The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
Doryctobracon areolatus (Szépligeti, 1911), formerly Parachasma cereum (Gahan), is a parasitoid of Anastrepha spp. in the Neo- and subtropics (Ovruski et al. 2000). It was introduced into Florida and the Dominican Republic for control of the Caribbean fruit fly, Anastrepha suspensa (Loew) (Diptera: Tephritidae), and the West Indian fruit fly, A. obliqua (Macquart) (Baranowski et al. 1993; Serra et al. 2011) (see Host Table below).
Distribution
Doryctobracon areolatus is the most widely distributed, Neotropical/subtropical, larval-prepupal parasitoid of Anastrepha (Ovruski et al. 2000; López et al. 1999). Its range extends from Florida (where it was introduced in 1969) deep into South America (Sivinski et al. 1997). At one time, it was abundant in the Florida peninsula to well north of Lake Okeechobee (Eitam et al. 2004). Recently, its numbers appear to have declined, perhaps from competition with other fruit fly parasitoids and/or climate change.
Description
Adult
Doryctobracon areolatus is a larval-prepupal synovigenic (produce eggs over the life of the adult), endoparasitic koinobiont (parasitoid allows the host to continue development and does not kill the host until the parasitoid larva pupates) that develops particularly well in 2nd instar larvae (Wharton and Marsh 1978). The adult body coloration is yellow to orange with clear wings, and the apical abdominal tergites in males are often black (Wharton and Marsh 1978). The labrum is usually visible and the clypeus is relatively short compared to some of the other Doryctobracon species, with an ovipositor length of ~3.8 mm (Sivinski and Aluja 2003).

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
There is a very distinctive banding pattern on the hind tibia.

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
The Cu2 submarginal cell of the forewing is 4-sided (Wharton and Marsh 1978; Sivinski et al. 2001) and this shape distinguishes it from the two other opiine braconid wasps attacking A. suspensa in Florida.
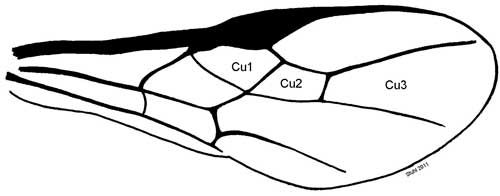

