The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The red palm mite, Raoiella indica Hirst, a pest of several important ornamental and fruit-producing palm species, has invaded the Western Hemisphere and is in the process of colonizing islands in the Caribbean, as well as other areas on the mainland.
Distribution
Until recently, the red palm mite was found in India, Egypt, Israel, Mauritius, Reunion, Sudan, Iran, Oman, Pakistan, and the United Arab Emirates. However, in 2004, this pest was detected in Martinique, Dominica, Guadeloupe, St. Martin, Saint Lucia, Trinidad, and Tobago in the Caribbean. In November 2006, this pest was found in Puerto Rico.
In 2007, the red palm mite was discovered in Florida. As of April 2009, this pest was in five counties: Broward, Miami-Dade, Martin, Monroe, and Palm Beach (FDACS 2009).
It is spreading rapidly and is expected to establish in other subtropical regions of the Western Hemisphere.
This mite is easily distributed by wind currents and movement of infested plants through nursery stock and cut branches of plants. It is likely to establish throughout tropical and subtropical areas throughout the Western Hemisphere.
Description and Life Cycle
Mites in the family Tenuipalpidae are commonly called "false spider mites" and are all plant feeders. However, only a few species of tenuipalpids in a few genera are of economic importance. The tenuipalpids have stylet-like mouthparts (a stylophore) similar to that of spider mites (Tetranychidae). The mouthparts are long, U-shaped, with whiplike chelicerae that are used for piercing plant tissues. Tenuipalpids feed by inserting their chelicerae into plant tissue and removing the cell contents. These mites are small and flat and usually feed on the under surface of leaves. They are slow moving and do not produce silk, as do many tetranychid (spider mite) species.
Adults
Females of Raoiella indica average 245 microns (0.01 inches) long and 182 microns (0.007 inches) wide, are oval and reddish in color. Females develop dark markings on the dorsum of the body after feeding. The dorsum is smooth, except for the presence of punctae (sculptured depressions). The male is smaller, but similar to the female in shape except for having a tapering of the posterior end of the body.

Credit: Rita Duncan, University of Florida
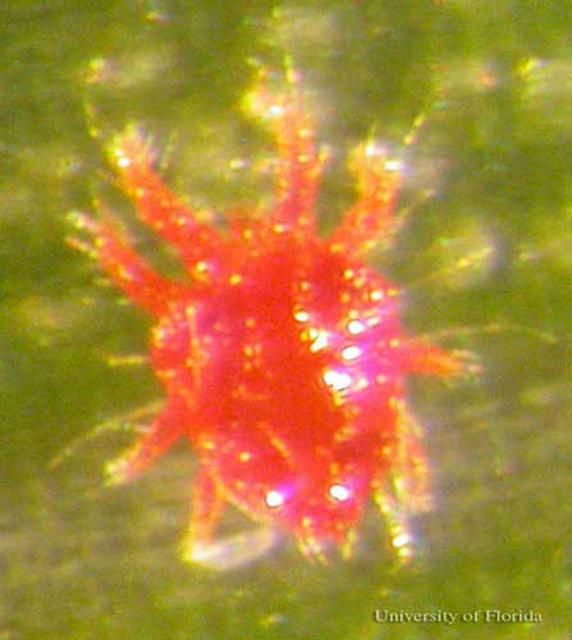
Credit: Rita Duncan, University of Florida
Adult females are larger than males and less active. Males and females are sexually mature when they emerge and males actively seek out females, suggesting there is a sex pheromone involved. When a male locates a female deutonymph in the quiescent stage, he will settle close to it and wait for up to two days for her to molt. When female deutonymphs begin to molt, the male becomes active and moves under her, bending his posterior up and forwards to mate. Mites remain in the mating posture for about 16 minutes.
The life cycle from egg to adult typically requires 23 to 28 days for females and 20 to 22 days for males. Mated females have a 5- to 6-day preoviposition period and oviposit for 47 days under laboratory conditions. Unmated females deposit an average of 18 eggs after a 2-day preoviposition period, oviposit for 40 days and live for ca. 48 days. Males, produced by unmated females, live an average of 22 days.
