Introduction
The small hive beetle, Aethina tumida Murray, is native to sub-Saharan Africa where it is an occasional pest of African subspecies of the western honey bee, Apis mellifera Linnaeus (Hymenoptera: Apidae). However, the beetle has been found outside of its native range where it can cause considerable damage to colonies of European subspecies of honey bees (http://entnemdept.ufl.edu/creatures/MISC/BEES/euro_honey_bee.htm). Because of its rapid spread, the small hive beetle is studied increasingly, both for its economic importance and biological significance.
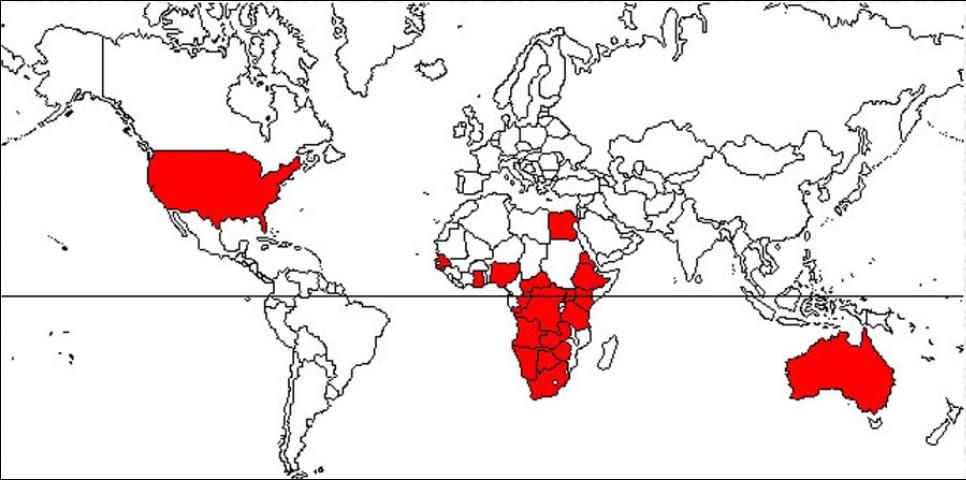
Distribution
Small hive beetles were confirmed in the southeastern United States in 1998 in a commercial apiary in Florida, but previously unidentified specimens indicate its presence in the US since at least 1996. The small hive beetle initially appeared in port cities such as Charleston, South Carolina and Savannah, Georgia, as well as parts of Florida, and since has spread to much of the US.
Small hive beetles were found in the port cities of Sydney and Brisbane, Australia, in the early 2000s and have spread within Australia since that time. The small hive beetle also was found in Portugal, but it was eradicated quickly after its detection.
Description
Eggs
Small hive beetle eggs are 1.4 x 0.26 mm (l x w) and pearly white in appearance.
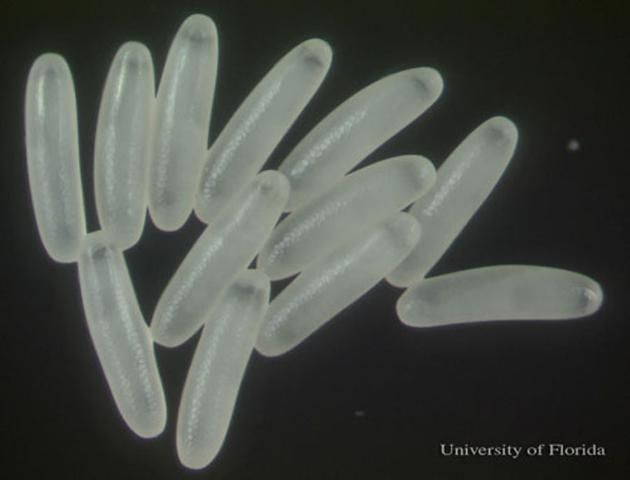
Credit: Josephine Ratikan, UF/IFAS
Larvae
Newly emerged small hive beetle larvae have relatively large heads and numerous protuberances covering their bodies. Upon full maturation, larvae will have reached a length and width of 9.5 mm and 1.6 mm, respectively.

Credit: Josephine Ratikan, UF/IFAS

Credit: Josephine Ratikan, UF/IFAS
Pupae
Once they reach maturity, beetle larvae drop from the hive and burrow into the soil where they pupate. Early-stage pupae of small hive beetles are pearly white, having characteristic projections on the thorax and abdomen. Later-stage pupae darken as their exoskeleton develops and hardens.

