study the life span of females was 101.3 days (range: 74 to 147 days). The average number of eggs laid per female was 99.5 (33 to 304 eggs).
Three to four generations have been reported in the latitude of Illinois and two in Ohio (Dowd and Nelson 1994). The number of generations encountered in the literature was found to vary and may be as result of differences in latitude, temperature, availability of suitable food sources, position of monitoring traps and some confusion with overlapping generations.
Glischrochilus quadrisignatus, commonly known as a picnic beetle, overwinters as an adult in Illinois. Hibernation sites are similar to that of Carpophilus lugubris. Picnic beetles become active on warm days in late winter or early spring. Most eggs are deposited in May. Females can oviposit up to 400 eggs in their lifetime. Females reproduce primarily near decomposing plant material. Eggs are deposited at random near decomposing plant material rather than on the material itself. To be suitable for oviposition and larval development, food material must be either buried in the soil or be in contact with the soil and it must be moist. Active adults of the new generation begin leaving the soil in June. They fly to fields of ripening or damaged berries, tree wounds, and corn. Adults live a long time, and in late June various life stages including both the new and overwintering generation can be found together in soil. In Illinois picnic beetles have been observed feeding on the silk and pollen of undamaged corn causing primary damage and allowing subsequent entry and oviposition by the dusky sap beetle. They also invaded fields that had corn stalks damaged by the European corn borer. They were secondary invaders in fields where ears were damaged by the Japanese beetle. Two generations were noted in Ohio the second occurring on field corn left in the field after harvest (Dowd and Nelson 1994).
Stelidota geminata, the strawberry sap beetle, migrates each spring from overwintering sites. Temperature plays a key role in regulating spring migration of Stelidota geminata (Gertz 1968, Weiss 1979). First generation beetles develop in the strawberry fields. Dowd and Nelson (1994) reported early migration peaks in both raspberry and corn at the end of the strawberry season which may indicate that these sites are utilized by the beetle for over-wintering. Stelidota geminata was not found to overwinter in strawberry plantings. However, Lobiopa insularis (Cast.) is the most frequently observed sap beetle in Florida strawberries (Price 2004), although several other smaller species inhabit the fields.

Credit: James F. Price, University of Florida

Credit: Lyle J. Buss, University of Florida

Credit: Lyle J. Buss, University of Florida
Okumura and Savage (Potter 1995) found that Epuraea luteolus has a more rapid development and mature larval development is attained after 11 days when placed in culture.
Host Range
Sap beetles have demonstrated a wide range of feeding habits with the majority of the group being primarily saprophagous and mycetophagous (Parsons 1943). Sap beetles of agricultural importance such as the dusky sap beetle, corn sap beetles (Carpophilus spp.), strawberry sap beetle (Stelidota geminata), and a picnic beetle (Glischrochilus quadrisignatus) have become abundant coincident with the expansion in production of suitable crop hosts in Florida. E. luteolus although a pest of dried fruit in California was reported as the dominant Nitidulid species on strawberry in Eastern Hillsborough county in Florida (Potter 1995).
These pest species generally feed on fruits and other plant parts that are ripening or decomposing. Their host range may include tree and small fruits such as peaches, figs, blueberries, raspberries, strawberries, pineapples, melons, field and sweet corn, stored corn, and dried fruit products. The species mentioned above are characterized by their affiliation with a specific host or group of related hosts. However, the beetles are not restricted in their host range.
Damage
Both direct and indirect damage can be caused by Carpophilus spp., Glischrochilus quadrisignatus, and Stelidota geminata (Luckmann 1963, Dowd 2000). Ear wounding of corn by primary insect pests such as the corn ear worm provides entry sites for sap beetles. The insect and corn volatiles associated with the damage are powerful attractants for the beetles. Dowd (2000) compared the incidence of sap beetle adults, larvae, and their damage on two varieties of corn, Bt corn and non-Bt hybrid corn. Dowd's study showed that direct damage can be induced by dusky sap beetles. In Ohio, the strawberry sap beetle attacks ripe, nearly ripe, or decaying strawberry fruit by boring into the berry and devouring a portion in the field. In the Florida study (Potter 1995) Epuraea luteolus apparently feeds on decomposing fruit material. The study however did not demonstrate the economic impact, if any, on strawberry fruit production. The presence of Epuraea luteolus on strawberry fruit may be more of an issue of contamination by beetles and possibly larvae.