Egg
The ovoid egg is reddish, ca. 100 microns long (0.003 inches), and 80 microns wide. The freshly laid egg is attached to the leaf surface and a fine white stipe (slender hairlike structure) as long as or longer than the egg is present at one end. The tip of the stipe may be coiled and have a droplet of water clinging to it. The egg turns opaque white about 24 hours before hatching. The incubation period averages eight days for fertilized eggs and 7.3 days for unfertilized eggs. According to Chandra and Channabasavanna (1984), this species has an unusual genetic system, in which all eggs laid by unmated females develop into males and mated females produce only female progeny.
Larva
The newly hatched larva is red and has three pairs of legs. A blackish tinge may develop on the posterior end of the dorsum after feeding. The larva typically feeds for three to five days and then becomes quiescent for 1.7 to 1.9 days before molting to the protonymphal stage.
Protonymph
The reddish protonymph emerges with four pairs of legs and feeds for two to five days prior to becoming quiescent. The quiescent phase lasts from one to four days before deutonymphs emerge from the exoskeleton (exuvium). The female protonymph has an ovoid body with a rounded posterior but the male protonymph has a pointed posterior and a nearly triangular body.
Deutonymph
Deutonymphs are larger than protonymphs but resemble protonymphs with regard to feeding and other habits. The active phase lasts two to five days and the subsequent quiescent phase lasts from two to four days.
Hosts
The red palm mite is a pest of several ornamental and fruit-producing palm species such as coconut and areca palms, and has been found attacking bananas and plantains in the Caribbean. Ornamental palms, including hurricane or princess palm (Dictyosperma album) and Christmas palm (Veitchia merrillii), have also been damaged. Palms are important components of our tropical landscapes, both indoors and out, and in many countries coconut palms, Cocos nucifera, and date palms, Phoenix dactylifera, are important food crops.
The red palm mite has been collected from ornamental plants other than palms (such as heliconia, Heliconia rostrata R., bird of paradise, Strelitzia reginae Banks, gingers, Alpinia purpurata Vieill., and Etlingera elatior (Jack.)) in the Caribbean islands where it has invaded, but it is not clear whether these are valid host plants or whether the enormous mite populations on coconuts and other palms have temporarily moved on to substory plants under the palms. Detailed studies on the reproduction of this mite on plants other than palms have not been completed. In any case, the red palm mite is expected to cause economic damage to tropical and subtropical agriculture and to urban and indoor environments where palms are grown in the Western Hemisphere. Palm nurseries, landscape palms, and horticultural gardens will be affected by this new pest, as well.
Survey, Detection, and Damage
The reddish mites are easily seen against green leaves. Heavy infestations of the mites are typically on the lower surface of the leaves, and yellow speckles and blotches on the leaves are seen from the feeding damage. Yellowing of the leaves may often be severe. These mites do not produce webbing.
Palms affected by Raoiella indica show scattered yellow spots on both leaflet surfaces to a strong yellowish coloration on the entire leaflet, with most of the leaflets affected located in the middle area of the leaf. Coconut palms severely affected by the mite showed entirely yellow leaves, particularly on the lower third region of the plant. It is unknown if this condition is solely the result of mite feeding in combination with the dry season or the presence of a plant pathogen transmitted by the mite; there is no information as to whether this mite is a disease vector. Very young coconut palms to very old palms (more than 50 feet tall) can be severely affected, with most of their lower leaves yellow. Young coconut plants may be the most affected by the feeding of the mite. The yellow color of the leaflets is followed by the abortion of the flowers or small nuts in coconut palms.
In less than a year after its introduction to Martinique, the mite appeared on coconut palms on nearby islands. The presence of Raoiella indica populations on very tall and older coconut palms in St. Lucia strongly support the premise that the mite was primarily dispersed by wind currents. The mite is spreading to other exotic and ornamental palms. In Dominica, where coconut is grown with bananas, the mite shifted to this new host with numbers close to 200 mites/cm2. The lower leaves of banana and plantain turn yellow with small patchy green-yellow areas. Most of the mites are on the lower leaf of the host plant and the presence of yellow coloration on the upper surface of the leaf corresponds to the presence of a mite colony on the underside of the leaf.