Credit: Lyle J. Buss, UF/IFAS
Adults
Newly emerged adult small hive beetles are light brown, becoming progressively darker (almost black) as their exoskeleton sclerotizes (hardens). These color changes may occur in the pupal cell before the adult emerges. Adult female (5.7 ± 0.02 mm) small hive beetles generally are longer than males (5.5 ± 0.01 mm) but both are nearly identical in width (about 3.2 mm). Adult females (14.2 ± 0.2 mg) are also heavier than males (12.3 ± 0.2 mg) and occur in greater proportions of the population. Naturally occurring small hive beetles can vary greatly in size, possibly depending on diet, climate, and other environmental factors.

Credit: Lyle J. Buss, UF/IFAS

Credit: Josephine Ratikan, UF/IFAS
Life Cycle
Although not fully known, small hive beetle biology is becoming better understood (Ellis and Hepburn 2006; Hood 2004). Upon emerging from the ground (where they pupate), adult small hive beetles search for honey bee colonies, probably identifying the host colony by a suite of olfactory cues. Investigators have shown that small hive beetles fly before or just after dusk and odors from adult bees and various hive products (honey, pollen) are attractive to flying small hive beetles (Elzen and Neumann 2004). Some investigators have suggested that small hive beetles also may find host colonies by detecting the honey bee alarm pheromone (Elzen and Neumann 2004). Additionally, small hive beetles carry a yeast (Kodamaea ohmeri) on their bodies that produces a compound very similar to honey bee alarm pheromone when deposited on pollen reserves in the hive.
Upon locating and entering the host colony, adult small hive beetles seek out cracks and crevices where they hide from bee aggression. These locations are often referred to as 'prisons' (Ellis 2005). Remarkably, honey bees station guards around the prisons where small hive beetles hide. The 'prison guard' bees keep the small hive beetles confined to the cracks and out of the brood combs where there is an ample supply of honey, pollen, and brood on which small hive beetles reproduce. Small hive beetles do not starve in these prisons as they are able to solicit food from their bee captors. In this behavior, small hive beetles use their antennae to rub the bees' mandibles and induce the bees to regurgitate. Small hive beetles then feed on the regurgitated food (Ellis 2005; Ellis and Hepburn 2006).
Mating behavior of small hive beetles (including whether female small hive beetles mate once or multiple times) is not understood, but adult small hive beetles do not appear to be sexually mature until about one week post-emergence from the soil. If allowed to reproduce, female small hive beetles will oviposit directly onto food sources such as pollen or brood combs. Alternatively, female small hive beetles may deposit irregular masses of eggs in crevices or cavities away from the bees as female ovipositors are long and flexible, being perfectly designed to lay eggs in tiny and concealed places. A female small hive beetle may lay 1,000 eggs in her lifetime, although data suggest that the number of eggs produced in one female's lifetime might be upwards of 2,000 (Hood 2004). The majority of these eggs hatch within three days; however, some eggs are still viable and hatch after five days. Humidity appears to be a crucial factor influencing hatch rates as small hive beetle eggs are prone to desiccation if exposed to circulating air and relative humidity below 50%.
Newly hatched larvae immediately begin feeding on whatever food source is available, including honey, pollen, and bee brood, though they have demonstrated a preference for bee brood. Maturation time for larvae is generally 10–16 days, although some may feed longer than a month. Once the larvae finish feeding, a wandering phase is initiated where larvae leave the food source and migrate out of the colony to find suitable soil in which to pupate. It is believed that the majority of larvae do this at night in the cover of darkness (Ellis and Hepburn 2006).
Larvae in the wandering stage may wander great distances from the hive to find suitable soil. Despite this, most larvae pupate within 90 cm of the hive. Nearly 80% of the larvae burrow down into the soil less than 10 cm from the soil surface but not generally more than 20 cm. Once larvae cease burrowing, they construct a smooth-walled, earthen cell in which they pupate. The period of time spent in the ground pupating can vary greatly depending on factors such as soil temperature, moisture, and composition. However, the majority of adults emerge from the soil in approximately three to four weeks. Upon adult emergence, the entire life cycle begins again. The turnover rate from egg to adult can be as little as four to six weeks; consequently, there may be as many as six generations in a 12-month period under moderate climatic conditions.
Economic Importance
In its native range, the small hive beetle regularly occurs in honey bee colonies but it does not cause damage in strong, healthy colonies. In the US and Australia though, small hive beetle damage in European colonies follows a characteristic pattern:
-
adult invasion into colonies
-
population build-up of small hive beetles
-
reproduction of small hive beetles
-
significant damage to brood, pollen, and honey stores by feeding small hive beetle larvae
-
mass exodus of larvae from the colony
-
pupation in the soil, and
-
emergence as adults and subsequent re-infestation of colonies.
There is fermentation of hive products (particularly honey) associated with feeding larvae. This likely occurs due to specific yeasts associated with the small hive beetle. Honey damaged by small hive beetles is rendered foul and unfit for human consumption. Colonies heavily infested with adult small hive beetles may abscond (entirely leave the nest), although the number of adults needed to elicit this behavior often must be high (>1000 small hive beetle adults/colony).
Small hive beetle damage to living colonies is not the only loss experienced by beekeepers. Adult and larval small hive beetles can be a significant problem in unprocessed honey crops stored in the honey house. As a result, beekeepers realize the necessity of extracting honey quickly and moving the equipment out of honey houses to discourage ensuing build-up of small hive beetle larvae. Further, stored supers of honey or supers containing pollen residues are prime targets for small hive beetle reproduction and subsequent damage.

