The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The genus of widow spiders, Latrodectus, has limited records of the pathogens, parasites, and parasitoids known to affect them.
In the United States, a known associated parasitoid is Philolema latrodecti (Fullaway). Little is documented regarding this species, but it is confirmed to target multiple widow spider species as hosts for reproduction (Boucek 1988, Fullaway 1953).
Another parasitoid of note is a scelionid wasp, Baeus latrodecti Dozier, which was recorded as emerging from the egg sacs of the southern black widow, Latrodectus mactans Fabricius (Dozier 1931).

Credit: Christopher S. Bibbs, University of Florida
Synonymy
Philolema latrodecti was originally described as Eurytoma latrodecti (Fullaway 1953). It was later moved into the new genus Desantisca with other egg sac parasitoids that were originally described in the genus Eurytoma (Burks 1971). Research to phylogenetically reclassify the large family Eurytomidae using morphological differences placed it into the genus Philolema (Lotfalizadeh et al. 2007).
Baeus latrodecti was described twice, once as B. latrodecti (Dozier 1931) and again as B. californicus (Pierce 1939, Krombein et al. 1979).
Neither P. latrodecti nor B. latrodecti have an official common name sanctioned by the Entomological Society of America.
Distribution
Philolema latrodecti was originally described from several collections in Hawaii, and was found during collections of brown widow, Latrodectus geometricus (Koch), egg sacs from Honolulu and Oahu during 1945 and 1946 (Fullaway 1953). Cataloguing of spider parasitoids in Florida has revealed its presence in the continental United States (Brambila & Evans 2001). The species appears to occupy warmer climate zones (Boucek 1988, Herting 1971, Fullaway 1953), and the known widow hosts have a wider distribution, but any movement in the United States since discovery in Florida has not been recorded. For the U.S., distribution information is incomplete and more verifible records are necessary to provide the range of P. latrodecti since introduction into the contiguous 48 states.
Philolema latrodecti has also been observed outside of the United States, emerging from L. hasselti Thorell egg sacs in Queensland, Australia, and the island chains near the Philippines (Boucek 1988, Herting 1971).
For B. latrodecti, the 1979 Catalogue of Hymenoptera in America North of Mexico indicates occurrence in California, Hawaii, Kansas, Louisiana, Maryland, Missouri, South Carolina, and Texas (Krombein et al. 1979).
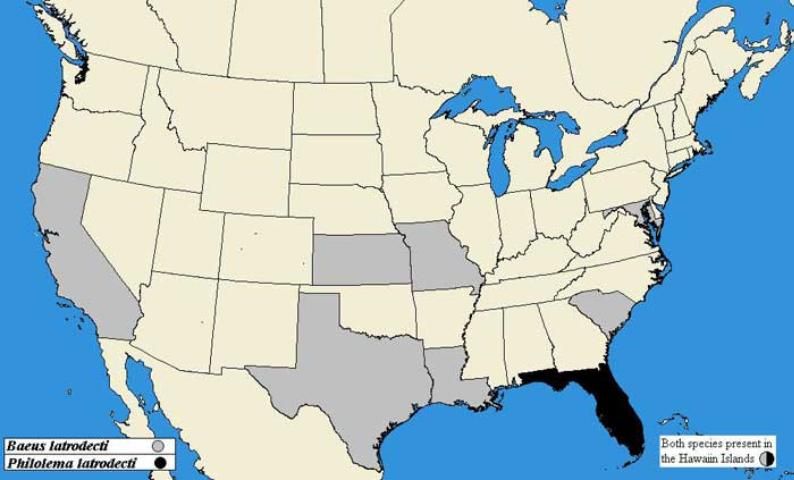
Credit: Map based on Brambila & Evans 2001, Fullaway 1953, and Krombein et al. 1979
Description
Immatures
Eggs and larvae of both species have no formal descriptions to date.
Adults
Philolema latrodecti (Fullaway)
Philolema latrodecti adults appear as small, black, ant-like or wasp-like organisms. The antennae are less than half of the body length, appearing short and thick.

Credit: Christopher S. Bibbs, University of Florida
Under magnification, the abdomen is seen as approximately equal to or shorter than the thorax. The abdomen appears smooth and polished with a rounded surface. The head and thorax are black, but silvery hairs dull the luster. The head and thorax are rough in texture, which is in contrast to the smooth texture of the abdomen. The body is black, but the leg segments below the femur are significantly lighter in color.

Credit: Christopher S. Bibbs, University of Florida
Males and females are difficult to distinguish. Females average 1.5–2.0 mm in length and have a slightly protruding ovipositor at the tip of the abdomen. Their antennae have a 3-segmented club. Males average 1.0–1.75 mm in length, with a 2-segmented antennal club. The petiole joining the abdomen to the thorax is longer in males. The terminal end of the male abdomen is telescoped, resulting in a short, somewhat tapered tip.