Credit: Jorge Pea, University of Florida

Credit: Jorge Pea, University of Florida

Credit: Jorge Pea, University of Florida
Mites may be collected by visual examination of the individual plant or plant part under a dissecting microscope or by beating the plants over a sieve screen fitted to a plastic funnel with a vial attached to it. Both methods allow tenuipalpids as well as predatory mites (Phytoseiidae) to be collected.
In India, populations of Raoiella indica are negatively affected by rainfall and high relative humidity and populations are highest during hot, sunny and dry conditions on palms, so it is likely that populations will be found more readily during hot, dry periods in the Western Hemisphere.
Management
Chemical
Chemical control is difficult and expensive when the palm trees are very tall. However, chemical control may be needed to manage these mites if they occur in palm nurseries without adequate natural enemies present. At present, it is unclear what chemicals are appropriate, although oil and sulfur are common miticides that should be tested for their effectiveness.
Biological
It is common that invasive arthropods are more serious pests in their new environment than in their original home because there are fewer natural enemies that can suppress them. Several natural enemies exist in India, Mauritius and Egypt, where predators have been cited as important, but only a few studies have been conducted to resolve which are effective.
In India, the predatory mite Amblyseius channabasavanni Gupta (Acari: Phytoseiidae) is known to feed on Raoiella indica and developed rapidly and reproduced at a high rate (2.7 eggs per day) on this food (Daniel 1981). In Mauritius, the phytoseiid Amblyseius caudatus Berlese is considered the "main predator" of Raoiella indica on coconut palms (Moutia 1959). Ueckermann (2004) found the phytoseiid Amblyseius largoensis (Muma) "in association" with Raoiella indica, but did not indicate how effective it was as a natural enemy. Whether these phytoseiids have a narrow prey range currently is unknown. Many phytoseiid species feed on pollen, fungal spores, and honeydew when prey is not available and only a few are obligatory predators. Of those that are obligatory predators, many will feed on several species of mites, including tetranychids, tenuipalpids, eriophyids, or tydeids, when available.
In addition to phytoseiid predators, the lady beetle Stethorus tetranychi Kapur (Coleoptera: Coccinellidae) is known to feed voraciously on Raoiella indica in Kerala, India, preferring eggs followed by larvae, nymphs and adults. Likewise, Stethorus parcempunctatusi Puttarudria and Channabasavanna has been reported to feed on Raoiella indica in Karnataka, India, as has Stethorus karalicus. Stethorus lady beetles are known to be effective, and relatively specific, predators of spider mites and false spider mites, but require relatively high prey densities to maintain population growth.
Native natural enemies in the Western Hemisphere may provide some control of the red palm mite. For example, it is possible that coccinellids endemic to the Western Hemisphere will feed on this abundant new prey. Other natural enemies native to the Western Hemisphere also may attack the red palm mite. The phytoseiid Neoseiulus longispinosus (Evans), an Old World species probably native to the Asian region, was found attacking Raoiella indica in the Caribbean. In Florida, a number of phytoseiid species are known to prey on tenuipalpid mites (Muma and Denmark 1970), and should be evaluated as natural enemies of Raoiella indica once it is here. Likewise, it is likely that endemic fungal pathogens, such as Hirsutella, will be effective against this pest, at least during rainy seasons.
A great deal is unknown about the biology and host range of this invasive pest, so it is difficult to predict the long-term impact of the red palm mite in the Western Hemisphere. Efforts are under way, however, to develop more information and responses to this pest.
Selected References
Chandra BKN, Channabasavanna GP. 1984. Development and ecology of Raoiella indica Hirst (Acari: Tenuipalpidae) on coconut. pp. 785-790. In Griffiths DA, Bowman CE (eds.). Acarology VI. Vol. 2, Ellis Horwood Publishers, Chicester, UK.