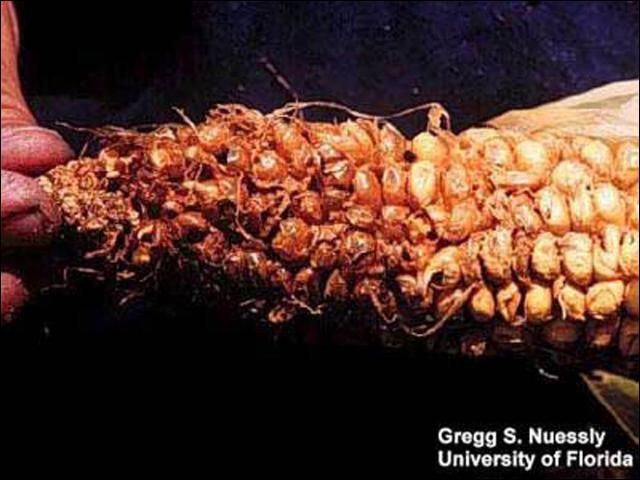
Credit: Gregg S. Nuessly, University of Florida

Credit: James F. Price, University of Florida
Sap beetles can also vector mycotoxin producing fungi to corn and strawberries (Dowd and Nelson 1994). Maize sap beetles appear to be well adapted for vectoring mycotoxigenic fungi, including species in the genera Aspergillus, Penicillium, and Fusarium. The adults feed on corn plant residues left in the field after harvest. These residues usually contain spores of Aspergillus or Fusarium, which not only damage crops but produce toxins harmful to humans and animals. They have also been implicated as vectors of forest pathogens causing wood rots (Peng and Williams 1991).
Some species, such as the dusky sap beetle, Carpophilus lugubris, and the beetles in the genus Glischrochilus, are implicated as vectors of tree diseases such as oak wilt, Ceratocystis fagacearum.
Management
Sap beetles are considered minor pests of field and sweet corn and strawberries in Florida. However high population levels may cause considerable damage, resulting in the spread of mycotoxin producing fungi that warrants their control.
Sanitation
Field sanitation appears to be an important means of control. It is best to harvest sweet corn, tomatoes, melons, berries, and other produce immediately they ripen. Damaged, diseased, and overripe fruits and vegetables should be removed from the area at regular intervals. Such material should be destroyed, or, if buried, should be buried deep below the soil.
Chemicals
Chemical treatment is recommended for the control of sap beetles in Florida. Previously, chemical control of corn earworm usually indirectly controlled sap beetle. However, in recent years chemicals suitable for corn earworm and fall armyworm control have not been effective for sap beetle control. With the advent of Bt corn to control corn earworm, one study has indicated that direct feeding damage of sap beetles on Bt corn is possible in the absence of corn earworm (Dowd 2000), hence warranting chemical intervention or a suitable management protocol for these pests. Several products are registered for use on corn and strawberries for the control of sap beetles. These pesticides can be incorporated in baits to trap incoming sap beetles.
Sap Beetle (Coleoptera: Nitidulidae) Management in Strawberries
Insect Management Guide for Sweet Corn
Baits
Research has shown that sap beetles are strongly attracted to certain volatile plant compounds in ripening or decaying fruits, and themselves produce pheromones/kairomones that elicit an aggregating behavior. Baits using such material can be effective in trapping and monitoring sap beetle populations, and hence determine when treatment is necessary.
Varietal Selection
Tight, long-husked corn varieties have been recommended for corn. These varieties are more resistant to corn earworms and the beetle itself and therefore are less likely to be infested by sap beetles. Varieties shown to be resistant to sap beetles include Country Gentleman, Golden Security, Tender Joy, Trucker's Favorite, Stowell's Evergreen, and Victory Golden. Though these varieties sustain less injury than susceptible varieties they are not immune to attack.
Biological Control
Research continues in the area of biological control. A nematode, Psammomermis nitidulesis, was found in living sap beetles collected near Illinois corn fields. Scientists believe that the nematode enters the body of the beetle in late summer when they are pupating in the soil. Scientists are studying ways to move infected sap beetles into areas where the nematode does not exist so the nematode can be used as a biological control agent. Researchers at Ohio State University evaluated a tiny parasitic wasp Brachyserphus abruptus for the control of the strawberry sap beetle. They are mass rearing the parasite for release at the time of strawberry fruit set. The parasite keeps the larva from maturing into an adult beetle, thereby eliminating reproduction and reducing subsequent sap beetle populations.