Credit: Christopher S. Bibbs, University of Florida
Baeus latrodecti Dozier
Baeus latrodecti adults average less than 1.0 mm in body length. Antennae appear to have a two jointed club because of a constriction in the middle of the club. Males have wings, but females are wingless with stronger hind legs and stouter body; the eyes of both sexes are black. Body color ranges from a honey yellow to a dark orange.

Credit: Lyle J. Buss, University of Florida

Credit: Lyle J. Buss, University of Florida
Hosts and Fecundity
Philolema latrodecti (Fullaway)
Observational data regarding either obligate egg parasitoid are limited. Laboratory observations in Honolulu and Oahu detailed the emergence of P. latrodecti from the egg sacs of the southern black widow, L. mactans, and the brown widow, L. geometricus Koch (Fullaway 1953). Philolema latrodecti has also been documented emerging from the egg sacs of L. geometricus in Florida (Brambila & Evans 2001). Outside of the United States, this parasitoid has been recorded emerging from the egg sacs of the redback spider, L. hasselti (Boucek 1988).
Rearing by Lyle Buss from an L. geometricus egg sac collected in Gainesville, Florida, in 2010, yielded 16 wasps for the single egg sac. Rearing by Christopher Bibbs from seven L. geometricus egg sacs collected in Gainesville, Florida, in 2010, yielded 13–16 wasps per egg sac. Four L. mactans egg sacs collected in Balm, Florida, in 2011, yielded 15–17 wasps per egg sac.

Credit: Lyle J. Buss, University of Florida
Based on these records, P. latrodecti can use multiple members of the genus Latrodectus as a host. Other U.S. species of interest are the red widow, Latrodectus bishopi (Fabricius), the northern black widow, Latrodectus variolus Walkenaer, and the western black widow, Latrodectus hesperus Chamberlin & Ivie.
Baeus latrodecti Dozier
Baeus latrodecti was recorded emerging from egg sacs of black widows in southern California and the island of Haiti, and was introduced into Hawaii (Bianchi 1945, Dozier 1931, Pierce 1939). After oviposition, emergences occur within seven days; the number of parasitoids per emergence was not indicated. Dozier (1931) and Pierce (1939) described the parasitoid as emerging from L. mactans. It has not been documented since the formal description separating L. hesperus from L. mactans (Chamberlin and Ivie 1935) as to whether or not B. latrodecti will emerge from egg sacs of L. hesperus.
Selected References
Bianchi FA. 1945. Notes on the abundance of the spiders Lactrodectus mactans, L. geometricus and Argiope avara, and of their parasites on the Island of Hawaii. Proceedings of the Hawaiian Entomological Society 12: 245-247.
Boucek Z. 1988. Australasian Chalcidoidea (Hymenoptera): A Biosystematic Revision of Genera of Fourteen Families, with a Reclassification of Species. CAB International, Wallingford, Oxon, U.K., Cambrian News Ltd; Aberystwyth, Wales. 108 pp.
Brambila J, Evans GA. 2001. Hymenopteran parasitoids associated with spiders in Florida. Insecta Mundi 15: 18.
Burks BD. 1971. A synopsis of the genera of the family Eurytomidae (Hymenoptera: Chalcidoidea). Transactions of the American Entomological Society 97: 1-89.
Chamberlin RV, Ivie W. 1935. The black widow spider and its varieties in the United States. Bulletin of the University of Utah 25: 1-29.
Dozier HL. 1931. A new scelionid egg parasite of the black widow spider. Proceedings of the Entomological Society of Washington 33: 27-28.
Fullaway DT. 1953. Three new species of Eurytoma (Hymenoptera: Eurytomidae). Proceedings of the Hawaiian Entomological Society 15: 33-36.
Herting B. 1971. Arachnida to Heteroptera: A Catalogue of Parasites and Predators of Terrestrial Arthropods. Section A. Host or Prey/Enemy. Commonwealth Agricultural Bureaux, Slough, England. 1. 129 pp.
Krombein KV, Hurd PD, Smith DR, Burks BD. 1979. Catalog of Hymenoptera in America North of Mexico. Vol. 1 Smithsonian Institution Press, Washington, D.C. 1160 pp.
Lotfalizadeh H, Delvare G, Rasplus JY. 2007. Phylogenetic analysis of Eurytominae (Chalcidoidea: Eurytomidae) based on morphological characters. Zoological Journal of the Linnaean Society 151: 441-510.
Pierce WD. 1939. The black widow spider and its parasites. Bulletin of the Southern California Academy of Sciences 37: 101-104.