Daniel M. 1981. Bionomics of the predaceous mite Amblyseius channabasavanni (Acari: Phytoseiidae) predaceous on the palm mite. pp. 167-172. In Channabasavanna GP (ed.). Contributions to Acarology in India. Bangalore, Anubhava Printers.
Etienne J, Fletchmann CW. 2006. First record of Raoiella indica (Hirst, 1924) (Acari: Tenuipalpidae) in Guadeloupe and Saint Martin, West Indies. International Journal of Acarology 32: 331-332.
FDACS-DPI. Red Palm Mite Raoiella indica Hirst (Acari: Tenuipalpidae). http://www.freshfromflorida.com/Divisions-Offices/Plant-Industry/Pests-Diseases/Red-Palm-Mite (February 2019).
FDACS. (May 2009). Protecting Florida palms: Effectively managing red palm mites in nursery environments. FDACS-Division of Plant Industry. (no longer available online).
Flechtmann CHW, Etienne J. 2004. The red palm mite, Raoiella indica Hirst, a threat to palms in the Americas (Acari: Prostimagata: Tenuipalpidae). Systematic and Applied Acarology 9: 109-110.
Gupta SK. 1991. The mites of agricultural importance in India with remarks on their economic status. pp. 509-522. In Dusbabek F, Bukva V (eds.). Modern Acarology Vol. 1, Academia Prague and SPB Academic Publ. Bv, The Hague.
Gupta SK. 1998. A conspectus of natural enemies of phytophagous mites and mites as potential biocontrol agents of agricultural pests in India. pp. 484-497. In Acarology: Proceedings of the 10th International Congress, Halliday RB, Walter DE, Proctor HC, Norton RA, Colloff MJ (eds). CSIRO Publications, Melbourne, Australia.
Hirst S. 1924. On some new species of red spider. Annals and Magazine of Natural History 14: 522-527.
Jagadish PS, Nagesha Chandra BK. 1982. Biology of Typhlodromips tetranychivorus (Acari: Phytoseiidae) on red palm mite Raoiella indica (Acari: Tenuipalpidae) Acarological Newsletter (1982) (11): 9-10.
Jagadish PS, Nagesha Chandra BK. 1979. Biology of Typhlodromips tetranychivorus (Acaari: Phytoseiidae) on red palm mite Raoiella indica (Acari: Tenuipalpidae) in Contributions to Acarology in India, Proceedings of the First All India Symposium on Acarology, Apr. 23-25, 1979, Bangalore, ChanaBasavanna GP, (ed.). Acarology Society of India, Bangalore, India. pp 173-179.
Jeppson LR, Keifer HH, Baker EW. 1975. Mites Injurious to Economic Plants. University of California Press, Berkeley. 614 pp.
Kane EC, Ochoa R. (May 2006). Detection and Identification of the red palm mite Raoiella indica Hirst (Acari: Tenuipalpidae). UF/IFAS Pest Alert. (no longer available online).
McMurtry JA 1984. A consideration of the role of predators in the control of acarine pests. 109-121 pp. In Griffiths DA, Bowman CD, (eds.). Acarology VI. Vol. 1, Ellis Horwood Publishers, Chicester, UK.
Muma MH, Denmark HA. 1970. Arthropods of Florida and Neighboring Land Areas. Phytoseiidae of Florida. Vol. 6, Contribution No. 148. Bureau of Entomology, Florida Department of Agriculture and Consumer Services, Division of Plant Industry. 149 pp.
Nangia N, ChannaBasavanna GP. 1984. The biology of Typhlodromips tetranychivorus Gupta (Acari: Phytoseiidae) on selected plant feeding mites of economic importance. pp. 725-732, In Griffiths DA, Bowman CE, (eds.), Acarology VI. Vol. 2, Ellis Horwood Publishers, Chicester, UK.
Osborne LS. (2009). (May 2009). Red Palm Mite. Entomology and IPM: MREC. http://mrec.ifas.ufl.edu/lso/RPM/RPM.htm (February 2019).