The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Calleida decora (Fabricius) is a small arboreal ground beetle, predaceous both as larva and adult. Common on various cultivated crops, it is apparently the only carabid to complete its larval development on Florida soybean foliage (Neal 1974). It is believed to be a major factor in suppression of several lepidopterous pests, e.g., velvetbean caterpillar, Anticarsia gemmatalis Hübner, on soybeans.
Synonymys
Erwin et al. (1977) listed synonyms:
Calleida cordicollis Putzeys,
Calleida cyanoptera LeConte, and
Calleida coeruleipennis Gemminger & Harold.
Distribution
Calleida decora is reported from the southeastern United States, extending into the midwest, Baja California, Mexico, and Belize (Erwin et al. 1977).
Description
Adults
The adults are slender, 7 to 10 mm (0.3 to 0.4 in) long, 2.5 to 3.5 mm (~0.1 in) wide. Head and elytra are green or blue-black, thorax and legs yellowish-red with tips of femora and tarsi dark. Antennae are dark with three or four basal segments lighter.

Credit: Lyle Buss, UF/IFAS
Males have a double row of papillate hairs on the undersurface of the first three protarsal segments and first two metatarsal segments (appearing white), but female tarsi are pubescent (straw-colored) (Horn 1882; McWhorter et al. 1984).
Eggs
The eggs are round, white, semi-opaque, approximately 0.75 mm (0.03 in) in diameter, covered with sand particles, and attached by a silken thread to a leaf or other available surface such as a stem or twig.
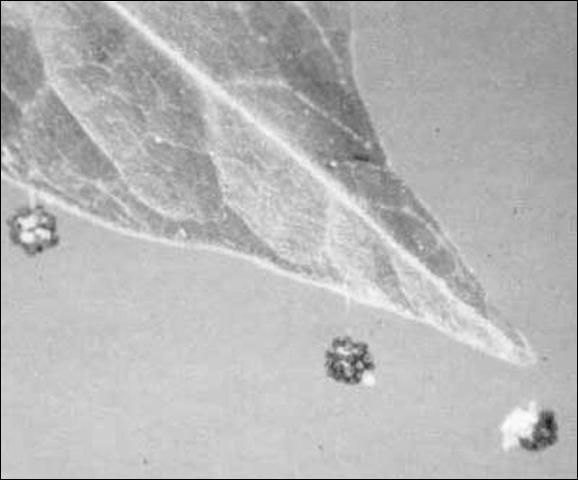
Credit: Shepard, reproduced from McWhorter et al. by permission of the editor, Journal of Agricultural Entomology.
Larvae
The larvae are active, black, campodeiform (in early stages having no abdominal appendages except cerci), with yellowish-red head capsules. Larvae are approximately 1.5 mm (0.06 in) long at hatching and may grow to 10 mm total body length prior to pupation. Mean head capsule widths for the three larval instars are 0.60, 0.90, and 1.23 mm (0.02, 0.04, and 0.05 in) respectively.

Credit: Lyle Buss, UF/IFAS
Pupae
The pupae are white, exarate and approximately 5 mm (0.2 in) long. The pupal cell is usually constructed ca. 7 to 15 mm (0.3 to 0.6 in) beneath the soil surface (Hasse 1971).
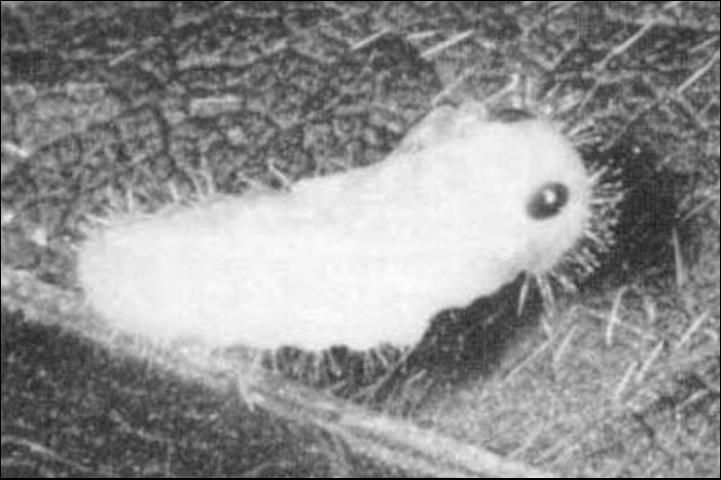
Credit: Shepard, reproduced from McWhorter et al. by permission of the editor, Journal of Agricultural Entomology.