Selected References
Abogast RT, Throne JE. 1997. Insect infestation of farm stored maize in South Carolina; Towards characterization of a habitat. Journal of Stored Products Research 33: 187–198.
Dowd PF. 2000. Dusky sap beetle (Coleoptera: Nitidulidae) and other kernel damaging insects in Bt and non-Bt sweet corn in Illinois. Journal of Economic Entomology 93: 1714–1720.
Dowd PF, Nelson TC. 1994. Seasonal variation of sap beetle (Coleoptera: Nitidulidae) populations in central Illinois cornfield-oak woodland habitat and potential influence of weather patterns. Environmental Entomology 23: 1215–1223.
Dowd PF, Moore DE, Vega FE, Mcguire MR, Bratlet RJ, Nelson TC, Miller DA. 1995. Occurrence of a Mermithid nematode parasite of Carpophilus lugubris (Coleoptera: Nitidulidae) in Central Illinois. Environmental Entomology 24: 1245–1251.
Luckmann WH. 1963. Observations on the biology and control of Glischrochilus quadrisignatus. Journal of Economic Entomology 56: 681–686.
Lyons-Johnson D. 1997. Sap beetle has a nematode nemesis. Agricultural Research. Washington D.C.: Agricultural Research Service, United States Department of Agriculture 45, no. 4.
McCoy CE, Brindley TA. 1961. Biology of the four-spotted fungus beetle, Glishrochilus q. quadrisignatus and its effect on European corn borer populations. Journal of Economic Entomology 54: 713–717.
Miller KV, Williams RN. 1981. Seasonal abundance of Stelidota geminata (Say) in selected habitats. Journal of the Georgia Entomological Society 17: 112–117.
Nuessly GS, Pernezny K, Stansly P, Sprenkel R, Lentini R. (1999). Sap (picnic) beetles: Carpophilus spp., including corn sap beetle, C. dimidiatus, Nitidulidae. Florida Corn Insect Identification Guide. (no longer available online)
Parsons CT. 1943. A revision of Nearctic Nitidulidae (Coleoptera). Bulletin of Comparative Zoology 92: 121–248.
Peng C, Williams RN. 1990. Pre-oviposition period, egg production and mortality of six species of hibernating sap beetles (Coleoptera: Nitidulidae). Journal of Economic Entomology 23: 453–457.
Peng C, Williams RN. 1991. Influence of food on development, survival, fecundity, longevity and sex ratio of Glischrochilus quadrisignatus (Coleoptera: Nitidulidae). Environmental Entomology 20: 205–210.
Potter MA. 1995. The Nitidulidae (Coleoptera) associated with strawberry in eastern Hillsborough County, Florida. MSc. Thesis, University of Florida. 98 pp.
Price J. 2001. Sap beetles and fruit fly damage final fruit quality in March. Berry Times. Volume 1.
Price JF. (2004). Adult and larval large sap beetle (picnic beetle, nitidulid), Lobiopa insularis. (no longer available online)
Sanford JW, Luckman WH. 1963. Observations on the biology and control of the dusky sap beetle in Illinois. Proceedings North Central Branch Entomological Society of America 18: 39–43.
Schmidt CT. 1935. Biological studies on the Nitidulid beetles found in pineapple fields (Nitidulidae Coleoptera). Annals of the Entomological Society of America 28: 475–511.
Webber RG, Connel WA. 1975. Stelidota geminata (Say): Studies of its biology (Coleoptera: Nitidulidae). Annals of the Entomological Society of America 68: 649–653.
Weiss MJ, Williams RN. 1980. Some host parasite relationships of Microtonus nitidulidis. Annals of the Entomological Society of America 73: 323–326.
Williams RN, Weiss MJ, Miller KV, Werner JJ. 1984. A summary of experiments for control of sap beetles which attack fruit crops. Research circular - Ohio Agricultural Research and Development Center 283: 66–68.
Williams R, Fickle D, Ellis S. 1995. Strawberry sap beetle control parasite. Strawberry IPM Update 1: 9–10.
Windels CE, Windels MB, Kommedahl T. 1976. Association of Fusarium species with picnic beetles on corn ears. Phytopathology 66: 328–331.