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
Life Cycle
Doryctobracon areolatus forages for larvae in ripe fruit on the tree and, unlike some related species, seldom investigates fallen fruit. The females are attracted to fruit volatiles in their search for food and fly hosts. Host location within the fruit is mediated by antennation (sensing information by touching antennae) and perhaps probing with the ovipositor to detect compounds unique to larval hosts (Stuhl et al. 2011b). The adult female inserts a single egg inside the body of the fly larvae. Upon hatching, the parasitoid larva remains in the first instar stage until the host pupates. The development time from egg to adult parasitoid is temperature dependent, but usually takes about two weeks.
Adult foods consist of fruit juices expelling from ovipositor-wounded or infested fruit (Stuhl et al. 2011a) and other plant-produced substances such as extrafloral nectar and hemipteran honeydew. Fruit juice consumption allows the parasitoid to forage for both food and hosts in the same habitat and thus eliminates the expense and danger of separate forays to locate carbohydrates (Stuhl et al. 2011a).
Hosts
Some fruit fly hosts and fruit fly host plant species of Doryctobracon areolatus (Aluja et al. 2000; Aluja et al. 2003).
Economic Importance
Doryctobracon areolatus parasitism rate is highly dependent on fruit size (the larger the fruit the more difficult it is to reach hosts). In its native habitats, parasitism of fruit flies in certain fruit can reach >80%. Mean parasitism of Caribbean fruit fly following original establishment in Florida was ~40% and, while it is too early to tell for releases in the Dominican Republic (Serra et al. 2011), parasitism is already similar to that exerted by the native parasitoid Utetes anastrephae.
Selected References
Aluja M, Piñero J, López J, Ruiz C, Zuniga A, Piedra E, Díaz-Fleischer F, Sivinski J. 2000. New host plant and distribution records in Mexico for Anastrepha spp., Toxotrypana curvicauda Gerstacker, Rhagoletis zoqui Bush, Rhagoletis sp., and Hexachaeta spp (Diptera : Tephritidae). Proceedings of the Entomological Society of Washington 102: 802–815.
Aluja M, Rull J, Sivinski J, Norrbom L, Wharton RA, Macías-Ordoñez R, Díaz-Fleischer F, López M. 2003. Fruit flies of the genus Anastrepha (Diptera: Tephritidae) and associated native parasitoids (Hymenoptera) in the tropical rainforest biosphere reserve of Montes Azules, Chiapas, Mexico. Environmental Entomology 32: 1377–1385.
Baranowski R, Swanson R. 1970. Introduction of Parachasma (=Opius) cereus (Hymenoptera: Braconidae) into Florida as a parasite of Anastrepha suspensa (Diptera: Tephritidae). Florida Entomologist 53: 161–162.
Baranowski R, Glenn H, Sivinski J. 1993. Biological control of the Caribbean fruit fly (Diptera, Tephritidae). Florida Entomologist 76: 245–251.
Eitam A, Sivinski J, Holler T, Aluja M. 2004. Biogeography of braconid parasitoids of the Caribbean fruit fly (Diptera: Tephritidae) in Florida. Annals of the Entomological Society of America 97: 928–939.
Gahan AB. 1919. Descriptions of seven new species of Opius (Hymenoptera-Braconidae) Proceedings of the Entomological Society of Washington 21: 161–170.
López M, Aluja M, Sivinski J. 1999. Hymenopterous larval pupal and pupal parasitoids of Anastrepha flies (Diptera: Tephritidae) in Mexico. Biological Control 15: 119–129.
Ovruski S, Aluja M, Sivinski J, Wharton R. 2000. Hymenopteran parasitoids of fruit-infesting Tephritidae (Diptera) in Latin America and the southern United States: Diversity, distribution, taxonomic status and their use in fruit fly biological control. Integrated Pest Management Review 5: 81–107.
Serra A, Ferreira M, García S, Santana L, Castillo M, Nolasco C, Morales P, Holler T, Roda A, Aluja M, Sivinski J. 2011. Establishment of the West Indian fruit fly (Diptera: Tephritidae) parasitoid Doryctobracon areolatus (Hymenoptera: Braconidae) in the Dominican Republic. Florida Entomologist 94: 809–816.
Sivinski J, Aluja M. 2003. The evolution of ovipositor length in the parasitic Hymenoptera and the search for predictability in biological control. Florida Entomologist 86: 143–150.
Sivinski J, Aluja M. López M. 1997. Spatial and temporal distributions of parasitoids of Mexican Anastrepha species (Diptera: Tephritidae) within the canopies of fruit trees. Annals of the Entomological Society of America 90: 604–618.
Sivinski J, Vulinec K, Aluja M. 2001. Ovipositor length in a guild of parasitoids (Hymenoptera: Braconidae) attacking Anastrepha spp. fruit flies (Diptera: Tephritidae) in southern Mexico. Annals of the Entomological Society of America 94: 886–895.
Sivinski J, Rendón P, Holler T, Bloem K, Martínez A, López Valenzuela M. 2000. Distribution of hymenopterous parasitoids of Anastrepha spp. in guava (Psidium guajava) along an altitudinal transect in the Soconusco region (Chiapas, Mexico): practical implications for a potential augmentative biocontrol program with native parasitoids, pp: 115–116. In 4th Meeting of the Working Group on Fruit Flies of the Western Hemisphere, Mendoza, Argentina, Nov. 25–30.
Stuhl C, Cicero L, Sivinski J, Teal P, Lapointe S., Paranhos B, Aluja M. 2011a. Longevity of multiple species of tephritid (Diptera) fruit fly parasitoids (Hymenoptera: Braconidae: Opiinae) provided exotic and sympatric-fruit based diets. Journal of Insect Physiology 57: 1463–1470.
Stuhl C, Sivinski J, Teal P, Paranhos B. Aluja M. 2011b. A compound produced by fruigivorous Tephritidae (Diptera) larvae promotes oviposition behavior by the biological control agent Diachasmimorpha longicaudata (Hymenoptera: Braconidae). Environmental Entomology 40: 727–736.
Wharton RA, Marsh PM. 1978. New world Opiinae (Hymenoptera: Braconidae) parasitic on Tephritidae (Diptera). Journal of the Washington Academy of Science 68: 147–167.