Credit: Mark Dykes, UF/IFAS
Despite the fact that small hive beetles may cause considerable damage in bee colonies in the US, they increasingly are considered a secondary pest of bee colonies, much like the greater (Galleria mellonella Linnaeus) and lesser (Achroia grisella Fabricius) wax moths (Ellis and Hepburn 2006). Colonies hosting other bee diseases/pests appear more likely to suffer small hive beetle damage than healthy ones.
Management
Chemical Control
Since the introduction of small hive beetles into the US, little progress toward developing chemical control methods has been made (Hood 2004). The two available chemical controls for small hive beetles are GuardStar™ a ground drench used around colonies to kill pupating beetles, and Checkmite+™ a plastic strip used under pieces of cardboard in colonies to kill adult beetles. As is the case for all insecticides, it is important to follow the product label when using these products to control small hive beetles.
For controlling small hive beetles, more attention has been given to cultural, biological, and genetic control possibilities.
Cultural and Mechanical Controls
Cultural/mechanical controls result from a change in practice with the intention of limiting, but not eradicating, a pest. Practices such as removing honey, bits of comb and cappings from around the honey house will minimize foodstuffs to which small hive beetles may be attracted. It is also important to extract supers of honey quickly to reduce the damage that small hive beetle adults and larvae do to standing, unprotected crops. Reducing the relative humidity to 50% in honey houses and other places where honey is stored inhibits small hive beetle eggs from hatching. In the apiary, one should eliminate, requeen, or strengthen weak colonies to reduce colony stress and to make the colony better able to deal with small hive beetles. One should avoid other conditions that might lead to colony stress such as brood diseases, mite problems, wax moth activity, failing queens, excessive swarming, and over-supering.
At least five in-hive trapping devices have been developed for small hive beetle control. They range from the Hood beetle trap (which fits in a standard hive frame and is filled with apple cider vinegar to attract and mineral oil to drown adult small hive beetles) to the West beetle trap which is designed to rest on the bottom board of a colony. Others exist as well but they are based typically on the same principle: provide an attractant (often apple cider vinegar) and a killing agent (mineral oil) in a trap to attract and kill adult small hive beetles. The traps vary in efficacy but provide some control of small hive beetle adults.
Biological Control
Other control measures are being developed. For example, two species of soil-dwelling nematodes have demonstrated activity against pupating small hive beetles. These include Steinernema riobrave and Heterorhabditis indica. Additionally, researchers have shown that some honey bee colonies are able to detect and remove brood that has been oviposited on by small hive beetles (Ellis 2005). This behavior, called hygienic behavior, can be selected for in breeding programs and can help reduce small hive beetle problems. In conclusion, small hive beetles will be controlled best by integrating a number of these control methods. For example, one may consider using hygienic queens (bees remove beetle eggs and young larvae), in-hive traps (catch adult beetles), and nematodes around colonies (kill pupating beetles) to attack all beetle life stages. While no single beetle control boasts 100% efficacy, employing multiple control strategies may limit beetle populations to manageable levels.
For more information on small hive beetles and beetle control, visit http://www.UFhoneybee.com/ and view our free educational videos.
Selected References
Ellis JD. 2005. "Reviewing the confinement of small hive beetles (Aethina tumida) by western honey bees (Apis mellifera)." Bee World 86: 56–62.
Ellis JD, Hepburn HR. 2006. "An ecological digest of the small hive beetle (Aethina tumida), a symbiont in honey bee colonies (Apis mellifera)." Insectes Sociaux 53: 8–19.
Elzen PJ, Neumann P. 2004. "The biology of the small hive beetle (Aethina tumida, Murray): Gaps in our knowledge of an invasive species." Apidologie 35: 229–247.
Hood WM. 2004. "The small hive beetle, Aethina tumida: A review." Bee World 85: 51–59.