Biology
Caged adult females live an average of 230 days with a mean preovipositional period of 11 days and lay an average of 800 eggs (McWhorter et al. 1984). While an egg is still held by the abdominal tip, the female covers it with sand or dust particles and binds it with silken thread to form a purse. The "egg purse" is attached to a leaf by a silken thread. Developmental times at 22°C to 28°C (71.6°F to 82.4°F) for eggs, larvae, and pupae are approximately four to six, 12 to 18, and four to six days, respectively (McWhorter et al. 1984; unpublished data 1973, 1982). Larvae are predaceous except while undergoing sclerotization following hatching and molting (McWhorter et al. 1984). They are highly cannibalistic and must be reared in individual containers. They feed readily on lepidoptera eggs as well as small larvae.
Economic Importance
On soybeans, populations were estimated as high as 5400/ha in Gadsden County, Florida (Neal 1974) and 9600/ha in Alachua County, Florida (Elvin 1983). Calleida decora adults and larvae have been observed feeding on velvetbean caterpillar, Anticarsia gemmatalis Hübner; cabbage looper, Trichoplusia ni Hübner; soybean looper, Pseudoplusia includens (Walker); and other lepidopterous larvae (Whitcomb and Bell 1964; McCarty et al. 1980; McWhorter et al. 1984; unpublished data 1973, 1982).
Over 10% of the total insect predation (almost 20% during one season) of Anticarsia gemmatalis larvae (1st to 4th instar) artificially placed on soybean foliage was by Calleida decora. Of the 21 predation observations involving Calleida decora during a total of four seasons, 19 were by larvae and two by adults (Elvin 1983; unpublished data 1974). Adult Calleida decora, confined in small field cages on potted soybeans, consumed an average of 6.4 small (1st to 3rd instar) Pseudoplusia includens larvae/24 hr (Richman et al. 1980).
Survey and Detection
Larval and adult populations on crops may be sampled by direct observation or examination, vacuum sampling, sweeping, shaking, or beating of the foliage. This species should not be confused with destructive leaf beetles of similar size and coloration.
Selected References
Elvin MK. 1983. Quantitative estimation of rates of arthropod predation on velvetbean caterpillar (Anticarsia gemmatalis Hübner) eggs and larvae in soybeans. Ph.D. Dissertation, University of Florida. 249 pp.
Erwin TL, Whitehead DR, Ball GE. 1977. Family 4 Carabidae. In Arnett RH Jr. Checklist of the beetles of North and Central America and the West Indies. Vo1.1, The ground beetles, water beetles, and related groups. Gainesville: Flora and Fauna Publications. 68 pp.
Hasse WL. 1971. Predaceous arthropods of Florida soybean fields. M.S. Thesis, University of Florida. 67 pp.
Horn GH. 1882. Synopsis of the species of the tribe Lebiini. Transactions of the American Entomological Society 10: 126–164.
McCarty MT, Shepard M, Turnipseed SG. 1980. Identification of predaceous arthropods in soybeans by using autoradiography. Environmental Entomology 9: 199–203.
McWhorter RE, Grant JF, Shepard M. 1984. Life history of a predator, Calleida decora, and the influence of temperature on development. Journal of Agricultural Entomology 1: 68–77.
Neal TM. 1974. Predaceous arthropods in the Florida soybean agroecosystem. M.S. Thesis, University of Florida. 194 pp.
Richman DB, Hemenway RC Jr, Whitcomb WH. 1980. Field cage evaluation of predators of the soybean looper, Pseudoplusia includens (Lepidoptera: Noctuidae). Environmental Entomology 9: 315–317.
Whitcomb WH, Bell K. 1964. Predaceous insects, spiders and mites of Arkansas cotton fields. Arkansas Agricultural Experiment Station Bulletin 690: 1–84.